07 July 2021: Clinical Research
Application of the RCOG Risk Assessment Model for Evaluating Postpartum Venous Thromboembolism in Chinese Women: A Case-Control Study
Ying-Zhou Ge123ACDEF, Chen Zhang123CD, Yan-Qing Cai123ABE, He-Feng Huang123AEG*DOI: 10.12659/MSM.929904
Med Sci Monit 2021; 27:e929904
Abstract
BACKGROUND: Since China has not yet constructed its own risk assessment model (RAM) for pregnancy-related venous thromboembolism (VTE), more and more hospitals use the RCOG RAM for VTE risk prediction. However, the RCOG RAM was established based on Western populations, and its applicability in China is still uncertain. Thus, we aimed to evaluate the validity of the RCOG RAM in predicting postpartum VTE in Chinese maternity.
MATERIAL AND METHODS: This retrospective case-control study was conducted at the International Peace Maternity and Child Health Hospital (IPMCHH) from June 2016 to June 2020. The VTE group consisted of 38 women with postpartum VTE. For each VTE patient, 4 women without VTE who gave birth on the same day were randomly selected as the control group (n=152). The receiver operating characteristic (ROC) curve, calibration curve, and decision curve analysis (DCA) were used to evaluate the discrimination, accuracy, and validity of the RCOG RAM. Univariable analysis and multivariable logistic regression analysis were used to identify other related factors for postpartum VTE.
RESULTS: Compared with the low-risk group, the risk of VTE was 9.75-fold greater in the intermediate-risk group, and 90.00-fold greater in the high-risk group. The area under curve (AUC) of the model was 0.828 (95% CI: 0.762-0.894), with a score of 2 as its best cut-off value, which exactly matched the criterion recommended by the RCOG guidelines for pharmacological thromboprophylaxis. The calibration curves and DCA of the model also showed good accuracy. In addition to the factors included in the RCOG RAM, glucocorticoid therapy during pregnancy (adjusted OR=6.72, 95% CI: 1.56-28.91) and previous use of IUD (adjusted OR=7.11, 95% CI: 1.45-34.93) were associated with increased risk of postpartum VTE.
CONCLUSIONS: The RCOG RAM was found to be effective in predicting postpartum VTE, and has certain guiding significance for postpartum thromboprophylaxis in China.
Keywords: Chemoprevention, Glucocorticoids, Intrauterine Devices, Risk Assessment, venous thromboembolism, Postpartum Period, Pregnancy, Pregnancy Complications, Cardiovascular, Risk Factors
Background
Venous thromboembolism (VTE) comprises deep venous thrombosis (DVT) and pulmonary embolism (PE). Because of increased concentrations of coagulation factors, decreased anticoagulant and fibrinolytic components, and increased compression of the uterus to the iliac vein and inferior vena cava [1], the risk of VTE for women during pregnancy and the puerperium period is approximately 4–5 times higher than that in non-pregnant women of the same age [2,3]. Meanwhile, the risk of postpartum VTE is 20 times higher than that of antepartum VTE [4]. The incidence of VTE is 1–2 per 1000 pregnancies in Europe and America [5,6]. Nearly 80% of pregnancy-related VTEs are isolated DVTs, and 20% are PEs or coexisting PE and DVT [7], whose fatality rate can be as high as 15% [8]. Currently, VTE has become one of the leading causes of maternal mortality worldwide [9].
