10 February 2021: Lab/In Vitro Research
Knockdown of Dual Oxidase 1 (DUOX1) Promotes Wound Healing by Regulating Reactive Oxygen Species (ROS) by Activation of Nuclear Factor kappa B (NF-κB) Signaling
Hui Wang1BC, Haowei Lu2DF, Yige Wu1AE*DOI: 10.12659/MSM.926492
Med Sci Monit 2021; 27:e926492
Abstract
BACKGROUND: The aim of this study was to evaluate the potential role of dual oxidase 1 (DUOX1) in wound healing.
MATERIAL AND METHODS: Primary fibroblasts were isolated from wound granulation tissue. Fibroblasts cell lines were established using DUOX1 overexpression and interference. Cell proliferation and reactive oxygen species (ROS) production were measured and compared among the groups.
RESULTS: DUOX1 expression was highest in the slow-healing tissues (P<0.05). Knockdown of DUOX1 significantly increased cell proliferation and inhibited ROS production and cell apoptosis (P<0.01). Moreover, expression of malondialdehyde (MDA) was significantly reduced, while expression of superoxide dismutase (SOD) expression was significantly increased (P<0.01). In addition, DUOX1 silencing significantly upregulated collagen I, collagen III, and NF-κB protein levels in the cytoplasm, and inhibited the protein levels of P21, P16, and NF-κB in the nucleus (P<0.01). Overexpression of DUOX1 caused a reverse reaction mediated by knockdown of DUOX1. When DUOX1-overexpressing cells were treated with the ROS inhibitor N-acetyl-L-cysteine (NAC), the protein levels that were increased by DUOX1 overexpression were reversed.
CONCLUSIONS: These results suggest that knockdown of DUOX1 significantly benefits wound healing, likely by the regulation of oxidative stress via NF-κB pathway activation.
Keywords: NADPH Oxidase, Wound Healing, Acetylcysteine, Dual Oxidases, Fibroblasts, Malondialdehyde, primary cell culture
Background
In humans, the skin is the organ with the largest surface area, and it plays key roles in protecting internal tissues from various types of damage, including mechanical, bacterial, and ultraviolet rays. The skin is also susceptible to extensive burns and skin wounds, which impose a heavy financial burden on healthcare systems [1]. Abnormal wound healing is problematic, and recent interventions for wound healing are only moderately effective; thus, more effective therapies for wound repair are required.
Oxidative stress represents a state of imbalance between oxidants and antioxidants within the body, which leads to increased production of free radicals and decreased antioxidant capacity. Moreover, excessive oxidative stress promotes the development of many diseases, as well as tissue damage [2]. Oxygen is essential for tissue renewal, and reactive oxygen species (ROS) are described as radical derivatives of oxygen, which play roles as secondary messenger-signaling molecules. Many cells in the wound microenvironment can produce ROS to varying degrees [3], and high levels of ROS lead to lipid peroxidation. As a consequence, the level of malondialdehyde (MDA), as an end-product of ROS-induced peroxidation, also increases. Superoxide dismutase (SOD) is a type of antioxidant enzyme that can remove superoxide free radicals [4,5].
Wound healing is a complex and orderly biological process, which involves multiple sequential and intersecting pathophysiological processes, such as inflammatory response, cell proliferation, migration, and wound remodeling [6,7]. Dual oxidase (DUOX) belongs to the NADPH oxidase family, and has potential roles in cell signaling, hypoxic response, and immune function [8]. DUOX1 and DUOX2 are 2 isoforms of DUOX, which have more than 85% homology at the amino acid level. NADPH oxidase is partially homologous to the gp91phox domain of the phagocyte Nox2, and may be the source of H2O2 production in human tracheobronchial epithelial cells [9–11]. Chan et al demonstrated that NADPH oxidase-mediated cell proliferation has a significant impact on wound healing [12]. Although DUOX is important in wound repair, its function and roles in regulation remain unknown.
Thus, in this study, primary fibroblasts were isolated from slow-healing burn wounds, fast-healing burn wounds, and skin tissues of traumatic resection in order to evaluate the potential role of DUOX1 in wound healing. We found that the expression level of DUOX1 was correlated with the speed of wound healing. Moreover, the expression of DUOX1 was significantly correlated with cell proliferation and ROS production. In addition, DUOX1 silencing significantly upregulated the protein levels of collagen I, collagen III, and NF-κB in the cytoplasm, and inhibited the protein levels of P21, P16, and NF-κB in the nucleus. Notably, the expression levels of these proteins exhibited a reverse trend following treatment with the ROS inhibitor N-acetyl-L-cysteine (NAC).
Material and Methods
PATIENT SAMPLES:
After informed consent was obtained, 12 clinical samples (male: n=6, female: n=6; age, 25–30 years) of granulation tissues with good healing; 12 cases (male: n=6, female: n=6; age, 25–30 years) of granulation tissues with slow healing from patients with burned wounds (second-degree burns); and 6 cases (male: n=3, female: n=3; age, 25–30 years) of normal skin tissues were collected. Burns were classified as follows: First-degree (superficial) burns: First-degree burns affect only the outer layer of skin, the epidermis. The burn site is red, painful, dry, and with no blisters. Mild sunburn is an example. Long-term tissue damage is rare and often consists of an increase or decrease in the skin color; Second-degree (partial thickness) burns: Second-degree burns involve the epidermis and part of the lower layer of skin, the dermis. The burn site looks red, blistered, and may be swollen and painful; Third-degree (full thickness) burns: Third-degree burns destroy the epidermis and dermis. They may go into the innermost layer of skin, the subcutaneous tissue. The burn site may look white or blackened and charred; Fourth-degree burns: Fourth-degree burns go through both layers of the skin and underlying tissue as well as deeper tissue, possibly involving muscle and bone. There is no feeling in the area since the nerve endings are destroyed. In the current study, the healing speed was judged according to the wound healing (the area size) of the burn area in the same time (Supplementary Figure 1). This study was approved by the Ethics Committee of GongLi Hospital, Pudong District, Shanghai, and was performed in accordance with the Declaration of Helsinki.
ISOLATION OF PRIMARY FIBROBLASTS AND CELL CULTURE:
Primary fibroblasts were isolated from granulation tissue of slow-healing wounds. The tissues were first washed 3 times with PBS containing 1% penicillin and streptomycin, before the heavily polluted granulation tissues and blood vessels were removed. The tissues were cut into pieces in a petri dish using ophthalmic scissors, and then digested in 0.1% collagenase and 0.1% bovine serum albumin (1: 2; C0130, Sigma) at 37°C on a shaker (T1300-100, Solarbio) for 2 h. The reaction was terminated by addition of 10% fetal bovine serum (FBS; 16000-044, GIBCO), and the tissue suspension was filtered using a 200-molybdenum filter. The filter fluid was centrifuged for 10 min at 1000 rpm, and the pellet was resuspended in DMEM medium (SH30243.01; HyClone) containing 10% FBS and 1% penicillin and streptomycin. Finally, the cell suspension was seeded in 6-well plates and cultured in a CO2 incubator (5% CO2, Thermo Forma 3111; Thermo) at 37°C. The culture medium was refreshed after the first 48 h in vitro, and was subsequently changed every 2 to 3 days according to the cell morphology and growth. Subculturing was performed when the cell confluence reached 80%.
