23 April 2021: Clinical Research
Effect of Improving Dietary Structure on Serum 25(OH)VitD Level in Hypertensive Patients in a Chinese Rural Area
Jia Wang1BCDE, Nan Wang1F, Zhiwen Ge1F, Bingjie Ding1F, Yanxia Bi1F, Wei Li1F, Zhongxin Hong1A*DOI: 10.12659/MSM.929218
Med Sci Monit 2021; 27:e929218
Abstract
BACKGROUND: In this study, we explored the effect of an improved dietary pattern on 25-hydroxyvitamin D3 [25(OH)VitD₃] level in hypertensive patients in a rural setting and investigated the possible mechanism for lowering blood pressure.
MATERIAL AND METHODS: Thirty-three participants with overweight or obesity were randomly divided into 2 groups. A 24-h dietary review method was used for dietary assessment. Participants in both groups were provided the same balanced diet except for the breakfast staple food for 4 weeks. General characteristics, body composition analysis, blood pressure, serum lipid profile, inflammatory indices, and serum 25(OH)VitD₃ level were assessed at baseline and at the completion of the 4-week dietary intervention. Nutrient intake was analyzed by dietary analysis software, and SPSS software was used for correlation and regression analyses.
RESULTS: Thirty-one participants completed the study. The prevalence of vitamin D deficiency was 100% at baseline. After the dietary intervention, the prevalence of vitamin D deficiency decreased to 45%. Levels of dietary intake of vitamin D, cholesterol, protein, fat, and calcium showed a significantly positive correlation with the serum 25(OH)VitD₃ level. Body fat, visceral fat, waist-to-hip ratio, serum triglyceride, and Toll-2 expression were negatively correlated with the serum 25(OH)VitD₃ level. Blood pressure had a significant negative correlation with 25(OH)VitD₃ level. Participants in both groups experienced a significant decrease in blood pressure.
CONCLUSIONS: Intake of a balanced diet rich in vitamin D, with appropriate amounts of cholesterol, protein, calcium, and fat, helped improve body composition, ameliorated lipid metabolism disorder, reduced inflammation, and improved serum 25(OH)VitD₃ level, thus lowering blood pressure.
Keywords: Blood Pressure, Body Composition, Hypertension, Nutritional Physiological Phenomena, Asians, Calcifediol, Diet, Diet Therapy, Lipid Metabolism, Prevalence, Rural Population, Vitamin D Deficiency
Background
Vitamin D is involved in the regulation of cardiovascular function, mental health, muscle function, and brain function [1–3], in addition to its well-recognized role in regulating calcium and phosphorus metabolism and bone transformation. Recent studies have demonstrated the relation of vitamin D with cancer [4] and type 2 diabetes mellitus [5]. In our previous studies, hypertension showed a close relation with increased body weight and changes in blood lipid profile and inflammatory indices. An increase in the inflammatory indices can increase the blood pressure level, while dietary interventions to decrease inflammatory indices can decrease the blood pressure level [6]. Recent studies have also shown a close relation of 25-hydroxyvitamin D3 [25(OH)VitD3] with increased body weight and changes in blood lipid profile and inflammatory indices [4,7–9]. Previous studies have also demonstrated the relationship of 25(OH)VitD3 deficiency with cardiovascular diseases (especially hypertension) [10,11]. A decreased serum level of 25(OH)VitD3 was shown to be an independent risk factor for cardiovascular diseases, and its level was negatively related to blood pressure [12]. According to a meta-analysis, an increase in the serum 25(OH)VitD3 level by 16 ng/mL may reduce the risk of hypertension by 16% [12]. A dietary intervention study on people with overweight or obesity entailed energy reduction of 15% for up to 4 weeks. After 2 weeks of the dietary intervention, the decrease in body fat and inflammatory indices and increase in serum 25(OH)VitD3 level were associated with a significant decrease in systolic blood pressure (SBP) [8]. When the energy intake was reduced by 20%, the decrease in body fat was more obvious if the 25(OH)D3 level was >20 ng/mL [13]. In a 12-month-long energy-reducing dietary intervention study, weight loss in participants was associated with a concomitant increase in 25(OH)VitD3 level; this outcome indicated a relation between body weight and 25(OH)VitD3 level [14]. However, all the above studies were aimed at improving the level of 25(OH)VitD3 by simply reducing the energy intake, which led to the changes in body weight, body fat, inflammatory indices, and SBP. However, no studies have investigated the change in serum 25(OH)VitD3 level in hypertensive patients due to improved dietary structure. In this study, improving the dietary pattern of participants led to a change in many nutrients. We observed a correlation between the nutrients and the level of 25(OH)VitD3; subsequently, we identified the nutrients that can significantly influence the level of 25(OH)VitD3. Furthermore, we observed the correlation of 25(OH)VitD3 with body composition, blood lipids, inflammatory indices, and blood pressure. The purpose of this study was to explore the effect of a reasonable improvement in dietary structure on the serum 25(OH)VitD3 level and to investigate the possible mechanism by observing changes in body composition, blood lipid profile, and inflammatory indices, as well as the effect on blood pressure. Our findings may provide a practical and feasible dietary pattern for prevention and treatment of hypertension.
Material and Methods
RESEARCH PARTICIPANTS:
From August 21, 2017, to September 30, 2017, 33 individuals with overweight or obesity and grade 1 hypertension who met inclusion criteria were selected from a database of 245 middle-aged and elderly (age range, 45–75 years) villagers residing in Zhang Fang village, Fangshan district, Beijing. In a series of studies conducted over the last 8 years, the prevalence of hypertension in middle-aged and elderly villagers was found to be significantly higher than that in their urban counterparts. Moreover, this group had a relatively homogenous dietary pattern. Therefore, the study population was suitable for this intervention study.
Inclusion criteria were (1) body mass index (BMI) >24.0 kg/m2 (overweight: 24< BMI <27.9 kg/m2; obese: BMI >28 kg/m2) [15]; (2) SBP 140–159 mmHg (1 mmHg=0.133 kPa) and/or diastolic blood pressure (DBP) 90–99 mmHg [15]; (3) diagnosis of grade 1 hypertension and regular use of antihypertensive drugs; and (4) provision of written informed consent after obtaining detailed information about the diet contents and provision of a 4-week diet.
Exclusion criteria were (1) hepatic or renal insufficiency; (2) cognitive impairment; (3) pregnancy or lactation; (4) alcoholism (consumption of >150 g of liquor or >500 mL of beer per day); (5) mental disorders; (6) being unable to take antihypertensive drugs regularly or to adjust antihypertensive drugs; (7) lactose intolerance or inability to consume dairy products; (8) intolerance or allergy to the anti-atherosclerosis cereal powder; (9) being vegetarian or semivegetarian; (10) traveling during the intervention and being unable to guarantee compliance with the dietary intervention and testing; (11) amputation or hemiplegia; (12) infectious diseases; and (13) regular use of vitamin D supplements.
Participants were randomly allocated to 2 groups using a random number table. We selected 33 series numbers from the random number table representing the 33 participants. Odd numbers were assigned to group A, and even numbers were assigned to group B. Group A had 17 participants and group B had 16 participants. The study was conducted according to the ethical principles of the Ministry of Health’s “Ethical Review Measures for Biomedical Research Involving Human Beings (Trial)” (2007), the State Food and Drug Administration’s Quality Management Standard for Clinical Trials of Drugs (2003) and Regulations on Clinical Trials of Medical Devices (2004), the World Medical Association’s Declaration of Helsinki, and Council for International Organizations of Medical Sciences’ International Ethical Guidelines for Biomedical Research of Human Beings. The study protocol was approved by the Ethics Committee of the Beijing Friendship Hospital affiliated with the Capital Medical University (ethics certificate number: 2017-P2-078-01). Written informed consent was obtained from all participants prior to their enrollment. This study is registered in the China Clinical Trial Registration Center (registration no. ChiCTR1800017457).
RESEARCH PROCESS:
On August 21, 2017, general information was collected from participants, including sex, age, smoking status (participants who smoked ≥1 cigarette per day were defined as smokers), drinking (consumption of any kind of alcoholic beverage per day was defined as drinking), sleep time, and exercise time (daily time spent in heavy physical labor and deliberate physical exercise was defined as exercise time).
On August 28, 2017, we organized a formal training session for villagers wherein they were informed in detail about the research objectives and the protocol. All consenting individuals underwent a trial of anti-atherosclerosis diet powder to rule out any allergy or intolerance to its ingredients. Thereafter, written informed consent was obtained for participation in the study.
From August 29, 2017, to September 02, 2017, the villagers were instructed to eat regularly every day and to maintain a record of their dietary intake (including the type and quantity of food) and activities during the 3-day period. This period was the adaptation period.
On September 3, 2017, before the intervention (T0) began, blood pressure, body composition, and blood lipid profile of the participants were measured, and a detailed dietary survey was conducted [16]. Group A received a standard balanced diet, while group B received a balanced diet supplemented with the anti-atherosclerosis diet powder.