In the past, insufficient attention was given to the prevention of pregnancy-related VTE in China, and until now, there has been no epidemiological analysis based on national data. The Wells score and the Caprini score are usually used in clinical practice for VTE risk assessment in the obstetric department, but neither is designed specifically for pregnant women [10]. In 2015, the Royal College of Obstetricians and Gynecologists (RCOG) released the latest edition of guidelines for the prevention of VTE during pregnancy and puerperium and updated the VTE risk scoring method (mentioned as RCOG risk assessment model below). The detailed scoring method for VTE is shown in Supplementary Table 1. This model has been widely used in Western countries for the assessment of the risk of pregnancy-related VTE in both the antenatal and postnatal periods, and a thromboprophylaxis management process based on the results of the model has been recommended by the RCOG guidelines. If the total score is ≥2 postnatally, thromboprophylaxis for at least 10 days is recommended [11]. However, the data used for building the RCOG RAM were mostly obtained from Western countries. At present, whether this model is suitable for Chinese maternity care remains poorly understood. In view of the higher prevalence of VTE during the postnatal period, this study aimed to evaluate the predictive value of the RCOG RAM for postpartum VTE and its guiding significance in postpartum thromboprophylaxis in China. Furthermore, we aimed to explore related factors for postpartum VTE other than those already included in the model.
Material and Methods
STUDY POPULATION:
This retrospective case-control study was conducted at the International Peace Maternity and Child Health Hospital (IPMCHH), a specialized hospital of obstetrics and genecology in Shanghai, from June 2016 to June 2020. Women diagnosed with VTE during the postpartum period constituted the VTE group. For each VTE patient, 4 women who gave birth at the same day and confirmed as without VTE were randomly selected as the control group. Inclusion criteria included women who gave birth beyond 28 weeks of gestation, regardless of delivery mode, and completed regular antenatal and postnatal examinations as well as diagnostic examinations for VTE. Exclusion criteria were missing or untraceable previous medical records.
The diagnosis of DVT was confirmed by compression ultrasonography of the lower limb veins [12,13], and once thrombotic events were detected, the patient was routinely checked by echocardiography and computer tomography pulmonary angiography (CTPA) to investigate the presence of PE. All women were encouraged to walk as soon as possible after delivery. For those who underwent cesarean section, a pneumatic compression device was routinely used. Very few women received pharmacological prophylaxis, which was mainly based on empirical medication.
DATA COLLECTION:
Data collected from medical records included socio-demographic characteristics, reproductive history, gynecologic history, previous contraceptive use, obstetric characteristics, and the results of laboratory tests (coagulation-related parameters) and imaging examinations. All women returned to the hospital for routine check-ups 42 days after delivery, and relevant data were also collected.
ASSESSMENT OF POSTPARTUM VTE RISK:
Based on the above information, the RCOG RAM was used to assess the risk score and risk level of postpartum VTE. The risk of postpartum VTE was divided into 3 levels: low risk (<2 points and without intermediate or high-risk factors, no need for thromboprophylaxis), intermediate risk (≥2 points and without high-risk factors, need for thromboprophylaxis), and high risk (with at least 1 high-risk factor, need for thromboprophylaxis) [11]. All scores were calculated independently by 2 researchers. If any difference in the RCOG score was present, a third researcher recalculated the score.
ETHICAL CONSIDERATIONS:
This study was approved by the Institutional Review Board of IPMCHH (GKLW-2020-03).
STATISTICAL ANALYSIS:
The Kolmogorov-Smirnov test was used to evaluate the normal distribution. Continuous variables were assessed using the
A 2-sided
Results
VTE EVENTS AND THE USE OF ANTICOAGULANT:
A total of 65 501 women gave birth in our hospital during the study period, and 39 thrombotic events occurred. The incidence of DVT was 0.060% and the incidence of PE was 0.018%. Among the VTE cases, 1 patient was excluded because delivery occurred at less than 28 gestational weeks. Therefore, 38 VTE cases (VTE group) and 152 women without VTE (control group) were finally included in this study (Figure 1).
Two women in the control group used low-molecular-weight heparin (LMWH) during pregnancy, one due to significant elevation in D-dimer and the other due to a history of 3 spontaneous abortions. In the VTE group, 1 woman with antiphospholipid syndrome (APS) used unfractionated heparin prenatally. Three women with antenatal DVT used LMWH during pregnancy, and the thrombus was dissolved before delivery. Among the 190 women, only the 3 with antenatal DVT used LMWH for thromboprophylaxis after delivery. Details of the VTE events are shown in Supplementary Table 2.