DUOX1 OVEREXPRESSION AND INTERFERENCE CONSTRUCTION:
The coding sequence of DUOX1 (NM_017434.5) was synthesized using primers containing the restriction enzyme cutting sites of HindIII and EcoR I integrated into a pcDNA3.1(+) vector (Addgene, USA) to elevate DUOX1 overexpression:
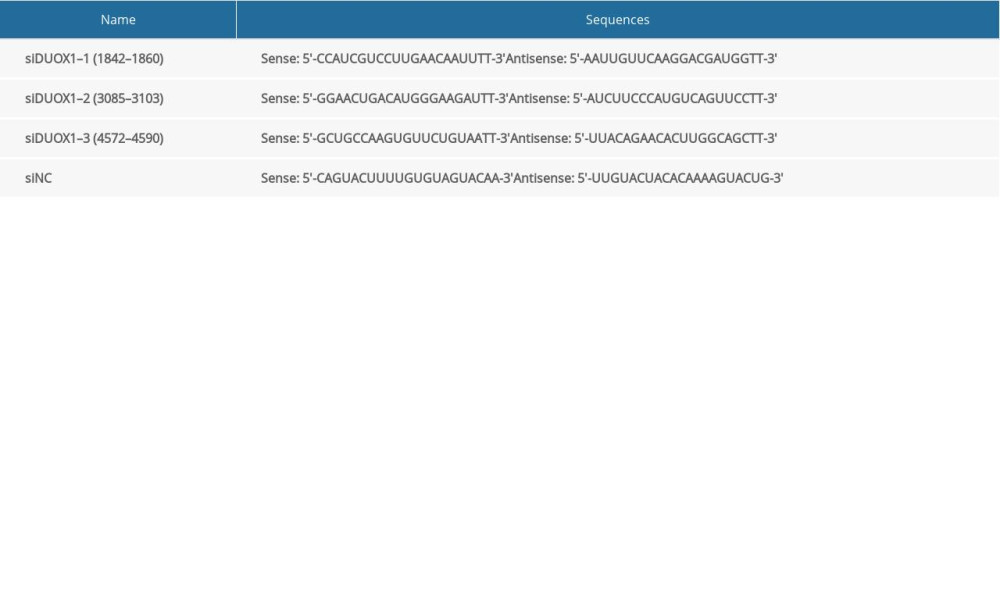
SiRNA targeting DUOX1 (NM_017434.5) coding sequences (CDS) were synthesized, and then inserted into a pcDNA3.1(+) vector (Addgene, USA) at Hind III/EcoR I sites. DUOX1 overexpression plasmids (oeDUOX1) were extracted using a Plasmid Extraction Kit (OMEGA). DUOX1 siRNA (siDUOX1) sequences (Table 1) were synthesized to downregulate DUOX1 expression.
CELL TRANSFECTION:
Primary fibroblasts in the logarithmic growth phase were trypsinized, and 2 ml of cell suspension was seeded into 6-well plates at a density of 2×105 cells/ml. The cells were cultured in serum-free transfer solution overnight at 37°C in a 5% CO2 incubator. The cells were transfected with siDUOX1-1, siDUOX1-2, siDUOX1-3 (200 pmol, 10 μl), and siNC (10 μl), or oeDUOX1 (200 pmol, 10 μl) and empty plasmids (vector, 10 μl) using Lipofectamine2000 (Invitrogen, USA) based on the manufacturer’s protocol. Serum-free transfer solution was used instead of DMEM medium supplemented with 10% FBS for the 6 h following transfection, and cells were harvested after a further 48 h.
BIOCHEMICAL DETECTION:
The cells were treated with siNC (negative control of siRNA) or siDOUX1-1, siDUOX1-2; or vehicle+vector, vehicle+oeDUOX1, NAC+vector, or NAC+oeDUOX1. Following treatment, the cells were collected and the supernatants were obtained to detect MDA and SOD levels using an MDA (A003-2) or SOD (A001-1) kit (Nanjing Jiancheng Biotechnology Research Institute, Jiangsu, China). Following the manufacturer’s instructions, the kit components were mixed with the samples in test tubes using a vortex mixer. The mouth of the test tube was fastened with a plastic wrap, and a small hole was made with a needle. The samples were incubated in a water bath at 95°C for 40 min, and the absorbance at 532 nm was measured.
QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (RT-PCR) ASSAY:
The cells were treated with control, siNC, siDUOX1-1, siDUOX1-2, siDUOX1-3, or control, vector, or oeDUOX1. The total RNA was extracted using TRIzol (1596-026; Invitrogen) according the manufacturer’s recommendations. After quantification and RNA integrity had been confirmed, the extracted RNA was reverse-transcribed into cDNA using a Reverse Transcription Kit (#K1622; Fermentas, Lithuania). Subsequently, using cDNA as template, qRT-PCR was conducted using an ABI 7300 Real-Time PCR system (ABI-7300; Applied Biosystems, Foster City, CA, USA) with a SYBR Green PCR Kit (#K0223; Thermo Fisher Scientific, Inc., Waltham, MA, USA). Reactions were performed in triplicate and were normalized to GAPDH. The ARHGAP15 mRNA level was calculated by the method of 2−ΔΔCq. The qRT-PCR procedure was as follows: 95°C for 10 min; (95°C for 15 s; 60°C for 45 s)×40. The primer sequences were as follows:
WESTERN BLOT ANALYSIS:
The cells were treated with control, siNC, siDUOX1-1, siDUOX1-2, or vehicle+vector, vehicle+oeDUOX1, NAC+vector, or NAC+oeDUOX1. Following treatment, the cells were collected, and the nuclear and cytoplasmic protein was extracted using a Nuclear/Cytoplasmic Protein Extraction Reagent Kit (P0028, Beyotime). The protein level was determined using the BCA protein assay kit (Thermo Fisher Scientific, PICPI23223). The sample was first boiled at 95ºC for 10 min, and 25 μg was then separated through SDS-PAGE gels with 10% concentration (JRDUN Biotechnology Co., Ltd., Shanghai, China). After electrophoresis, the samples were transferred to PVDF membranes (Millipore, HATF00010, Bedford, MA, USA), and subjected to blocking with 5% skim milk for 1 h at room temperature. The membrane was then incubated with primary antibodies against DUOX1 (1: 1000, orb589688; Biorbyt), collagen I (1: 1000, Ab138492; Abcam), collagen III (1: 1000, Ab184993; Abcam), p21 (1: 2000, Ab109520; Abcam), p16 (1: 5000, Ab51243; Abcam), NF-κB (1: 2000, Ab16502; Abcam), H3 (1: 1000, Ab1791; Abcam), and GAPDH (1: 2000, #5174; Cell Signaling Technology [CST]) overnight at 4°C with gentle shaking. After 5–6 washes with TBST (TBS+Tween), the membranes were incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (1: 1000; Beyotime, Shanghai, China) for 1 h at room temperature. After incubation, the PVDF membranes were washed with TBST another 5–6 times. Finally, the HRP signal was treated with chemiluminescent reagent (Millipore, WBKLS0100) and detected using an ECL imaging system (Tanon, Tanon-5200; Shanghai, China).