On September 30, 2017, at the completion of the 4-week intervention (T4), participants’ blood pressure, body composition, and blood lipid profile were again measured, and the detailed dietary survey was repeated [16]. We also recorded the compliance of villagers with the dietary intervention and any drug adjustment. The research protocol is presented in Figure 1.
At completion of the intervention, 1 participant was unable to comply with the dietary intervention due to personal reasons and 1 participant was hospitalized due to an oral disease. The remaining 31 participants received the meals and underwent the required blood tests and physical examinations.
THE DIETARY PLAN:
Group A received a standard balanced diet including 220 g of white flour, 30 g of coarse cereals, 200 g of fruits, 243 mL of whole-fat milk, 1 egg, 150 g of white meat/red meat, 1000 g of vegetables, and 25 mL of nontransgenic soybean oil. Group B received a balanced diet supplemented with the anti-atherosclerosis diet powder; 77 g of anti-atherosclerosis diet powder was provided instead of 120 g of white flour (the energy content in 77 g anti-atherosclerosis diet powder was equivalent to that in 120 g white flour). The rest of the dietary content in group B was identical to that in group A. A national invention patent has been obtained for the anti-atherosclerosis diet powder (invention patent no. 201310706278.6), which contains rye, black kerneled rice, black soya bean, American ginseng, walnut, lotus nut, Chinese yam, and jujube. More details about the anti-atherosclerosis diet powder are available elsewhere [17]. The total daily energy intake during the intervention was approximately 1720 kcal. The total daily energy intake before the intervention was 1500–1700 kcal. The energy intake during the intervention was not significantly different from the pre-intervention energy intake. Moreover, residents in this village typically perform physical labor and require more energy. Therefore, our energy supply used an energy factor of 30–35, and the energy intake was weight times the 30–35 factor.
BLOOD PRESSURE MEASUREMENT METHOD:
We used a medical-grade electronic sphygmomanometer (HEM-907, Japan Omron Matsuzaka Co. Ltd.) for measurement of blood pressure (including SBP and DBP). Prior to measurement, the participants were required to sit quietly for 5 min; subsequently, the blood pressure was measured 3 times and the average value was recorded.
HUMAN BODY COMPOSITION ANALYSIS:
The 722 Bioelectrical Impedance Analyzer (Inbody, South Korea) was used to analyze body composition. We recorded BMI, body fat, body fat mass, waist-to-hip ratio (WHR), and visceral fat area.
BLOOD TESTS:
For blood tests, 4-mL venous blood samples were obtained from the participants after overnight fasting. The samples were centrifuged at 2500×
BLOOD LIPID PROFILE:
The levels of total cholesterol (TC) and triglycerides (TG) were detected by the oxidase method. The level of high-density lipoprotein cholesterol (HDL-C) was detected by the direct method, and the level of low-density lipoprotein cholesterol (LDL-C) was detected by the enzyme method. The tests were carried out by Beijing Deyi Clinical Laboratory Co. Ltd.
:
Serum levels of 25(OH)VitD3 were determined using liquid chromatography with tandem mass spectrometry.
INFLAMMATORY INDICES:
The levels of soluble intercellular adhesion molecule-1 (sICAM-1), soluble vascular cell adhesion molecule-1 (sVCAM-1), adiponectin, selectin, nuclear factor-κB (NF-κB), integrin, Toll-2, and Toll-4 were measured by enzyme-linked immunosorbent assay. These indices were measured by the Beijing Deyi Clinical Laboratory Co. Ltd.
:
The 24-h dietary review method was used to assess the pre-intervention diet of participants. The nutrition diagnosis and treatment system of the Yingkang Hospital was used to analyze the total daily energy intake and the intake and proportions of various food items (staple foods, leafy vegetables, root vegetables, salt, edible oil, eggs, milk, red meat, white meat, tofu, fruit) and nutrients (carbohydrate, protein, fat, dietary fiber, vitamin A, vitamin D, vitamin B1, vitamin B2, vitamin C, calcium, iron, zinc, selenium, magnesium, manganese, cholesterol, nicotinic acid, folic acid, saturated fatty acids, monounsaturated fatty acids, and polyunsaturated fatty acids).
QUALITY CONTROL:
The entire study was conducted in the same season, and the study population resided in the same village. These factors helped exclude the influence of season and skin color on the test results.
We conducted a health survey of middle-aged and elderly residents of this village and established the health records of the villagers in 2011, and we subsequently conducted our research investigations in 2013 and 2015. We have in-depth information on the dietary habits and life style of the inhabitants of this village. The village is located in a fairly remote area of Beijing, and the middle-aged and elderly residents lead a relatively simple life. The participants showed good compliance with the dietary intervention and the testing arrangements. The dietary habits in this village are representative of most economically underdeveloped rural areas in China. Our findings may help inform large-scale dietary interventions for lowering blood pressure in rural areas of China.
The dietary habits, daily activities, and sleep habits of participants during the adaptation period remain unchanged, which minimized the influence of changes in living habits on the test results.
Serum levels of transaminases and creatinine were measured before the intervention (September 3, 2017) and after the intervention (September 30, 2017) to evaluate the liver and renal function of participants. These assessments helped eliminate the influence of liver and kidney dysfunction on the test results. There were no significant differences in the pre- and postintervention levels of transaminases and creatinine.
Our dietary investigation revealed several problems in the dietary intake of villagers. We prepared a balanced diet plan to address this issue. The energy, carbohydrate, and fat content in the intervention diet in the 2 groups was comparable to that in the routine diet consumed by the participants prior to the study; however, we added greater food variety, including milk, eggs, fruits, vegetables, nuts, and meat. The only difference between the 2 balanced dietary patterns was the breakfast content. Group A received a steamed staple food made with 120 g of white flour, while Group B received 77 g of anti-atherosclerosis powder. Nutritional chefs ensured the quantitative aspects of meal preparation. Nutritional caterers prepared the anti-atherosclerosis powder according to the requirements, and it was weighed and packed in small bags. The nutritional caterers were responsible for putting the daily meal contents into each person’s portions. They strictly distinguished group A from group B, and indicated the names of the villagers.
Management nutritionists designed the weekly diet plan, and nutritional chefs ensured quantitative standardization of the prepared food according to the plan. Nutritional caterers prepared the anti-atherosclerosis powder and packaged each person’s daily diet according to the plan. The village committee of Zhang Fang was responsible for collecting the prepackaged food every day, providing a centralized dining place, and distributing the meals on time. Each group had a specially designated person to monitor and record the compliance of the villagers. All participants complied with the instructions and consumed the daily food according to the requirements of the informed consent; no extra helpings/meals were provided. All villagers received telephonic follow-up by nutritionists. Nutritionists were responsible for follow-up management (1 nutritionist for 2–3 participants) and supervision of participants to monitor the compliance with the study protocol.
Blood pressure was measured at the same time in the morning in the same environment. All participants were required to fast for >12 h. A specially designated person measured the blood pressure using the same sphygmomanometer.
For body composition analysis, all participants were required to fast for >12 h. All measurements were performed by specially designated personnel using the same human body composition analyzer; alcohol swabs were used to clean the soles of the feet and the electrodes before the measurement.
STATISTICAL METHODS:
All statistical analyses were performed using SPSS for Windows, version 17.0 (Chicago, IL, USA). Prior to statistical analysis, the normality of the distribution of all variables was assessed using the Kolmogorov-Smirnov test, histograms, and p-p plots. Descriptive statistics (mean, standard deviation, and range) for hepatorenal function were compared between the 2 time points and the intake of energy and nutrients was compared between the 2 groups and between the 2 time points using the
Results
TOLERANCE OF PARTICIPANTS DURING THE TEST:
During the intervention, all participants were contacted by telephone on a weekly basis to assess their tolerance of the dietary intervention. The dietary intervention included a low-fat and low-salt diet; however, all participants showed good tolerance to the diet during the study, and there were no extra meals or leftovers. The participants exhibited a good mental outlook and had normal bowel movements. At the beginning of the first week, some participants felt hungry but subsequently adapted to the new diet. One person required reduction in insulin dosage (by 2 IU), while 1 person stopped taking oral hypoglycemic drugs. None of the participants developed hypoglycemia or experienced fatigue, dizziness, nausea, or vomiting.
Before the intervention, no significant between-group differences were observed with respect to blood pressure, nutrient intake, body composition, blood lipid profile, 25(OH)VitD3 level, or the inflammatory indices.
There was no significant difference between pre- and postintervention alanine transaminase levels in either group (Table 1A, 1B). In both groups, postintervention creatinine levels were significantly higher than the respective pre-intervention levels due to increased intake of protein. However, the postintervention level of creatinine was within the normal range in all participants.