Table 1 shows the comparison of socio-demographic characteristics of the women in the 2 groups. There were no significant differences in BMI, educational level, and occupation between the 2 groups. Significant differences in age (P=0.006) and birthplace (P=0.024) were observed.
UNIVARIABLE ANALYSIS OF RISK FACTORS IN THE RCOG RAM:
Risk factors contained in the RCOG RAM were compared between the 2 groups (Table 2). We found that the proportions of women in the VTE group with previous VTE (P=0.001), a family history of VTE (P=0.010), known low-risk thrombophilia (P=0.014), age >35 (P=0.007), smoking habit (P=0.046), multiple pregnancy (P=0.025), cesarean section in labor (P<0.001), and surgical procedure in pregnancy or puerperium (P=0.004) were higher than those in the control group. Other risk factors showed no significant differences between the 2 groups.
Among the 190 women, the most common risk factors were elective cesarean section (88/190), age >35 years (39/190), cesarean section in labor (38/190), and family history of VTE (18/190).
RISK SCORING AND RISK STRATIFICATION BASED ON THE RCOG RAM:
Table 3 shows that the RCOG scores of women in the VTE group were much higher than that of the control group (3.74±0.74 vs 1.49±0.22, P<0.001). A difference in risk stratification was also found between the 2 groups (P<0.001). The univariable logistic regression analysis revealed that the risk of postpartum VTE was 9.75-fold greater in the intermediate-risk group, and 90.00-fold greater in the high-risk group than in the low-risk group. Meanwhile, as the RCOG score increased, the risk of developing postpartum VTE increased almost linearly (Figure 2).
The ROC curve analysis determined that the AUC of the RCOG RAM was 0.828 (95% CI: 0.762–0.894, Figure 3A), and the Youden index was 0.50, with the best cut-off value being 2, which exactly matches the cut-off value recommended by the RCOG guidelines for pharmacological thromboprophylaxis after delivery. The sensitivity of the model was 89.47%, and its specificity was 60.53%. The positive predictive value was 36.17%, and the negative predictive value was 95.83%. In addition, the calibration plot showed good agreement between the prediction of the RCOG RAM and actual observation of VTE events (Hosmer-Lemeshow test p=0.214, see Figure 3B). The DCA also showed a certain net benefit value when the threshold probability is between 0.05–0.74 and 0.87–0.99, indicating that clinical decisions based on the predicted result of this model can benefit patients (Figure 3C).
UNIVARIABLE ANALYSIS OF OTHER CLINICAL DACTORS AND D-DIMER:
Other factors that might be associated with postpartum VTE were also analyzed. We found that endometriosis (P=0.024), a family history of diabetes (P=0.010), recurrent pregnancy loss (P=0.024), glucocorticoid therapy during pregnancy (P=0.004), previous use of oral contraceptive pills (P=0.005), and intrauterine devices (P=0.001) for contraception were more common in women with postpartum VTE, while the previous use of condoms, calendar rhythm method, or withdrawal method for contraception (P=0.009) was more common in the control group (Table 4). Women in the VTE group had significantly higher D-dimer levels before delivery, and on the first and third day after delivery (P<0.05, Table 4).
MULTIVARIABLE ANALYSIS:
RCOG risk stratification and factors (not included in the RCOG model) found to be significantly different in the univariable analysis were further entered into a multivariable logistic regression analysis. The results show that RCOG intermediate risk (adjusted OR=8.39, 95% CI: 2.64–26.60), RCOG high risk (adjusted OR=118.23, 95% CI: 18.05–774.46), previous use of IUD (adjusted OR=7.11, 95% CI: 1.45–34.93), and glucocorticoid therapy during pregnancy (adjusted OR=6.72, 95% CI: 1.56–28.91) were associated with elevated risk of postpartum VTE (Table 5).