β-actin was used as an endogenous reference, and the levels of relative protein were quantified by ImageJ software (Bethesda, MD, USA).
PROLIFERATION ASSAY:
Cell proliferation was assessed using a Cell Counting Kit-8 (CCK-8; Signalway Antibody [SAB], CP002, Pierland, Texas, USA) solution. Primary fibroblasts with a density of 3×103 cells/well were seeded in a 96-well plate (TRUELINE, TR4001; USA). The next day, the cells were transfected with siDUOX1-1, siDUOX1-2, and siNC, or oeDUOX1 and vector. Subsequently, the CCK-8 and serum-free medium (100 μl; 1: 10) was added to the plate after transfection for 0, 24, 48, and 72 h. The plate was further incubated for another 1 h at 37°C. Finally, cell proliferation was quantified using a microplate reader (Perlong, DNM-9602; Beijing, China) based on the absorbance value (OD) at 450 nm.
ROS DETECTION:
ROS were detected using DCFH-DA probe solution. To obtain a 10-μM staining working solution, a 10-mM staining working solution was diluted with serum-free medium (1: 1000). The cells were then incubated with the DCFH-DA staining working solution in the dark at 37°C for 20 min. The cells were then washed 3 times using serum-free medium, and ROS production was measured by flow cytometry.
CELL APOPTOSIS ASSAY:
Flow cytometry analysis was applied to evaluate cell apoptosis. Primary fibroblasts were collected and stained with Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) (Beyotime, C1063). According to the manufacturer’s instructions, 5×105-1×106 cells were resuspended in 195 μl Annexin V-FITC binding buffer, followed by incubation in 5 μl Annexin V-FITC for 15 min at 4°C in the dark. Subsequently, the cells were incubated in 5 μl PI for 5 min at 4°C in the dark. A tube without Annexin V-FITC and PI was used as a control. Percentages of apoptotic cells were determined on a flow cytometer with a BD Accuri™ C6 Software (Version 1.0.264.21; BD Biosciences, USA).
IMMUNOHISTOCHEMISTRY (IHC):
Primary fibroblasts were first washed with 0.02 M PBS on glass coverslips, followed by treatment with 4% paraformaldehyde (10010018, National Medicines Corporation Ltd.) for 30 min to fix cells. The cells were then permeated using 3% H2O2 (10011218; Shanghai Sinopharm) for 10 min. In order to reduce background interference, cells were blocked using 1% BSA (A8010, Solarbio) at room temperature for 1 h. Primary antibodies against vimentin (ab8978; Abcam) were added to cells incubating for 1 h in a humidifying box at room temperature. After incubation, the cells were further washed using PBST to remove residual antibodies. Subsequently, HRP-labeled secondary antibodies (D-3004; Changdao) were added, and the cells were incubated for 30 min at room temperature. The slices were washed with tap water after DAB staining (FL-6001, Changdao), and hematoxylin (714094; BASO) was used to counterstain the slices. Alcohol differentiation was further performed using 1% hydrochloric acid. After washing by tap water, the slices were heated, transparentized, and sealed, followed by imaging on a microscope (ECLIPSE Ni, NIKON).
STATISTICAL ANALYSIS:
All experiments were performed 3 times, and data are presented as the mean±standard deviation (SD). Statistical analysis was performed using GraphPad prism 7.0 software (GraphPad Software, San Diego, CA, USA). One-way analysis of variance with Tukey’s multiple comparison post hoc tests was applied for multiple groups. A value of
Results
EXPRESSION OF DUOX1 IN WOUND HEALING:
The mRNA levels of DUOX1 in the 3 groups (slow-healing tissues, fast-healing tissues, and normal tissues) were assessed using qRT-PCR. Figure 1A shows that expression of DUOX1 was higher in slow-healing tissues compared to that in fast-healing tissues (P<0.05). In contrast, slow-healing tissues had less expression of collagen I and III than fast-healing tissues (Figure 1B). In addition, cell proliferation was significantly increased in fast-healing tissues compared to that in slow-healing tissues (Figure 1C).
KNOCKDOWN OF DUOX1 SIGNIFICANTLY PROMOTED CELL PROLIFERATION, AND INHIBITED ROS:
IHC staining showed positive expression of vimentin, suggesting that the fibroblasts were successfully separated (Figure 2A). Moreover, both the mRNA and protein levels of DUOX1 were significantly decreased in the siDUOX1-1, siDUOX1-2, and siDUOX1-3 groups compared to the siNC group, suggesting that DUOX1 knockdown was successful (P<0.001) (Figure 2B, 2C). The CCK-8 assay showed that cell proliferation in the siDUOX1-1 and siDUOX1-2 groups was significantly increased by approximately 30% compared to the siNC group 72 h after transfection (P<0.001) (Figure 2D). Moreover, the MDA level was significantly reduced, and the SOD level was significantly increased in the siDUOX1-1 and siDUOX1-2 groups (P<0.001) (Figure 2E, 2F). ROS production and cell apoptosis were detected using flow cytometry, and the results showed that both were significantly inhibited after silencing DUOX1 (P<0.001) (Figure 2G, 2H). After silencing DUOX1, the protein levels of collagen I, collagen III, and NF-κB in the cytoplasm were significantly increased, while the protein levels of P21, P16, and NF-κB in the nucleus were significantly decreased (Figure 2I).