PRE- AND POSTINTERVENTION COMPARISON OF TOTAL ENERGY AND NUTRIENT INTAKE:
The pre- and postintervention total energy and nutrient intake in the 2 groups are shown in Table 2A–2D. No significant between-group differences were observed with respect to carbohydrate intake before the intervention. However, the postintervention carbohydrate intake in group A was significantly greater than that in group B (P=0.02). Postintervention protein intake in both groups was significantly greater than the respective pre-intervention levels (P<0.001); in addition, postintervention protein intake in group B was significantly greater than that in group A (P<0.001). There was no significant difference in fat intake between group A and group B before and after intervention. Postintervention dietary intake of fiber, vitamin B1, vitamin B2, nicotinic acid, folic acid, vitamin C, vitamin A, calcium, iron, zinc, magnesium, manganese, cholesterol, saturated fatty acid, and polyunsaturated fatty acids was significantly greater than the respective pre-intervention levels in both groups. The postintervention dietary intake of magnesium and manganese in group B was significantly greater than that in group A; however, there were no significant differences between group A and B with respect to postintervention intake of the other nutrients. There was no significant difference between the pre- and postintervention intake of selenium in group A; however, selenium intake in group B increased significantly after the intervention (P=0.027). Before the intervention, there was no significant difference between the 2 groups with respect to selenium intake; however, postintervention selenium intake in group A was significantly higher than that in group B (P=0.026). Vitamin D intake showed a significant increase after intervention in both groups (P<0.05); however, there was no significant difference between the 2 groups before and after intervention. After the intervention, there were significant differences between the 2 groups only with respect to the intake of carbohydrate, protein, and selenium. There was no significant between-group difference with respect to the intake of other nutrients. However, there were significant differences between the pre-intervention and postintervention intake of protein, fat, dietary fiber, vitamin B2, nicotinic acid, Folic acid, vitamin C, vitamin A, vitamin E, vitamin D, calcium, iron, zinc, magnesium, manganese, cholesterol, saturated fatty acids, and polyunsaturated fatty acids. Above all, there were no significant differences between the 2 groups with respect to nutrients; therefore, there were no significant differences between the 2 groups with respect to SBP and DBP.
:
The correlation analysis of the 25(OH)VitD3 level with SBP and DBP before and after intervention is shown in Table 3. Both SBP and DBP showed a significant negative correlation with 25(OH)VitD3 level (P<0.05). The higher the 25(OH)VitD3 level, the lower the SBP and DBP were.
:
The correlation analysis and regression analysis of 25(OH)VitD3 level and various nutrients before and after the intervention are shown in Table 4. The level of 25(OH)VitD3 showed a simple correlation with all nutrients except carbohydrates. In the regression analysis, the independent variables protein, fat, calcium, cholesterol, and vitamin D showed a significant positive effect on the dependent variable, 25(OH)VitD3 level (P<0.05). Increased intake of protein, fat, calcium, cholesterol, and vitamin D showed a significant positive correlation with increased 25(OH)VitD3 level.
:
The correlation analysis of 25(OH)VitD3 level with body composition, blood lipid level, and inflammatory indices before and after the intervention is shown in Table 5. The level of 25(OH)VitD3 had a simple linear correlation with body fat, WHR, visceral fat, TC, TG, LDL-C, and Toll-2. However, the level of 25(OH)VitD3 showed no significant correlation with the body fat content, HDL-C, adiponectin, selectin, sICAM-1, sVCAM-1, NF-κB, integrin, or Toll-4.
The regression analysis of 25(OH)VitD3 level with body composition, blood lipid level, and inflammatory indices before and after the intervention are shown in Table 6. On further regression analysis, the independent variables WHR, TG, and Toll-2 showed a significant negative association with the dependent variable 25(OH)VitD3 level (P<0.05). The level of 25(OH)VitD3 decreased with increases in WHR, TG, and Toll-2.
Discussion
:
The vitamin D content in food is typically low. Approximately 90% of vitamin D required by the body is synthesized from cholesterol through exposure to sunlight [23]. In studies conducted in Mexico [7] and Korea [19], the prevalence of VDD in rural areas was lower than that in urban areas, which was likely attributable to greater exposure to sunlight in rural areas. The dietary intake of vitamin D has been suggested to have a limited relationship with the serum 25(OH)VitD3 level [23]. Several studies have demonstrated a positive correlation between serum levels of 25(OH)VitD3 and HDL-C [24,25]. Our study population was exclusively composed of individuals residing in a rural setting; however, they had a very high prevalence of VDD. This high prevalence may be attributable to factors other than the lack of sunlight exposure.
The main dietary sources of vitamin D are milk, egg, fish, and seafood. In a Mexican study of 4162 women of childbearing age, the average intake of vitamin D was 2.56 μg/d, which was well below the 10 μg/d recommended by the Institute of Medicine [7]. The corresponding average intake in Japan was 10.7 μg/d [26], and that in the United States was 8.6 μg/d [27].
The routine diet of villagers in our study setting is centered around staple foods and pickled vegetables, all of which are deficient in vitamin D. The pre-intervention average daily intake of vitamin D was 8.72 μg/d, which increased to 12.21 μg/d during the intervention. This may have contributed to the increased serum 25(OH)VitD3 level after the intervention. At the same time, the intake of cholesterol showed a significant increase after the intervention. Cholesterol is the precursor for the synthesis of 7-dehydrocholesterol in the skin driven by ultraviolet irradiation. In the present study, the level of 25(OH)VitD3 had a significantly positive correlation with the intake of cholesterol and vitamin D. Moreover, dairy products, eggs, and other foods are rich in vitamin D and cholesterol; in addition, these foods are also rich in protein, fat, and calcium. Therefore, the above 3 nutrients also showed a positive correlation with 25(OH)VitD3.
Thus, adequate dietary intake of vitamin D and cholesterol may help increase the serum level of 25(OH)VitD3.
:
The body fat, visceral fat, and WHR of our participants showed a negative correlation with the serum 25(OH)VitD3 level. The possible reason is that vitamin D is fat soluble. Body fat can store vitamin D, which reduces its bioavailability [7,28] and further reduces the serum level of 25(OH)VitD3 in individuals with overweight or obesity. In our study, reduction in the body fat, visceral fat, and WHR increased the bioavailability of vitamin D, leading to a significant increase in the serum level of 25(OH)VitD3.
At the same time, some studies have shown that 25(OH)VitD3 can help improve the blood lipid profile (reduce TG, increase HDL-C) and ameliorate the degree of vascular atherosclerosis [4,9,19]. These findings are consistent with our study, which showed that the levels of TC and TG had a significant negative correlation with the level of 25(OH)VitD3. Homeostasis of human TC is closely related to the regulation of liver and is mainly regulated by the negative feedback of sterol regulatory element binding protein 2 (SREBP-2). 3-Hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR) is mainly regulated by SREBP-2 and is involved in cholesterol synthesis and uptake [29]. In a study excluding the effect of diet on cholesterol metabolism via depleting dietary cholesterol, Li et al [30] found that the fecal excretion of cholesterol in patients with vitamin D deficiency did not decrease. This outcome indicated that the increased TC in the blood mainly resulted from increased endogenous cholesterol synthesis. At the same time, the protein expression of HMGCR and its nuclear transcription factor SREBP-2 was significantly increased in patients with vitamin D deficiency. Insulin inducible gene 2 (Insig-2) can regulate the activation of SREBP, and its expression is mediated by the combination of 25(OH)VitD3 and vitamin D receptor (VDR). In the absence of vitamin D, the transcriptional activity of VDR decreased, resulting in the downregulation of the expression of Insig-2 and inhibition of the activation of SREBP-2. Vitamin D prevents the increase in cholesterol caused by VDD by maintaining the transcriptional activity of VDR and Insig-2 expression [31].
People with overweight or obesity have greater amounts of adipose tissue. Adipocytes perform an endocrine function and can secrete adipokines with proinflammatory effects, such as tumor necrosis factor α, interleukin-6, C-reactive protein, and phospholipase A2 related to lipoprotein [32]. The presence of inflammation has an adverse effect on the serum level of 25(OH)VitD3 [33,34]. Notably, Toll-2 inflammatory factor showed a significant negative correlation with 25(OH)VitD3 in our study.
:
An extensive body of literature supports the close relationship between 25(OH)VitD3 and hypertension. Several previous studies have implicated 25(OH)VitD3 deficiency in the causation of cardiovascular diseases (especially hypertension) [10]. A decrease in the serum 25(OH)VitD3 level was shown to be an independent risk factor for cardiovascular disease, and it also showed a negative relation with blood pressure [12]. In the present study, the serum level of 25(OH)VitD3 was negatively correlated with SBP and DBP. After the dietary intervention, both SBP and DBP decreased; in addition, the decrease in blood pressure had a significant correlation with the change in the 25(OH)VitD3 level. These findings are consistent with the results of previous studies.