Discussion
The RCOG risk assessment model is one of the most complex scales for pregnancy-related VTE. Since its first publication in 2004, it has been demonstrated to be effective in reducing the maternal mortality rate from PE in the UK [11], but its applicability in Chinese maternity care is uncertain. The traditional conception is that Asian people have a relatively low risk of VTE compared with people in Western countries [14]. As the data used to establish the RCOG model were obtained from Western countries, it is worrying that the use of RCOG RAM in China might lead to the overestimation of VTE risk as well as the overuse of LMWH.
Several domestic studies have compared the RCOG RAM and other VTE scoring models not specifically designed for pregnant women. Liang et al found that the discrimination and accuracy of the Wells score were better than that of the RCOG RAM, but they used the 2009 version of the RCOG model [15]. At the same time, Zhang et al came up with an opposite result [16]. Moreover, a prospective observational study has been carried out in Beijing to evaluate the feasibility of the RCOG RAM [17]. In our case-control study, we found that the RCOG RAM could screen out the majority of patients who eventually developed postpartum VTE (34/38), and for those assessed as low-risk by the model, nearly 96% would not develop VTE (90/94). Therefore, this model has a clear predictive value for postpartum VTE in Chinese women.
During its application in other Asian countries, it was found that the RCOG RAM might lead to the overuse of anticoagulant drugs in women who are less likely to develop postpartum VTE. A retrospective study in Malaysia suggested that 30.62% of postpartum women without VTE met the criteria for thromboprophylaxis [18]. Our study reached a similar conclusion.
Currently, the main concerns for the large-scale use of LMWH are its high price and the increased risk of hemorrhage and poor wound healing. However, compared with other anticoagulants such as heparin, LMWH is much safer [19]. Some studies have also confirmed that the use of LMWH does not increase the risk of postpartum hemorrhage and wound complications [20,21]. In view of the fatal consequences of VTE, it is critical to ensure that as many women as possible with potential risks of VTE receive anticoagulation treatment.
In our study, elective cesarean section, age >35 years, cesarean section in labor, and family history of VTE were the most common risk factors for postpartum VTE. Therefore, to reduce the incidence of VTE events, we strongly recommended that efforts should be made to prevent cesarean sections without indication and to avoid pregnancy in advanced age. We also found that in addition to the factors already contained in the RCOG RAM, previous use of IUD and glucocorticoid therapy during pregnancy might also increase the risk of postpartum VTE.
IUDs containing levonorgestrel (Mirena) are thought to increase the risk of VTE, but a copper IUD seems to be a safe alternative for contraception, even for people with a history of DVT or PE [22,23]. However, we surprisingly found that previous use of a copper IUD might increase the risk of postpartum VTE. Among the 7 women who had used IUD in the VTE group, only 1 used Mirena, and the rest used copper IUDs. Previous studies have not detected changes in coagulation-related parameters in IUD users, but the placement of an IUD has been shown to cause abnormal uterine bleeding by elevating fibrinolysis [24], which might cause further potential abnormalities in coagulation function that lead to thrombosis during pregnancy and the peripartum period. Furthermore, extensive microthrombosis in stromal capillaries has been observed in the endometrium in contact with an IUD [25].
The main purpose of using glucocorticoids during pregnancy is to promote fetal lung maturity in women who are at risk of preterm birth, or to treat thrombocytopenia during pregnancy. The use of glucocorticoids can cause the increased circulation level of fibrinogen, Factor VII, Factor VIII, and other hemostatic components [26]. Previous epidemiological studies also found that the use of systemic glucocorticoids can increase the risk of VTE by up to 3 times [27]. However, in the currently widely used risk assessment models for VTE, whether designed for pregnant women or not, the use of glucocorticoids is not regarded as a risk factor [10]. Therefore, it is recommended that clinicians strengthen the prevention and treatment of VTE in women who use glucocorticoids.