DUOX1 REGULATES WOUND HEALING, LIKELY VIA THE REGULATION OF ROS:
As shown in Figure 3A and 3B, fibroblasts with DUOX1 overexpression were successfully constructed at both the mRNA and protein levels. Our results demonstrated that overexpression of DUOX1 significantly increased MDA, ROS production, and cell apoptosis 24 h after pre-transfection of the primary fibroblasts (P<0.001); in contrast, a significant decrease in SOD and cell proliferation was observed (by approximately 27.49%) (P<0.001) (Figure 3C–3G). Moreover, the protein levels of collagen I, collagen III, and NF-κB in the cytoplasm were significantly decreased, while the protein levels of P21, P16, and NF-κB in the nucleus were significantly increased (Figure 3H). The ROS inhibitor NAC was added to further investigate the molecular mechanism of DUOX1 in wound healing. As shown in Figure 3C–3H, NAC partly canceled the effect of oeDUOX1 on MDA, ROS production, cell apoptosis, and protein expression of P21, P16, and NF-κB in the nucleus, as well as the inhibition of SOD, cell proliferation (increase by approximately 47.12%), and protein expression of collagen I, collagen III, and NF-κB in the cytoplasm (P<0.05). Therefore, we speculate that DUOX1 regulates wound healing by regulating ROS.
Discussion
Complex chronic wounds are a serious problem and impose a heavy financial burden on healthcare systems. In recent years, different types of wound dressing, such as films, hydrocolloids, hydrogels, and micro/nano fibers have been developed [13]. Several studies have revealed that bioactive hydrogel, electrospun membranes loaded with bioactive molecules, and biofunctionalized electrospun silk mats can improve the wound healing process [14–16]. However, more effective treatments are required to accelerate wound healing.
DUOX1 is a member of the NADPH oxidase (NOX) family [17, 18], which has a role in forming a hydrogen peroxide (H2O2) tissue gradient after injury [19]. NADPH oxidase 4 deficiency is reported to benefit wound healing [20]. In addition, a number of studies have shown the role of DUOX1 in wound healing, in which H2O2 produced by DUOX1 acts as a signaling molecule to regulate adhesion and migration in epithelial regeneration after injury [21–24]. Here, we found that the expression level of DUOX1 was negatively correlated with the speed of burn healing. Downregulation of DUOX1 significantly increased cell proliferation and decreased ROS production and apoptosis in fibroblasts isolated from the tissues with slow healing wounds. DUOX1 overexpression had an opposite effect on cell proliferation, ROS production, and cell apoptosis, which was potently reversed by NAC, a scavenger of ROS [25]. Additionally, in wound healing, collagen [26], P21, and P16 [27] are key components involved in tissue repair. Collagen is an important scaffold structure of the skin, and it was found that collagen type I and III are the 2 most abundant collagen components in the skin [28]. Our data show that DUOX1 silencing significantly upregulated the protein levels of collagen I and III and inhibited the protein levels of P21 and P16. Evidence has shown that ROS, which are second messengers in controlling cell proliferation and differentiation [29,30], have key roles in complex cellular processes, such as angiogenesis, inflammation, and proliferation. Pedro et al showed that the expression of DUOX1 was related to genes involved in oxidative stress [31]. Consistent with the findings that oxidative stress plays an important role in wound healing [32,33], our results suggest that DUOX1 downregulation promoted burn wound healing, probably by inhibiting ROS production.
Further, the potential mechanism of DUOX1 in regulating burn wound healing was explored. The wound microenvironment is important for the wound healing process, and a sequence of molecular and cellular events, such as inflammatory reaction, cell proliferation, and extracellular matrix synthesis, occur during wound repair [34]. NF-κB is a multipotent nuclear transcription factor that normally exists in the cytoplasm as an inactive form. Following stimulation by external factors, NF-κB can enter the nucleus and regulate the transcription and expression of target genes. Zhang et al found that inhibition of NF-κB activation could counteract oxidative stress-induced cardiomyocyte apoptosis [35]. The expression of several pro-inflammatory genes has been shown to be related with NF-κB, which is recognized as a master regulator of inflammation [36]. Although there are still many inconsistencies concerning the influence of oxidative stress on NF-κB activity, several studies have shown that oxidative stress can act as a regulator of NF-κB kinases using in vivo cultured cell models [10]. Previous studies have demonstrated that NF-κB activity is regulated by NADPH oxidases and is sensitive to redox [37]. Moreover, in the early modulation of inflammatory responses during wound healing, signals such as ATP and H2O2, as DUOX1 activation modulators, are necessary for NF-κB signaling [38]. Consistently, our results demonstrated that knockdown of DUOX1 inhibited NF-κB activation, while DUOX1 overexpression enhanced NF-κB activation. Several studies have shown that NF-κB plays an important regulatory role in lymphocyte activation, lymphocyte apoptosis, and lymphocyte-mediated cytotoxicity [39,40]. From these results, we speculated that downregulation of DUOX1 can regulate the immunomodulatory function of host stem cells by inhibiting the activation of NF-κB and oxidative stress, thereby regulating wound healing. Of course, this speculation needs to be verified by further experiments.
Conclusions
This research demonstrated the importance of DUOX1 in burn wound repair. Our data indicate that downregulation of DUOX1 may benefit burn wound healing by inhibiting oxidative stress via inhibiting NF-κB pathway activation. Although further investigations on this topic are needed, our study identifies a potential therapeutic approach for burn wound healing, and also provides a theoretical and experimental basis for subsequent research.
Figures
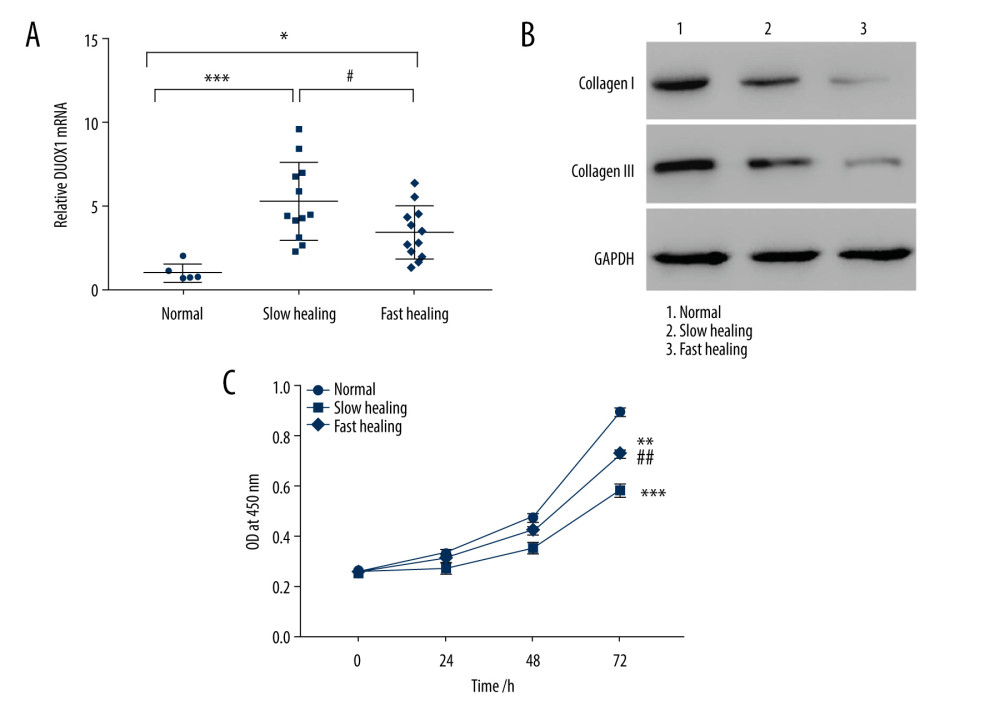 Figure 1. DUOX1 was highly expressed in slow-healing granulation tissue of burn wounds (A) Clinical burned wounds with fast healing (n=12) and slow healing (n=12) granulation tissue and normal tissue (n=6) were collected, and Q-PCR was used to detect the expression of DUOX1. Primary fibroblasts were isolated from fast-healing, slow-healing, and normal tissues. (B) Collagen I and III protein levels were detected by Western blotting. (C) Cell proliferation was detected by CCK8 at 0, 24, 48, and 72 h. * P<0.05, ** P<0.01, *** P<0.001 vs normal; # P<0.05, ## P<0.01 vs slow-healing. Normal tissues: skin tissue taken from the surface of traumatic injury, fast-healing granulation tissue: better recovery after 14 days’ treatment, slow-healing (n=12) granulation tissue: poor recovery after 14 days’ treatment.