The following mechanisms may explain the effect of 25(OH)VitD3 on blood pressure. First, an improvement in the blood lipid profile (eg, decrease in TG, increase in HDL-C) reduces blood viscosity and the degree of vascular atherosclerosis and thus lowers blood pressure [4,9,19]. This mechanism is consistent with the lowering of blood pressure and blood lipid levels observed in the present study. Second, reduced body weight and body fat helps to lower blood pressure [13,14], which is consistent with the lowering of blood pressure and decrease in body fat and WHR observed in our study. Third, an anti-inflammatory effect may reduce blood pressure [8, 35], and in the present study, we observed a decrease in the level of Toll-2 alongside a decrease in the blood pressure.
:
Vitamin D supplementation has been shown to help reduce body fat [7], and daily supplementation with 200 IU was found to reduce body weight and body fat [36]. Moreover, an increased serum 25(OH)VitD3 level can also inhibit the formation of adipose tissue [37]. Therefore, reducing body fat is a reliable method to increase the serum 25(OH)VitD3 level [38]. Patients with nonalcoholic fatty liver disease are prescribed a low-carbohydrate diet for a short period of time. An increase in dietary protein intake can ameliorate the inflammatory response, reduce body fat, and improve intestinal flora [39], as well as increase serum 25(OH)VitD3 level, thus reducing the blood pressure. The currently popular balanced diets across the world (including the DASH diet and the Mediterranean diet) are rich in fruits and vegetables that have a high content of phytochemicals, vitamin C, and other micronutrients. The consequent increase in protein level and decreased carbohydrate level have anti-inflammatory effects [40–43]. These diets can help ameliorate lipid metabolism disorders and increase serum 25(OH)VitD3 level, thus decreasing the blood pressure level.
The diet plan used in our study has many similarities with the DASH diet and the Mediterranean diet. The dietary intervention in our study resulted in increased intake of vitamin D, cholesterol, protein, fat, and calcium. After the intervention, the participants showed decreases in body fat, visceral fat, WHR, and TC and TG levels along with improvement in inflammatory indices. Previous studies [6,17] showed that blood pressure can be reduced by improving body composition, reducing blood lipid levels, and reducing the level of inflammation. In the present study, we found that increasing the level of 25(OH)VitD3 can improve the above-mentioned indices. These findings suggest that improving the dietary structure can help improve the body composition and reduce the levels of blood lipids and inflammatory indices by increasing the serum 25(OH)VitD3 level, so as to achieve the effect of reducing blood pressure.
STUDY STRENGTHS:
This investigation was a meticulously implemented dietary intervention study of hypertensive participants residing in a rural area. We used a standardized diet prepared according to the standards and provided free of cost, and strict quality control measures were in place throughout the study. The study entailed measurement of multiple indices, including blood pressure, blood sugar, body composition, and inflammatory indices. Our study demonstrates the safety and efficacy of our dietary intervention in hypertensive patients.
STUDY LIMITATIONS:
Some limitations of our study should be considered while interpreting the results. First, the sample size was relatively small, and the duration of the intervention was relatively short. In addition, we only measured the blood pressure for assessment of cardiovascular function. Other indicators, such as vasomotor function dependent on nitric oxide concentration or blood flow, were not measured.
Conclusions
A balanced diet pattern may help increase the serum 25(OH)VitD3 level and reduce blood pressure in hypertensive patients. The potential underlying mechanisms are: (1) increased dietary intake of vitamin D, (2) appropriate increase in dietary cholesterol intake, (3) reduction in body fat, (4) decrease in inflammation level, and (5) improvement in blood lipid profile. Increasing the dietary intake of vitamin D, promoting appropriate intake of cholesterol, and ensuring adequate intake of protein and fat can help improve body composition, ameliorate lipid metabolism disorder, have an anti-inflammatory effect, and increase the serum 25(OH)VitD3 level. All these factors may contribute to a decrease in blood pressure.
Figures
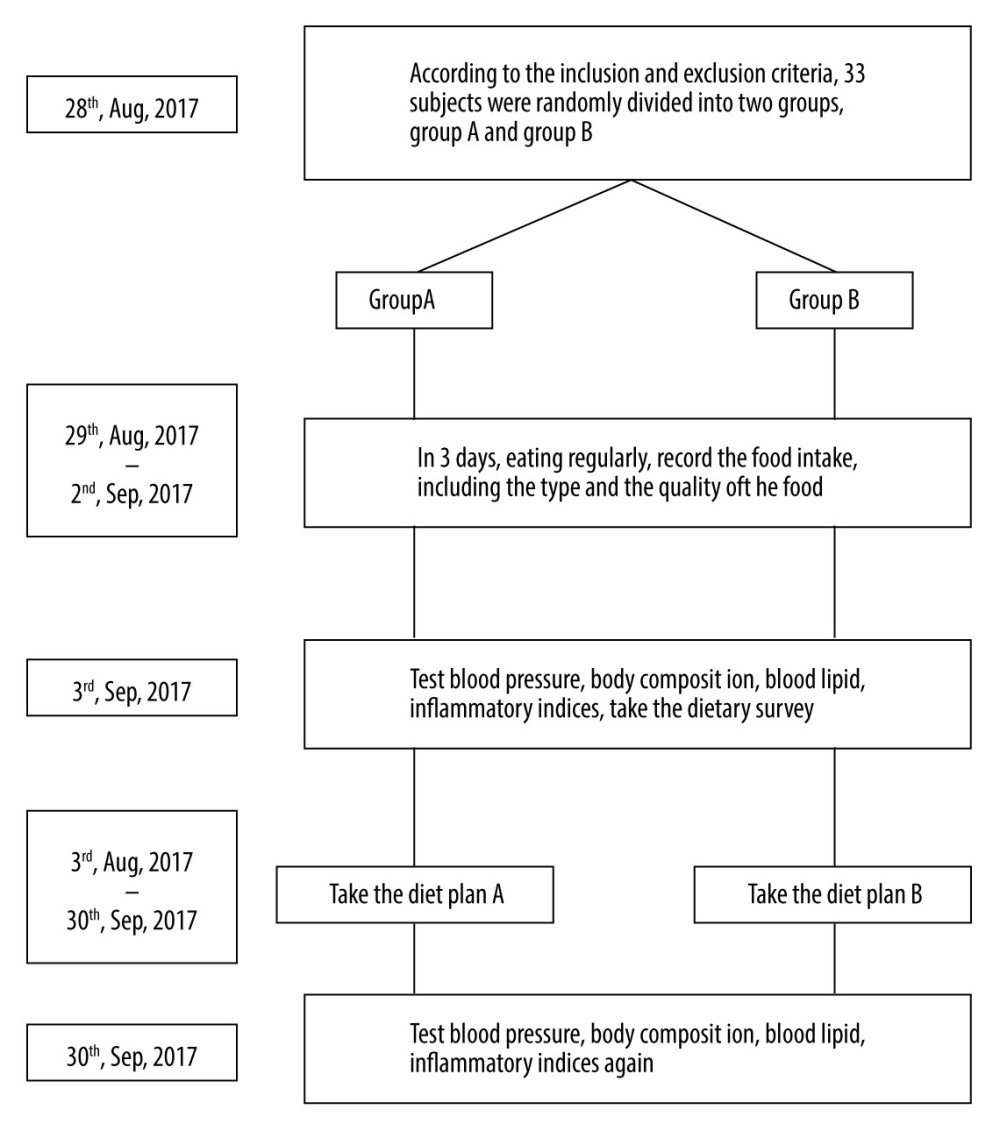 Figure 1. The research process.
Figure 1. The research process. Tables
Table 1A. Comparison of the baseline data between the 2 groups.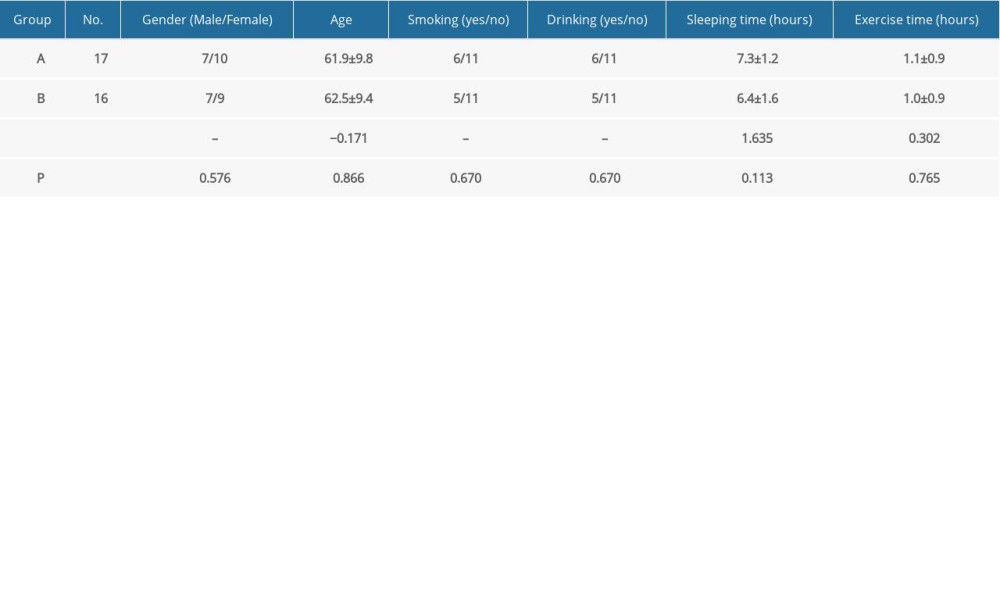 Table 1B. Comparison of hepatorenal function before and after intervention.