To our knowledge, this study is one of the few to assess the applicability of the RCOG RAM in China, and contains a relatively large number of VTE cases. At the same time, our study also has some limitations. Due to the low prevalence of postpartum VTE and the single-center design, although we enrolled all VTE cases within 4 years, the number of VTE events was relatively small. Additionally, the retrospective study design may limit the interpretation of the results and make it difficult to establish causal conclusions. To solve these problems, a prospective, large-sample, multicenter study will be required in the future. As far as we know, a large prospective observational study on the applicability of the RCOG RAM in Chinese women has already been carried out in Beijing, and the researchers hope to revise the model based on the study results to make it more suitable for Chinese population [17]. We hope that this study can provide a reference for the use of the RCOG RAM in Chinese maternity care, and provide valuable experience for subsequent large-scale studies.
Conclusions
Our results indicate that the RCOG RAM is effective in identifying women who were at risk of postpartum VTE, and that following the thromboprophylaxis methods recommended by the RCOG guidelines can effectively reduce the risk of VTE. But at the same time, we found that previous use of IUD and use of glucocorticoid during pregnancy might also increase the risk of postpartum VTE. Therefore, for the better prevention of pregnancy-related VTE, a risk assessment model based on national data needs to be established in the future.
Figures
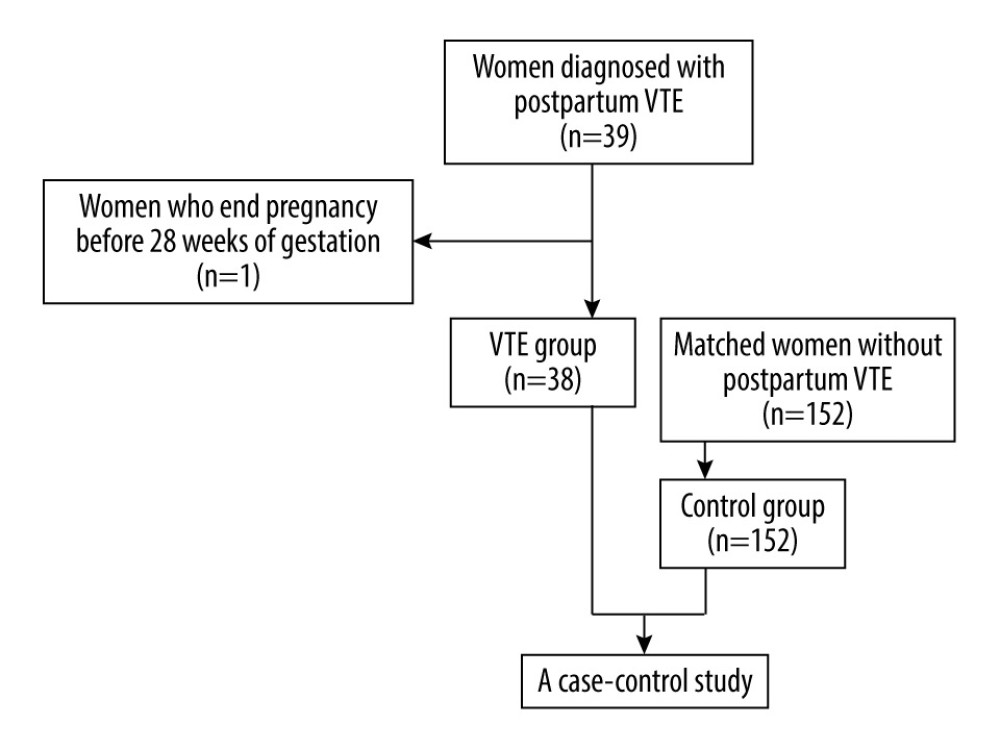 Figure 1. Flow chart.