Figure 1. DUOX1 was highly expressed in slow-healing granulation tissue of burn wounds (A) Clinical burned wounds with fast healing (n=12) and slow healing (n=12) granulation tissue and normal tissue (n=6) were collected, and Q-PCR was used to detect the expression of DUOX1. Primary fibroblasts were isolated from fast-healing, slow-healing, and normal tissues. (B) Collagen I and III protein levels were detected by Western blotting. (C) Cell proliferation was detected by CCK8 at 0, 24, 48, and 72 h. * P<0.05, ** P<0.01, *** P<0.001 vs normal; # P<0.05, ## P<0.01 vs slow-healing. Normal tissues: skin tissue taken from the surface of traumatic injury, fast-healing granulation tissue: better recovery after 14 days’ treatment, slow-healing (n=12) granulation tissue: poor recovery after 14 days’ treatment. 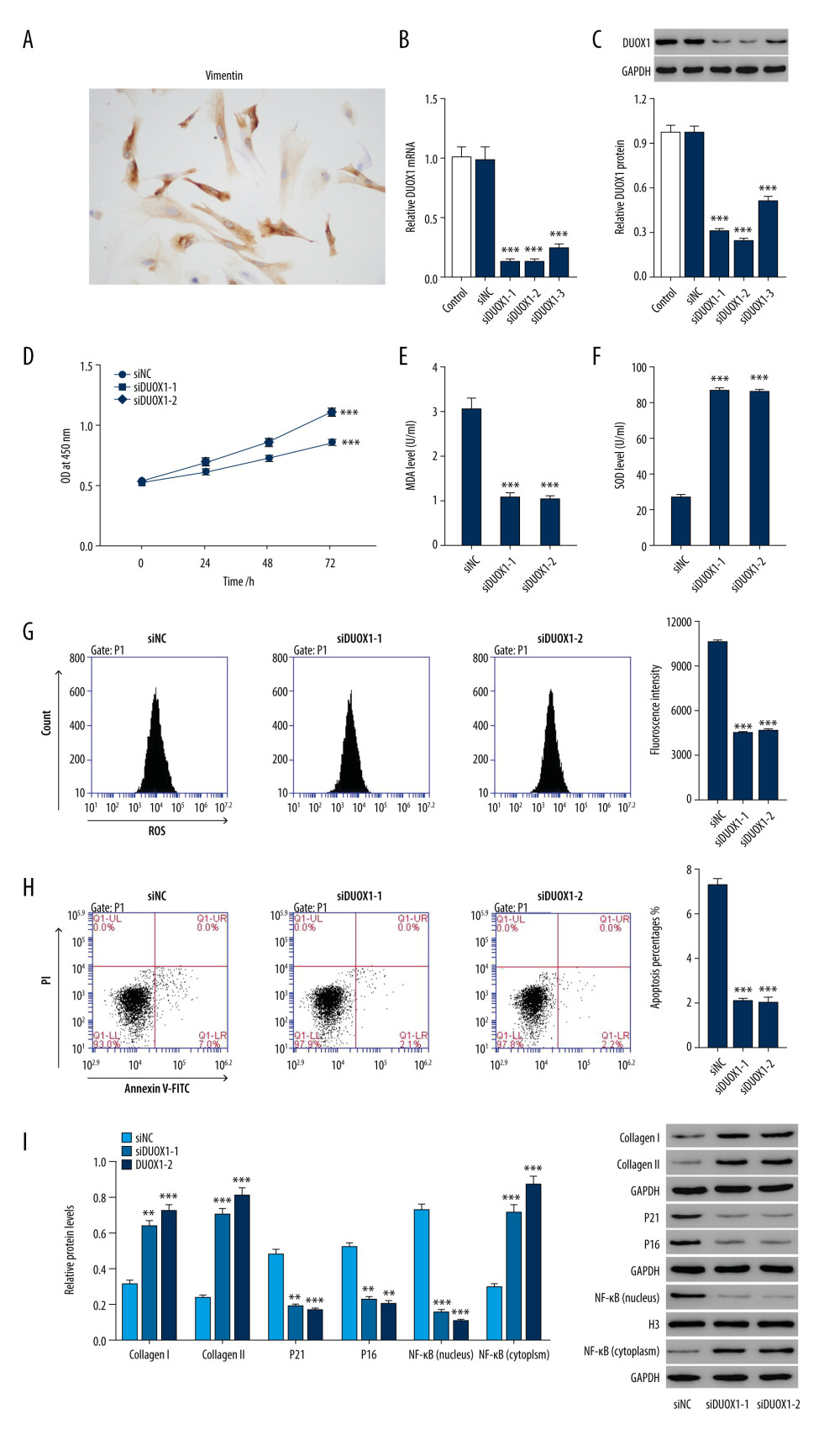 Figure 2. Knockdown of DUOX1 significantly increased cell proliferation and inhibited the production of reactive oxygen species (ROS). (A) Following primary isolation from wound granulation tissue, fibroblasts (Vimentin-positive) were identified by immunohistochemical staining. (B, C) Primary fibroblasts were transfected with human DUOX1 interferon, and the interference efficiency of DUOX1 was detected by Q-PCR (B) and Western blot (C). Following DUOX1 gene interference in primary fibroblasts, (D) a CCK-8 kit was used to detect cell proliferation; (E, F) biochemical detection of cell supernatant MDA (E) and SOD (F) expression; (G, H) flow detection of ROS and (G) cell apoptosis (H); (I) Western blotting of NF-κB (nuclear and plasma), collagen, collagen III, P21, and P16. ** P<0.01, *** P<0.001 vs siNC. siNC – negative control of DUOX1 interference.