Table 1B. Comparison of hepatorenal function before and after intervention.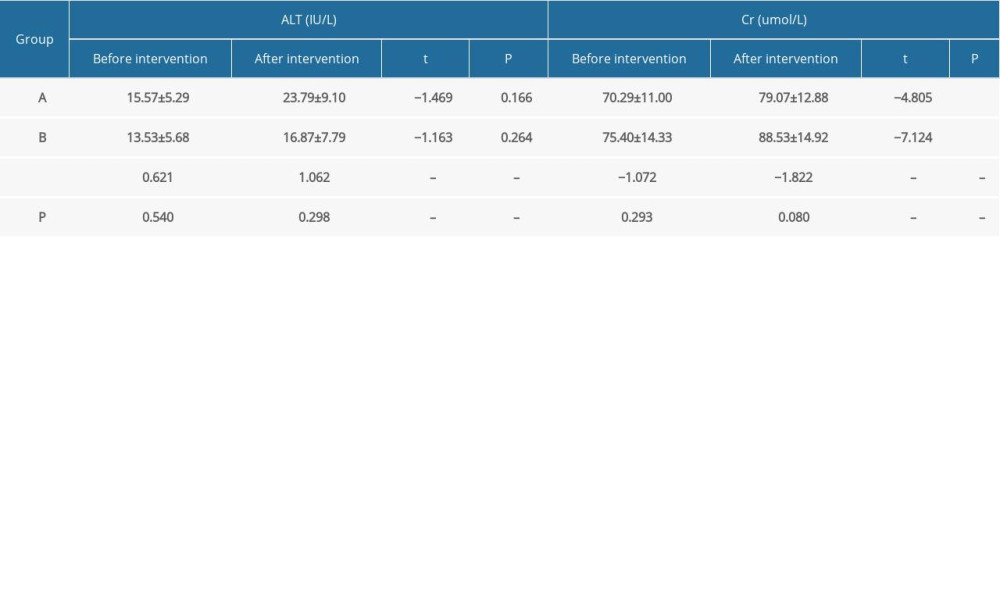 Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points.
Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points.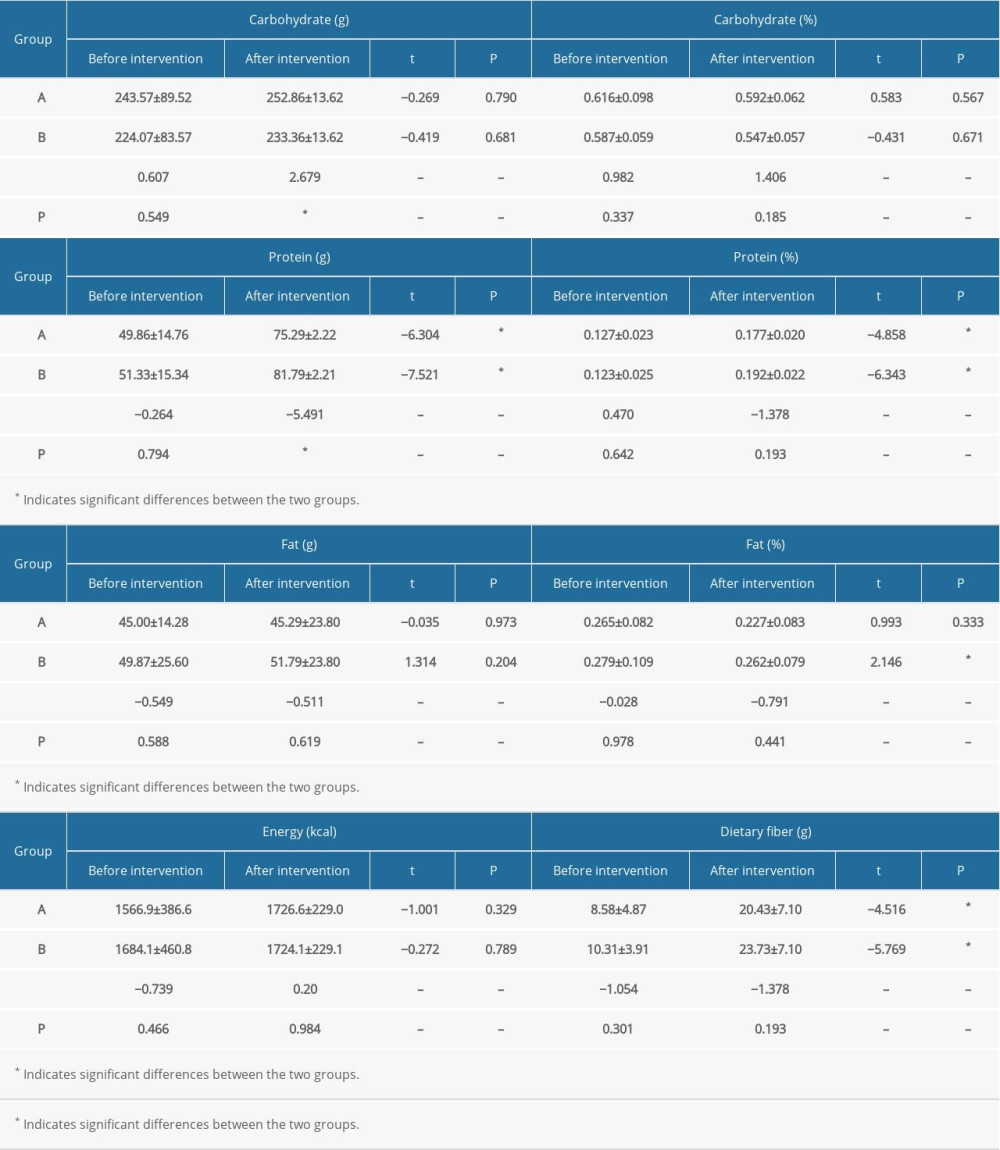 Table 2B. Comparison of vitamin intake between the 2 groups at different time points.
Table 2B. Comparison of vitamin intake between the 2 groups at different time points.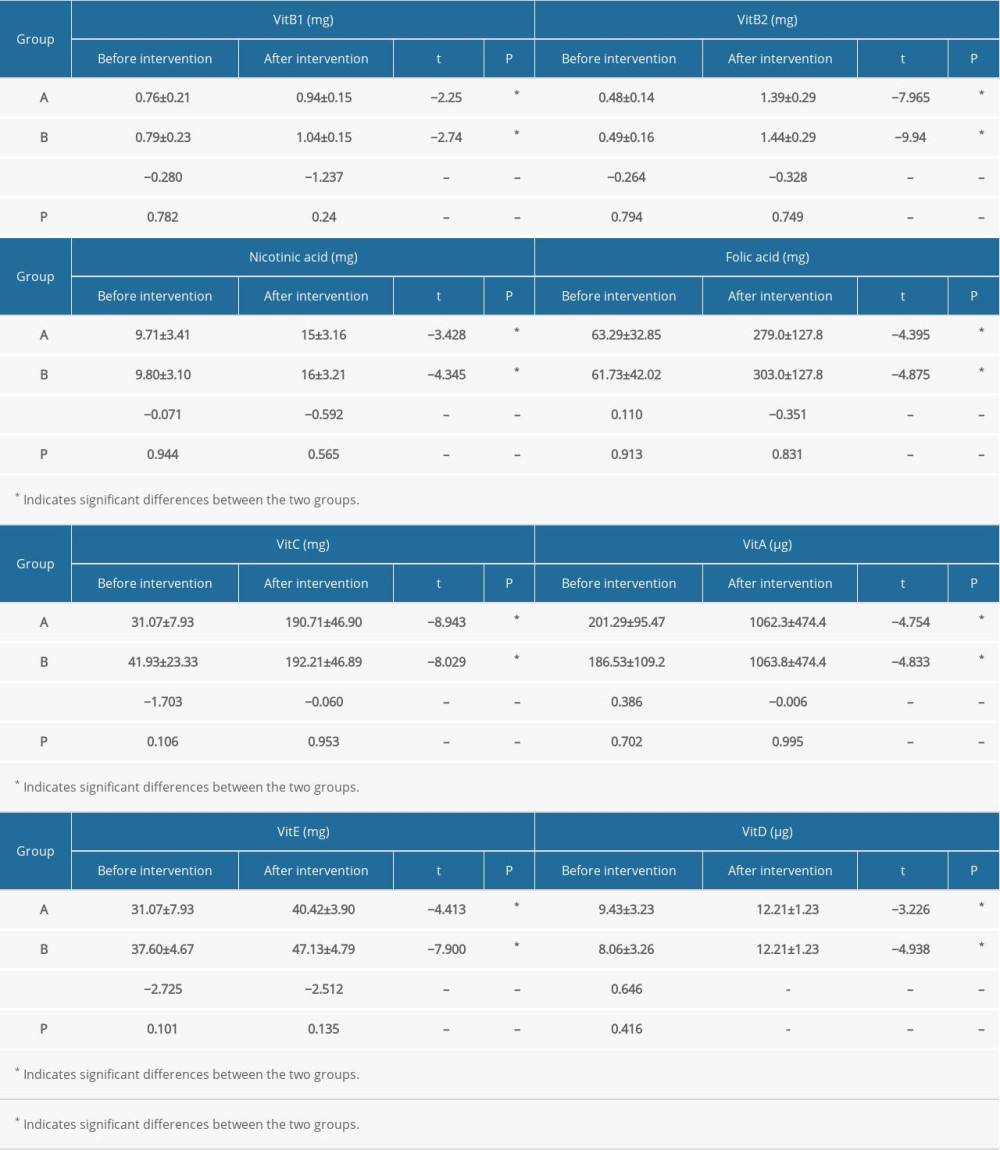 Table 2C. Comparison of mineral intake between the 2 groups at different time points.