Figure 1. Flow chart. 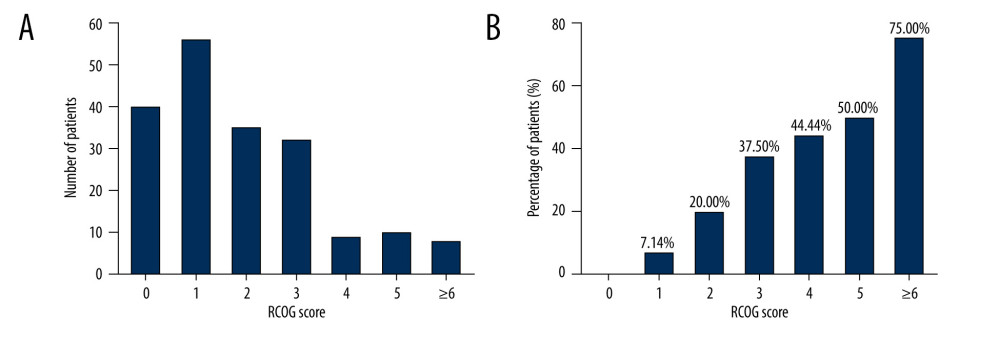 Figure 2. The distribution of the women in each score segment according to the RCOG assessment model. (A) Histogram of RCOG score (n=190). (B) Rates of VTE in each RCOG score segment.
Figure 2. The distribution of the women in each score segment according to the RCOG assessment model. (A) Histogram of RCOG score (n=190). (B) Rates of VTE in each RCOG score segment. 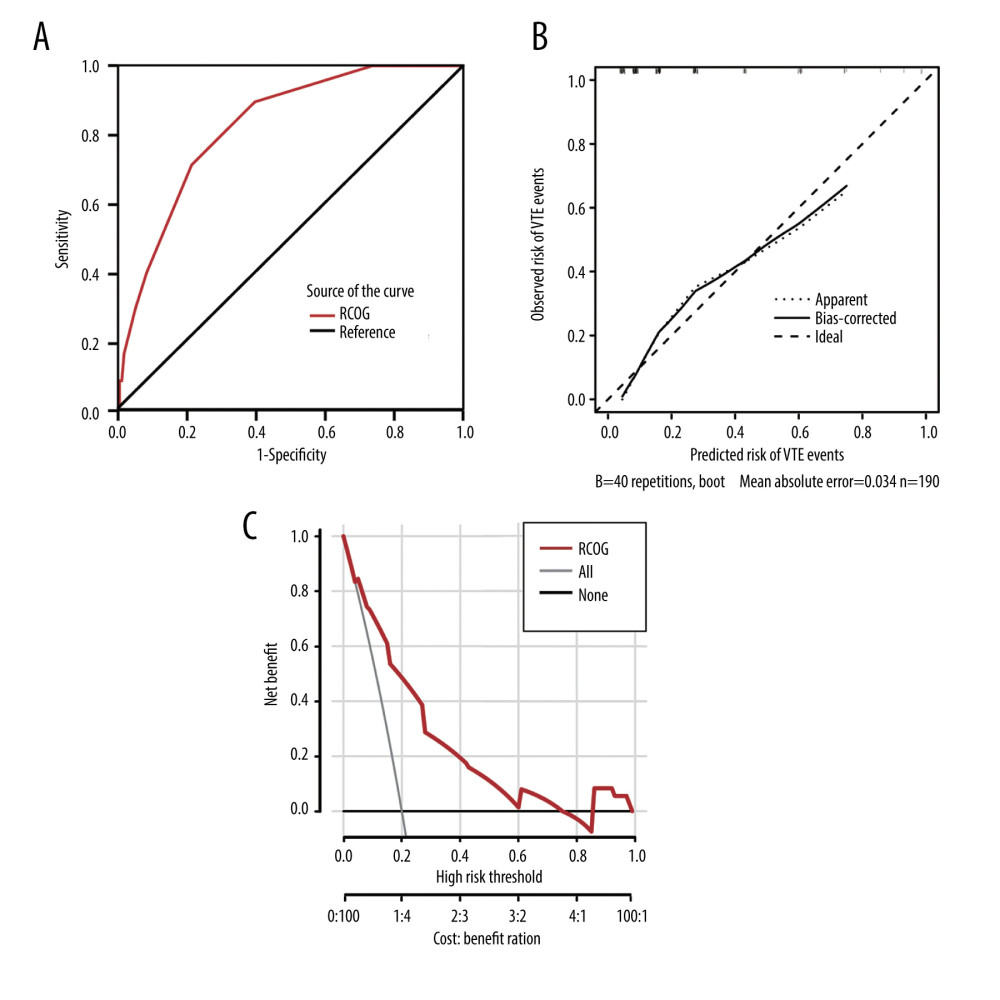 Figure 3. ROC curve, calibration plot and DCA of the RCOG risk assessment model. (A) ROC curve of the RCOG RAM. The AUC was 0.828 (95% CI: 0.762–0.894), and the Youden index was 0.50. (B) Calibration plot of the RCOG RAM. The apparent (thin dotted line) and bias-corrected (black solid line) nonparametric smoothed calibration curves are shown. (C) DCA of the RCOG RAM. The gray and black solid lines represent the net benefit of the strategy of treating all patients and no patients, respectively.