Figure 2. Knockdown of DUOX1 significantly increased cell proliferation and inhibited the production of reactive oxygen species (ROS). (A) Following primary isolation from wound granulation tissue, fibroblasts (Vimentin-positive) were identified by immunohistochemical staining. (B, C) Primary fibroblasts were transfected with human DUOX1 interferon, and the interference efficiency of DUOX1 was detected by Q-PCR (B) and Western blot (C). Following DUOX1 gene interference in primary fibroblasts, (D) a CCK-8 kit was used to detect cell proliferation; (E, F) biochemical detection of cell supernatant MDA (E) and SOD (F) expression; (G, H) flow detection of ROS and (G) cell apoptosis (H); (I) Western blotting of NF-κB (nuclear and plasma), collagen, collagen III, P21, and P16. ** P<0.01, *** P<0.001 vs siNC. siNC – negative control of DUOX1 interference. 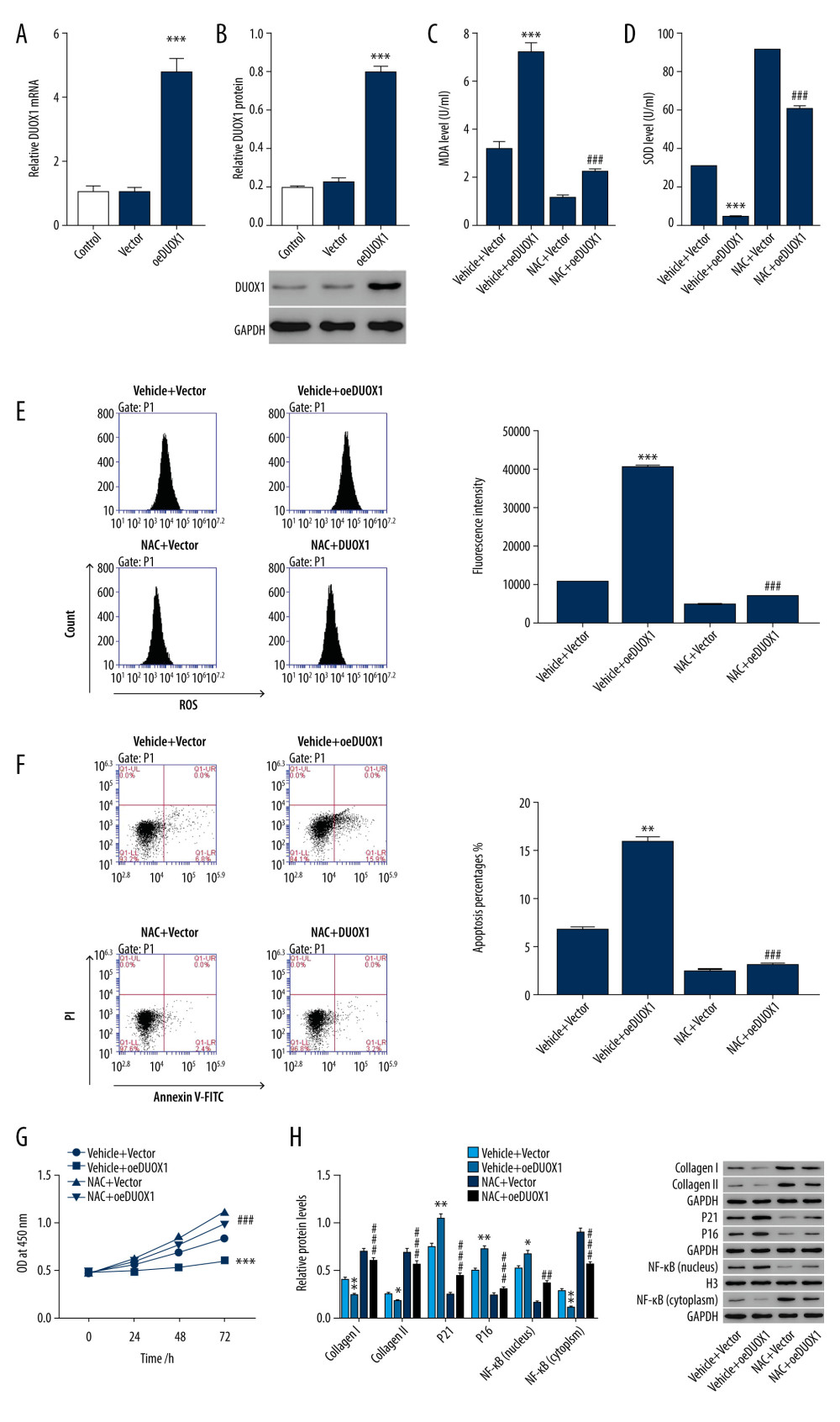 Figure 3. DUOX1-regulated wound healing, likely by regulation of reactive oxygen species (ROS). (A, B) Transfected primary fibroblasts with overexpression of human DUOX1 were constructed, and Q-PCR (A) and Western blot (B) were used to detect the efficiency of DUOX1 overexpression. Following pre-transfection of DUOX1 overexpression in primary fibroblasts for 24 h, cells were treated with NAC and ROS inhibitor (10 μM). (C, D) Expression of MDA (C) and SOD (D) were detected in cell supernatants; (E, F) flow cytometry was used to detect ROS and (E) cell apoptosis (F); (G) cell proliferation activity was detected using a CCK-8 kit; (H) expression of NF-κB (nuclear and plasma), collagen I, collagen III, P21, and P16 were detected by Western blotting. * P<0.05, ** P<0.01, *** P<0.001 vs vector or vehicle+vector; ## P<0.01, ### P<0.001 vs vehicle+oeDUOX1.