Table 2C. Comparison of mineral intake between the 2 groups at different time points.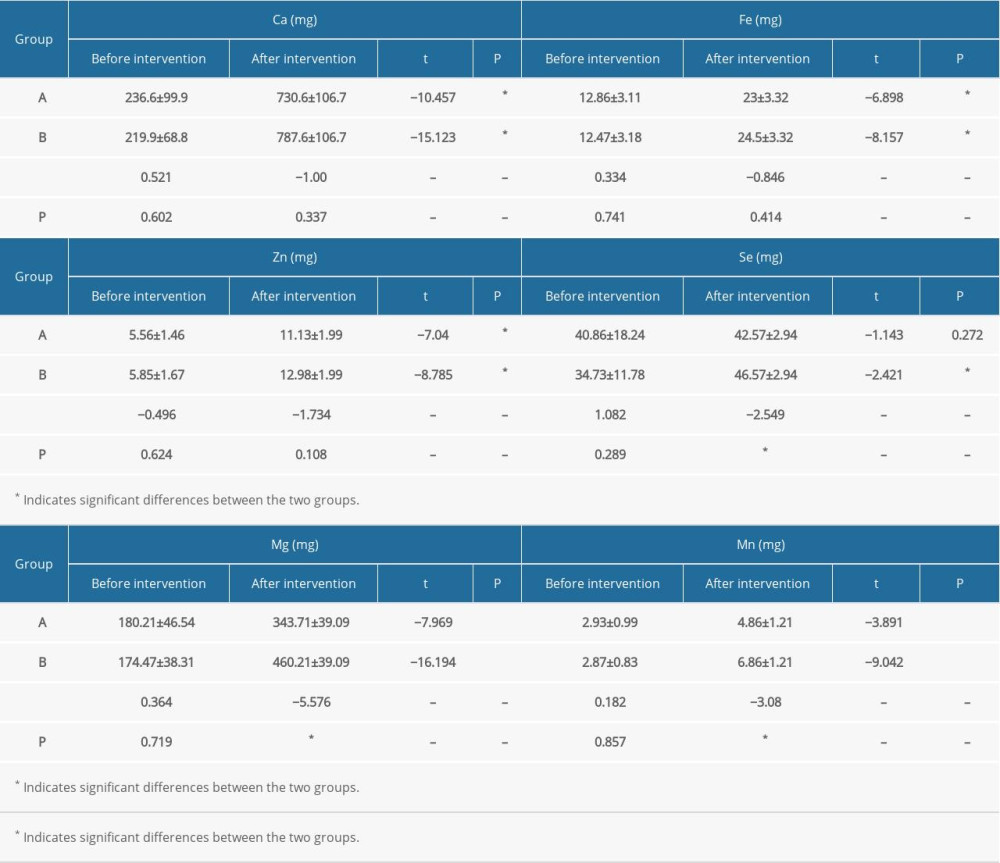 Table 2D. Comparison of lipid intake between the 2 groups at different time points.
Table 2D. Comparison of lipid intake between the 2 groups at different time points.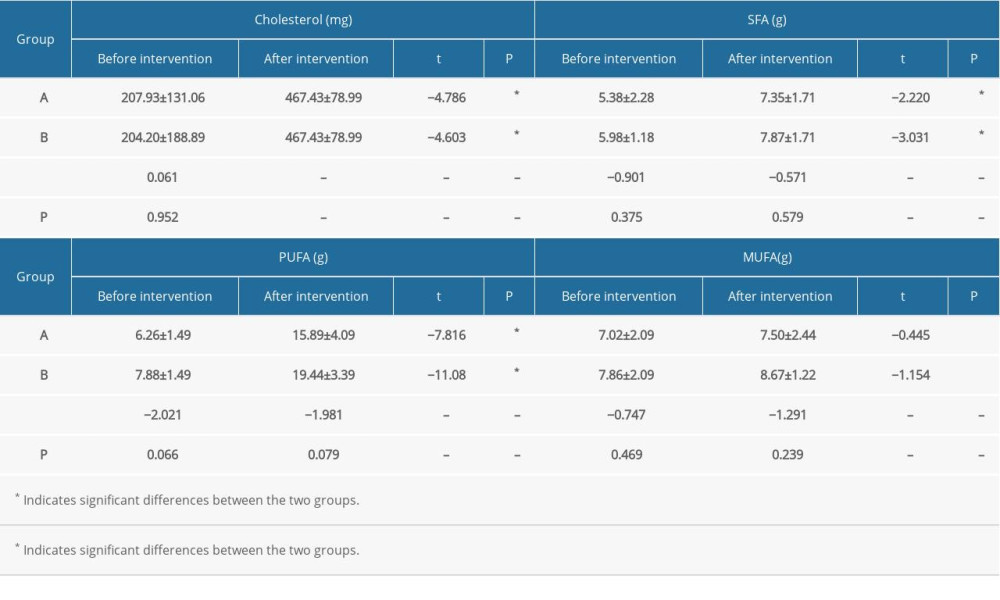 Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention.
Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention.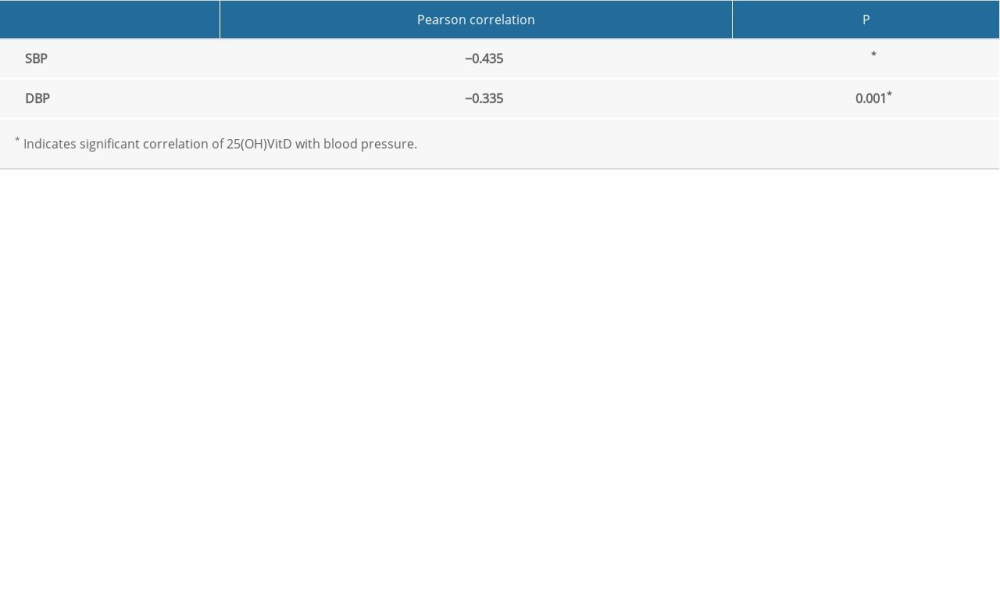 Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients.
Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients.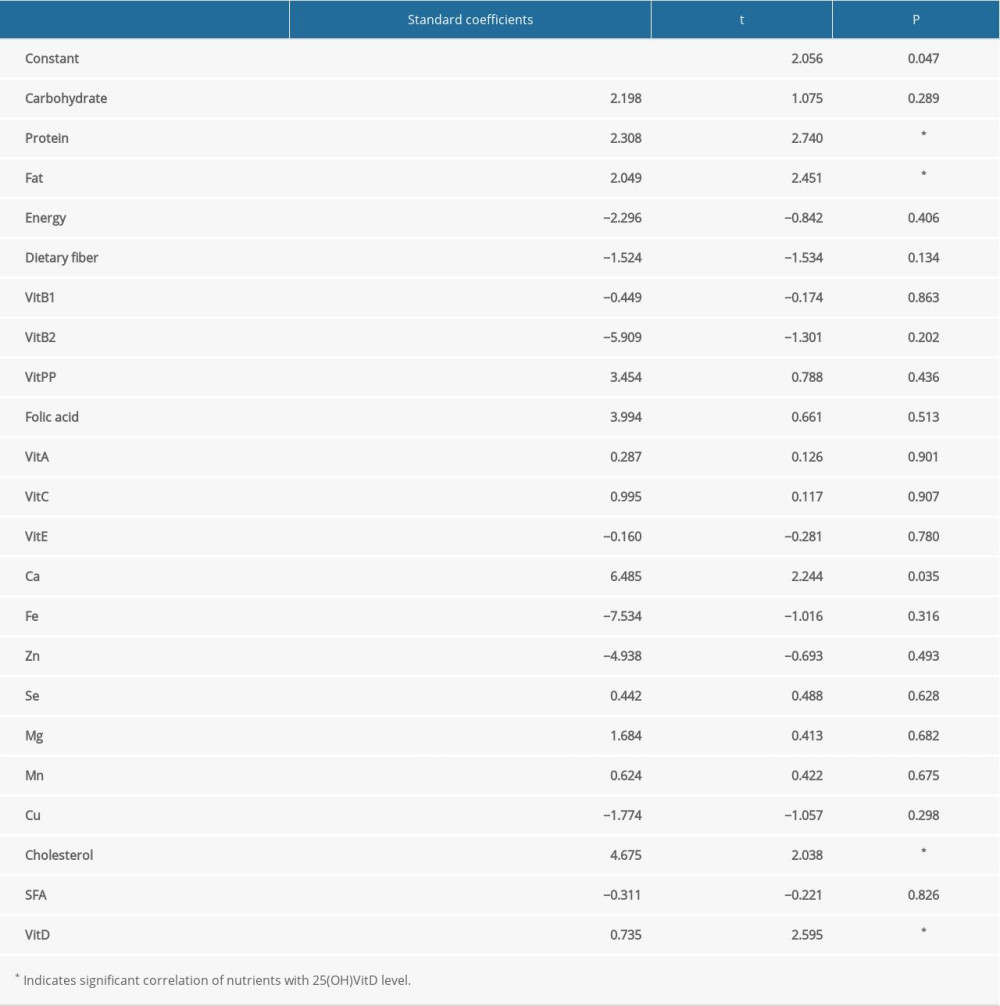 Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices.
Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices.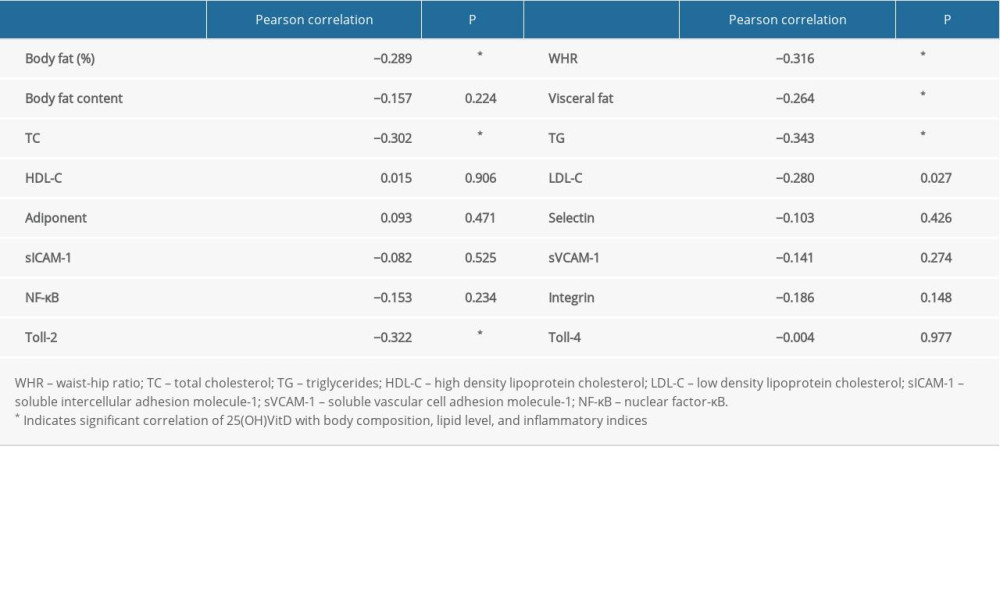 Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices.
Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices.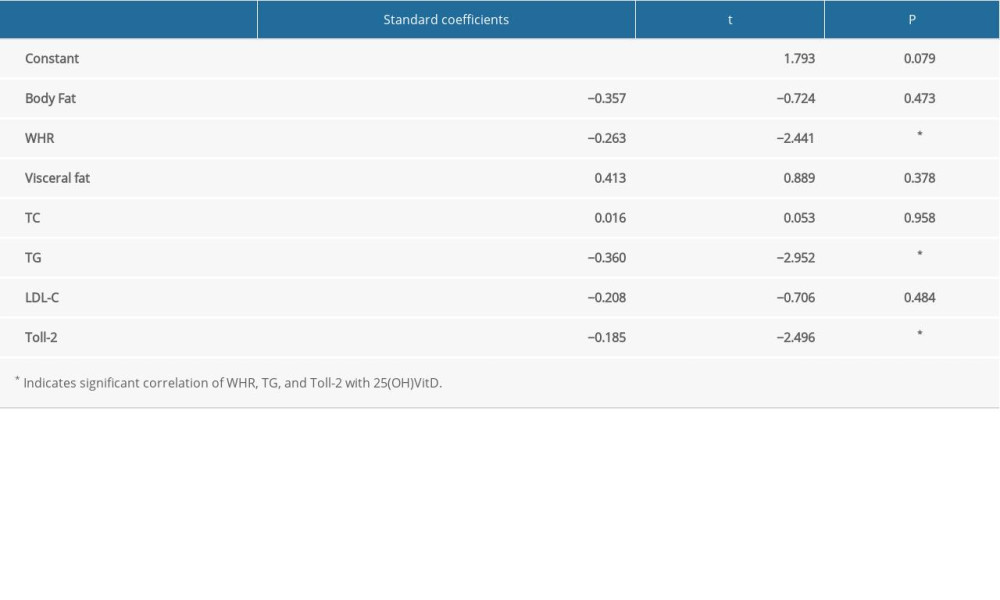
References
1. Prabhakar P, Chandra SR, Supriya M, Vitamin D status and vascular dementia due to cerebral small vessel disease in the elderly Asian Indian population: J Neurol Sci, 2015; 359; 108-11
2. Botella-Carretero JI, Alvarez-Blasco F, Villafruela JJ, Vitamin D deficiency is associated with the metabolic syndrome in morbid obesity: Clin Nutr, 2007; 26; 573-80
3. Stocklin E, Eggersdorfer M, Vitamin D, an essential nutrient with versatile functions in nearly all organs: Int J Vitam Nutr Res, 2013; 83; 92-100
4. Lopes MP, Giudici KV, Marchioni DM, Relationships between n-3 polyunsaturated fatty acid intake, serum 25 hydroxyvitamin D, food consumption, and nutritional status among adolescents: Nutr Res, 2015; 35; 681-88
5. Beveridge LA, Witham MD, Vitamin D and the cardiovascular system: Osteoporos Int, 2013; 24; 2167-80
6. Wang J, Hong Z, Wang N, Effects of a dietary supplement on inflammatory marker expression in middle-aged and elderly hypertensive patients: Clinics (Sao Paulo, Brazil), 2019; 74; e890
7. Contreras-Manzano A, Villalpando S, Robledo-Perez R, Vitamin D status by sociodemographic factors and body mass index in Mexican women at reproductive age: Salud Publica Mex, 2017; 59; 518-25
8. Ibero-Baraibar I, Navas-Carretero S, Abete I, Increases in plasma 25(OH)D levels are related to improvements in body composition and blood pressure in middle-aged subjects after a weight loss intervention: Longitudinal study: Clin Nutr, 2015; 34; 1010-17
9. Schmitt EB, Nahas-Neto J, Bueloni-Dias F, Vitamin D deficiency is associated with metabolic syndrome in postmenopausal women: Maturitas, 2018; 107; 97-102
10. Ku YC, Liu ME, Ku CS, Relationship between vitamin D deficiency and cardiovascular disease: World J Cardiol, 2013; 5; 337-46
11. Gu JW, Liu JH, Xiao HN, Relationship between plasma levels of 25-hydroxyvitamin D and arterial stiffness in elderly Chinese with non-dipper hypertension: An observational study: Medicine (Baltimore), 2020; 99; e19200
12. Zhang QW, Su L, Li WSerum 1,25-dihydroxy vitamin D3 level in elderly primary hypertension patients: Chinese Journal of Geriatric Heart Brain and Vessel Diseases, 2014; 16; 256-58 [in Chinese]
13. Ortega RM, Aparicio A, Rodriguez-Rodriguez E, Preliminary data about the influence of vitamin D status on the loss of body fat in young overweight/obese women following two types of hypocaloric diet: Br J Nutr, 2008; 100; 269-72
14. Mason C, Xiao L, Imayama I, Effects of weight loss on serum vitamin D in postmenopausal women: Am J Clin Nutr, 2011; 94; 95-103
15. Zhou BPredictive values of body mass index and waist circumference to risk factors of related diseases in Chinese adult population: Zhonghua Liu Xing Bing Xue Za Zhi, 2002; 23; 5-10 [in Chinese]
16. Wang J, Hong ZX, Wu L[Dietary pattern and pathogenesis of hypertension of middle aged and elderly people.]: Chinese General Practice, 2016; 19; 1706-10 [in Chinese]
17. Wang J, Hong ZX, Wu L[Effects of dietary pattern improvement on blood pressure, body composition and blood lipid profile of overweight or obese middle-aged and aged people with hypertension in Zhangfang Village of Fangshan District of Beijing.]: Chinese General Practice, 2017; 20; 290-98 [in Chinese]
18. Holick MF, High prevalence of vitamin D inadequacy and implications for health: Mayo Clin Proc, 2006; 81; 353-73
19. Kang JY, Kim MK, Jung S, The cross-sectional relationships of dietary and serum vitamin D with cardiometabolic risk factors: Metabolic components, subclinical atherosclerosis, and arterial stiffness: Nutrition, 2016; 32; 1048-56.e1
20. Ganji V, Zhang X, Tangpricha V, Serum 25-hydroxyvitamin D concentrations and prevalence estimates of hypovitaminosis D in the U.S. population based on assay-adjusted data: J Nutr, 2012; 142; 498-507
21. Greene-Finestone LS, Berger C, de Groh M, 25-Hydroxyvitamin D in Canadian adults: Biological, environmental, and behavioral correlates: Osteoporos Int, 2011; 22; 1389-99
22. Jung IK, Prevalence of vitamin D deficiency in Korea: Results from KNHANES 2010 to 2011: J Nutr Health, 2013; 46; 540-51
23. Holick MF, Chen TC, Vitamin D deficiency: A worldwide problem with health consequences: Am J Clin Nutr, 2008; 87; 1080s-86s
24. Ponda MP, Huang X, Odeh MA, Vitamin D may not improve lipid levels: A serial clinical laboratory data study: Circulation, 2012; 126; 270-77
25. Carbone LD, Rosenberg EW, Tolley EA, 25-Hydroxyvitamin D, cholesterol, and ultraviolet irradiation: Metabolism, 2008; 57; 741-48
26. Nakamura K, Kitamura K, Takachi R, Impact of demographic, environmental, and lifestyle factors on vitamin D sufficiency in 9084 Japanese adults: Bone, 2015; 74; 10-17
27. Moore CE, Radcliffe JD, Liu Y, Vitamin D intakes of adults differ by income, gender and race/ethnicity in the U.S.A., 2007 to 2010: Public Health Nutr, 2014; 17; 756-63
28. Foss YJ, Vitamin D deficiency is the cause of common obesity: Med Hypotheses, 2009; 72; 314-21
29. Han JS, Sung JH, Lee SK, Inhibition of cholesterol synthesis in HepG2 cells by GINST-decreasing HMG-CoA reductase expression via AMP-activated protein kinase: J Food Sci, 2017; 82; 2700-5
30. Li S, He Y, Lin S, Increase of circulating cholesterol in vitamin D deficiency is linked to reduced vitamin D receptor activity via the Insig-2/SREBP-2 pathway: Mol Nutr Food Res, 2016; 60; 798-809
31. Rodrigue-Way A, Caron V, Bilodeau S, Scavenger receptor CD36 mediates inhibition of cholesterol synthesis via activation of the PPARγ/PGC-1α pathway and Insig1/2 expression in hepatocytes: FASEB J, 2014; 28; 1910-23
32. Balistreri CR, Caruso C, Candore G, The role of adipose tissue and adipokines in obesity-related inflammatory diseases: Mediators Inflamm, 2010; 2010 802078
33. Kim M, Na W, Sohn C, Correlation between vitamin D and cardiovascular disease predictors in overweight and obese Koreans: J Clin Biochem Nutr, 2013; 52; 167-71
34. Laird E, McNulty H, Ward M, Vitamin D deficiency is associated with inflammation in older Irish adults: J Clin Endocrinol Metab, 2014; 99; 1807-15
35. Todd AS, Street SJ, Ziviani J, Overweight and obese adolescent girls: The importance of promoting sensible eating and activity behaviors from the start of the adolescent period: Int J Environ Res Public Health, 2015; 12; 2306-29
36. Mason C, Xiao L, Imayama I, Vitamin D3 supplementation during weight loss: A double-blind randomized controlled trial: Am J Clin Nutr, 2014; 99; 1015-25
37. Ching S, Kashinkunti S, Niehaus MD, Mammary adipocytes bioactivate 25-hydroxyvitamin D(3) and signal via vitamin D(3) receptor, modulating mammary epithelial cell growth: J Cell Biochem, 2011; 112; 3393-405
38. Ding C, Gao D, Wilding J, Vitamin D signalling in adipose tissue: Br J Nutr, 2012; 108; 1915-23
39. Mardinoglu A, Wu H, Bjornson E, An integrated understanding of the rapid metabolic benefits of a carbohydrate-restricted diet on hepatic steatosis in humans: Cell Metab, 2018; 27; 559-71.e5
40. Kelly OJ, Gilman JC, Kim Y, Micronutrient intake in the etiology, prevention and treatment of osteosarcopenic obesity: Curr Aging Sci, 2016; 9; 260-78
41. Calder PC, Ahluwalia N, Brouns F, Dietary factors and low-grade inflammation in relation to overweight and obesity: Br J Nutr, 2011; 106(Suppl.3); S5-78
42. Johnston CS, Strategies for healthy weight loss: From vitamin C to the glycemic response: J Am Coll Nutr, 2005; 24; 158-65
43. Medina-Remon A, Casas R, Tressserra-Rimbau A, Polyphenol intake from a Mediterranean diet decreases inflammatory biomarkers related to atherosclerosis: A substudy of the PREDIMED trial: Br J Clin Pharmacol, 2017; 83; 114-28
Figures
Tables
 Table 1A. Comparison of the baseline data between the 2 groups.
Table 1A. Comparison of the baseline data between the 2 groups. Table 1B. Comparison of hepatorenal function before and after intervention.
Table 1B. Comparison of hepatorenal function before and after intervention. Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points.
Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points. Table 2B. Comparison of vitamin intake between the 2 groups at different time points.
Table 2B. Comparison of vitamin intake between the 2 groups at different time points. Table 2C. Comparison of mineral intake between the 2 groups at different time points.
Table 2C. Comparison of mineral intake between the 2 groups at different time points. Table 2D. Comparison of lipid intake between the 2 groups at different time points.
Table 2D. Comparison of lipid intake between the 2 groups at different time points. Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention.
Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention. Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients.
Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients. Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices.
Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices. Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices.
Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices. Table 1A. Comparison of the baseline data between the 2 groups.
Table 1A. Comparison of the baseline data between the 2 groups. Table 1B. Comparison of hepatorenal function before and after intervention.
Table 1B. Comparison of hepatorenal function before and after intervention. Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points.
Table 2A. Comparison of intake of energy and generated energy nutrients between the 2 groups at different time points. Table 2B. Comparison of vitamin intake between the 2 groups at different time points.
Table 2B. Comparison of vitamin intake between the 2 groups at different time points. Table 2C. Comparison of mineral intake between the 2 groups at different time points.
Table 2C. Comparison of mineral intake between the 2 groups at different time points. Table 2D. Comparison of lipid intake between the 2 groups at different time points.
Table 2D. Comparison of lipid intake between the 2 groups at different time points. Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention.
Table 3. Correlation analysis of the 25-hydroxyvitamin D3 level with systolic and diastolic blood pressure before and after dietary intervention. Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients.
Table 4. Regression analysis of 25-hydroxyvitamin D3 level and various nutrients. Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices.
Table 5. Correlation analysis of 25-hydroxyvitamin D3 level with body composition, blood lipid level, and inflammatory indices. Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices.
Table 6. Regression analysis of 25-hydroxyvitamin D3 level and body composition, blood lipid level, and inflammatory indices. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