Figure 3. DUOX1-regulated wound healing, likely by regulation of reactive oxygen species (ROS). (A, B) Transfected primary fibroblasts with overexpression of human DUOX1 were constructed, and Q-PCR (A) and Western blot (B) were used to detect the efficiency of DUOX1 overexpression. Following pre-transfection of DUOX1 overexpression in primary fibroblasts for 24 h, cells were treated with NAC and ROS inhibitor (10 μM). (C, D) Expression of MDA (C) and SOD (D) were detected in cell supernatants; (E, F) flow cytometry was used to detect ROS and (E) cell apoptosis (F); (G) cell proliferation activity was detected using a CCK-8 kit; (H) expression of NF-κB (nuclear and plasma), collagen I, collagen III, P21, and P16 were detected by Western blotting. * P<0.05, ** P<0.01, *** P<0.001 vs vector or vehicle+vector; ## P<0.01, ### P<0.001 vs vehicle+oeDUOX1. References
1. Tricco AC, Cogo E, Isaranuwatchai W, A systematic review of cost-effectiveness analyses of complex wound interventions reveals optimal treatments for specific wound types: BMC Med, 2015; 13; 90
2. Smith RE, Ozben T, Saso L, Modulation of oxidative stress: Pharmaceutical and pharmacological aspects 2018: Oxid Med Cell Longev, 2019; 2019 6380473
3. Wlaschek M, Scharffetter-Kochanek K, Oxidative stress in chronic venous leg ulcers: Wound Repair Regen, 2005; 13; 452-61
4. Zhao HB, Lin SQ, Liu JH, Lin ZB, Polysaccharide extract isolated from ganoderma lucidum protects rat cerebral cortical neurons from hypoxia/reoxygenation injury: J Pharmacol Sci, 2004; 95; 294-98
5. Martinez MA, Rodriguez JL, Lopez-Torres B, Oxidative stress and related gene expression effects of cyfluthrin in human neuroblastoma SH-SY5Y cells: Protective effect of melatonin: Environ Res, 2019; 177; 108579
6. Dekoninck S, Blanpain C, Stem cell dynamics, migration and plasticity during wound healing: Nat Cell Biol, 2019; 21; 18-24
7. Lee J, Shin D, Roh JL, Promotion of skin wound healing using prevascularized oral mucosal cell sheet: Head Neck, 2019; 41; 774-79
8. Lambeth JD, Nox/Duox family of nicotinamide adenine dinucleotide (phosphate) oxidases: Curr Opin Hematol, 2002; 9; 11-17
9. Forteza R, Salathe M, Miot F, Regulated hydrogen peroxide production by Duox in human airway epithelial cells: Am J Respir Cell Mol Biol, 2005; 32; 462-69
10. Geiszt M, Witta J, Baffi J, Dual oxidases represent novel hydrogen peroxide sources supporting mucosal surface host defense: FASEB J, 2003; 17; 1502-4
11. Schwarzer C, Machen TE, Illek B, Fischer H, NADPH oxidase-dependent acid production in airway epithelial cells: J Biol Chem, 2004; 279; 36454-61
12. Chan EC, Jiang F, Peshavariya HM, Dusting GJ, Regulation of cell proliferation by NADPH oxidase-mediated signaling: Potential roles in tissue repair, regenerative medicine and tissue engineering: Pharmacol Ther, 2009; 122; 97-108
13. Miguel SP, Sequeira RS, Moreira AF, An overview of electrospun membranes loaded with bioactive molecules for improving the wound healing process: Eur J Pharm Biopharm, 2019; 139; 1-22
14. Gao L, Zhou Y, Peng J, A novel dual-adhesive and bioactive hydrogel activated by bioglass for wound healing: NPG Asia Materials, 2019; 11; 66
15. Schneider A, Wang X, Kaplan DL, Biofunctionalized electrospun silk mats as a topical bioactive dressing for accelerated wound healing: Acta Biomater, 2009; 5; 2570-78
16. Gama FM, Silva , João P, Improved burn wound healing using a bioactive peptide; 36
17. Sumimoto H, Structure, regulation and evolution of Nox-family NADPH oxidases that produce reactive oxygen species: FEBS J, 2008; 275; 3249-77
18. Bedard K, Krause KH, The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology: Physiol Rev, 2007; 87; 245-313
19. Razzell W, Evans IR, Martin P, Wood W, Calcium flashes orchestrate the wound inflammatory response through DUOX activation and hydrogen peroxide release: Curr Biol, 2013; 23; 424-29
20. Levigne D, Modarressi A, Krause KH, Pittet-Cuenod B, NADPH oxidase 4 deficiency leads to impaired wound repair and reduced dityrosine-crosslinking, but does not affect myofibroblast formation: Free Radic Biol Med, 2016; 96; 374-84
21. Niethammer P, Grabher C, Look AT, Mitchison TJ, A tissue-scale gradient of hydrogen peroxide mediates rapid wound detection in zebrafish: Nature, 2009; 459; 996-99
22. Rieger S, Sagasti A, Hydrogen peroxide promotes injury-induced peripheral sensory axon regeneration in the zebrafish skin: PLOS Biol, 2011; 9; e1000621
23. Juarez MT, Patterson RA, Sandovalguillen E, Mcginnis W, Duox, Flotillin-2, and Src42A are required to activate or delimit the spread of the transcriptional response to epidermal wounds in Drosophila: PLOS Genet, 2011; 7; e1002424
24. Gorissen S, Hristova M, Habibovic A, Dual oxidase-1 is required for airway epithelial cell migration and bronchiolar reepithelialization after injury: Am J Respir Cell Mol Biol, 2013; 48; 337-45
25. Downs I, Liu J, Aw TY, The ROS scavenger, NAC, regulates hepatic Valpha14iNKT cells signaling during Fas mAb-dependent fulminant liver failure: PLoS One, 2012; 7; e38051
26. Brett D, A review of collagen and collagen-based wound dressings: Wounds, 2008; 20; 347-56
27. Zhu X, Di Y, Hu C, Wang Z, Expression of positive and negative regulators of cell cycle during wound healing: Chin Med J (Engl), 2002; 115; 326-30
28. Riekki R, Parikka M, Jukkola A, Increased expression of collagen types I and III in human skin as a consequence of radiotherapy: Arch Dermatol Res, 2002; 294; 178-84
29. Sauer H, Wartenberg M, Hescheler J, Reactive oxygen species as intracellular messengers during cell growth and differentiation: Cell Physiol Biochem, 2001; 11; 173-86
30. Huo Y, Qiu WY, Pan Q, Reactive oxygen species (ROS) are essential mediators in epidermal growth factor (EGF)-stimulated corneal epithelial cell proliferation, adhesion, migration, and wound healing: Exp Eye Res, 2009; 89; 876-86
31. Pedro NF, Biselli JM, Maniglia JV, Candidate biomarkers for oral squamous cell carcinoma: Differential expression of oxidative stress-related genes: Asian Pac J Cancer Prev, 2018; 19; 1343-49
32. Ojalvo AG, Acosta JB, Marí YM, Healing enhancement of diabetic wounds by locally infiltrated epidermal growth factor is associated with systemic oxidative stress reduction: Int Wound J, 2017; 14; 214-25
33. Babajafari S, Akhlaghi M, Mazloomi SM, The effect of isolated soy protein adjunctive with flaxseed oil on markers of inflammation, oxidative stress, acute phase proteins, and wound healing of burn patients;A randomized clinical trial: Burns, 2018; 44; 140-49
34. Gonzalez AC, Costa TF, Andrade ZA, Medrado AR, Wound healing – A literature review: An Bras Dermatol, 2016; 91; 614-20
35. Zhang Q, Huang WD, Lv XY, Yang YM, Ghrelin protects H9c2 cells from hydrogen peroxide-induced apoptosis through NF-kappaB and mitochondria-mediated signaling: Eur J Pharmacol, 2011; 654; 142-49
36. Kawai T, Akira S, Signaling to NF-kappaB by Toll-like receptors: Trends Mol Med, 2007; 13; 460-69
37. Bonizzi G, Piette J, Schoonbroodt S, Reactive oxygen intermediate-dependent NF-kappaB activation by interleukin-1beta requires 5-lipoxygenase or NADPH oxidase activity: Mol Cell Biol, 1999; 19; 1950-60
38. de Oliveira S, Lopez-Munoz A, Candel S: J Immunol, 2014; 192; 5710-19
39. Li Q, Verma IM, NF-κB regulation in the immune system: Nat Rev Immunol, 2002; 2; 725-34
40. Taniguchi K, Karin M, NF-κB, inflammation, immunity and cancer: Coming of age: Nat Rev Immunol, 2018; 18; 309-24
Figures
 Figure 1. DUOX1 was highly expressed in slow-healing granulation tissue of burn wounds (A) Clinical burned wounds with fast healing (n=12) and slow healing (n=12) granulation tissue and normal tissue (n=6) were collected, and Q-PCR was used to detect the expression of DUOX1. Primary fibroblasts were isolated from fast-healing, slow-healing, and normal tissues. (B) Collagen I and III protein levels were detected by Western blotting. (C) Cell proliferation was detected by CCK8 at 0, 24, 48, and 72 h. * P<0.05, ** P<0.01, *** P<0.001 vs normal; # P<0.05, ## P<0.01 vs slow-healing. Normal tissues: skin tissue taken from the surface of traumatic injury, fast-healing granulation tissue: better recovery after 14 days’ treatment, slow-healing (n=12) granulation tissue: poor recovery after 14 days’ treatment.
Figure 1. DUOX1 was highly expressed in slow-healing granulation tissue of burn wounds (A) Clinical burned wounds with fast healing (n=12) and slow healing (n=12) granulation tissue and normal tissue (n=6) were collected, and Q-PCR was used to detect the expression of DUOX1. Primary fibroblasts were isolated from fast-healing, slow-healing, and normal tissues. (B) Collagen I and III protein levels were detected by Western blotting. (C) Cell proliferation was detected by CCK8 at 0, 24, 48, and 72 h. * P<0.05, ** P<0.01, *** P<0.001 vs normal; # P<0.05, ## P<0.01 vs slow-healing. Normal tissues: skin tissue taken from the surface of traumatic injury, fast-healing granulation tissue: better recovery after 14 days’ treatment, slow-healing (n=12) granulation tissue: poor recovery after 14 days’ treatment. Figure 2. Knockdown of DUOX1 significantly increased cell proliferation and inhibited the production of reactive oxygen species (ROS). (A) Following primary isolation from wound granulation tissue, fibroblasts (Vimentin-positive) were identified by immunohistochemical staining. (B, C) Primary fibroblasts were transfected with human DUOX1 interferon, and the interference efficiency of DUOX1 was detected by Q-PCR (B) and Western blot (C). Following DUOX1 gene interference in primary fibroblasts, (D) a CCK-8 kit was used to detect cell proliferation; (E, F) biochemical detection of cell supernatant MDA (E) and SOD (F) expression; (G, H) flow detection of ROS and (G) cell apoptosis (H); (I) Western blotting of NF-κB (nuclear and plasma), collagen, collagen III, P21, and P16. ** P<0.01, *** P<0.001 vs siNC. siNC – negative control of DUOX1 interference.
Figure 2. Knockdown of DUOX1 significantly increased cell proliferation and inhibited the production of reactive oxygen species (ROS). (A) Following primary isolation from wound granulation tissue, fibroblasts (Vimentin-positive) were identified by immunohistochemical staining. (B, C) Primary fibroblasts were transfected with human DUOX1 interferon, and the interference efficiency of DUOX1 was detected by Q-PCR (B) and Western blot (C). Following DUOX1 gene interference in primary fibroblasts, (D) a CCK-8 kit was used to detect cell proliferation; (E, F) biochemical detection of cell supernatant MDA (E) and SOD (F) expression; (G, H) flow detection of ROS and (G) cell apoptosis (H); (I) Western blotting of NF-κB (nuclear and plasma), collagen, collagen III, P21, and P16. ** P<0.01, *** P<0.001 vs siNC. siNC – negative control of DUOX1 interference. Figure 3. DUOX1-regulated wound healing, likely by regulation of reactive oxygen species (ROS). (A, B) Transfected primary fibroblasts with overexpression of human DUOX1 were constructed, and Q-PCR (A) and Western blot (B) were used to detect the efficiency of DUOX1 overexpression. Following pre-transfection of DUOX1 overexpression in primary fibroblasts for 24 h, cells were treated with NAC and ROS inhibitor (10 μM). (C, D) Expression of MDA (C) and SOD (D) were detected in cell supernatants; (E, F) flow cytometry was used to detect ROS and (E) cell apoptosis (F); (G) cell proliferation activity was detected using a CCK-8 kit; (H) expression of NF-κB (nuclear and plasma), collagen I, collagen III, P21, and P16 were detected by Western blotting. * P<0.05, ** P<0.01, *** P<0.001 vs vector or vehicle+vector; ## P<0.01, ### P<0.001 vs vehicle+oeDUOX1.
Figure 3. DUOX1-regulated wound healing, likely by regulation of reactive oxygen species (ROS). (A, B) Transfected primary fibroblasts with overexpression of human DUOX1 were constructed, and Q-PCR (A) and Western blot (B) were used to detect the efficiency of DUOX1 overexpression. Following pre-transfection of DUOX1 overexpression in primary fibroblasts for 24 h, cells were treated with NAC and ROS inhibitor (10 μM). (C, D) Expression of MDA (C) and SOD (D) were detected in cell supernatants; (E, F) flow cytometry was used to detect ROS and (E) cell apoptosis (F); (G) cell proliferation activity was detected using a CCK-8 kit; (H) expression of NF-κB (nuclear and plasma), collagen I, collagen III, P21, and P16 were detected by Western blotting. * P<0.05, ** P<0.01, *** P<0.001 vs vector or vehicle+vector; ## P<0.01, ### P<0.001 vs vehicle+oeDUOX1. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952