Figure 3. ROC curve, calibration plot and DCA of the RCOG risk assessment model. (A) ROC curve of the RCOG RAM. The AUC was 0.828 (95% CI: 0.762–0.894), and the Youden index was 0.50. (B) Calibration plot of the RCOG RAM. The apparent (thin dotted line) and bias-corrected (black solid line) nonparametric smoothed calibration curves are shown. (C) DCA of the RCOG RAM. The gray and black solid lines represent the net benefit of the strategy of treating all patients and no patients, respectively. Tables
Table 1. Socio-demographic characteristics of women in the VTE group and control group.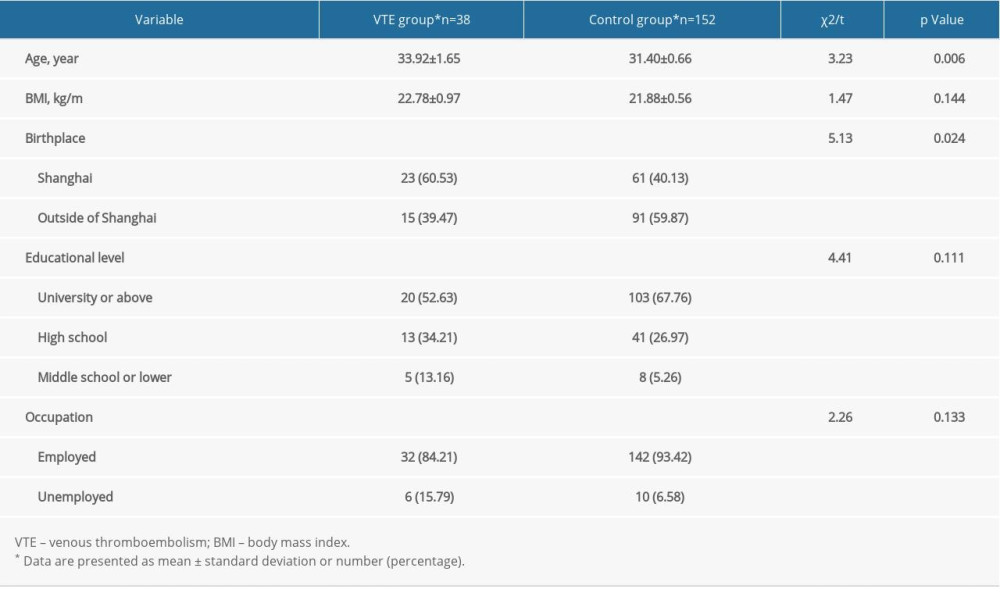 Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model.
Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model.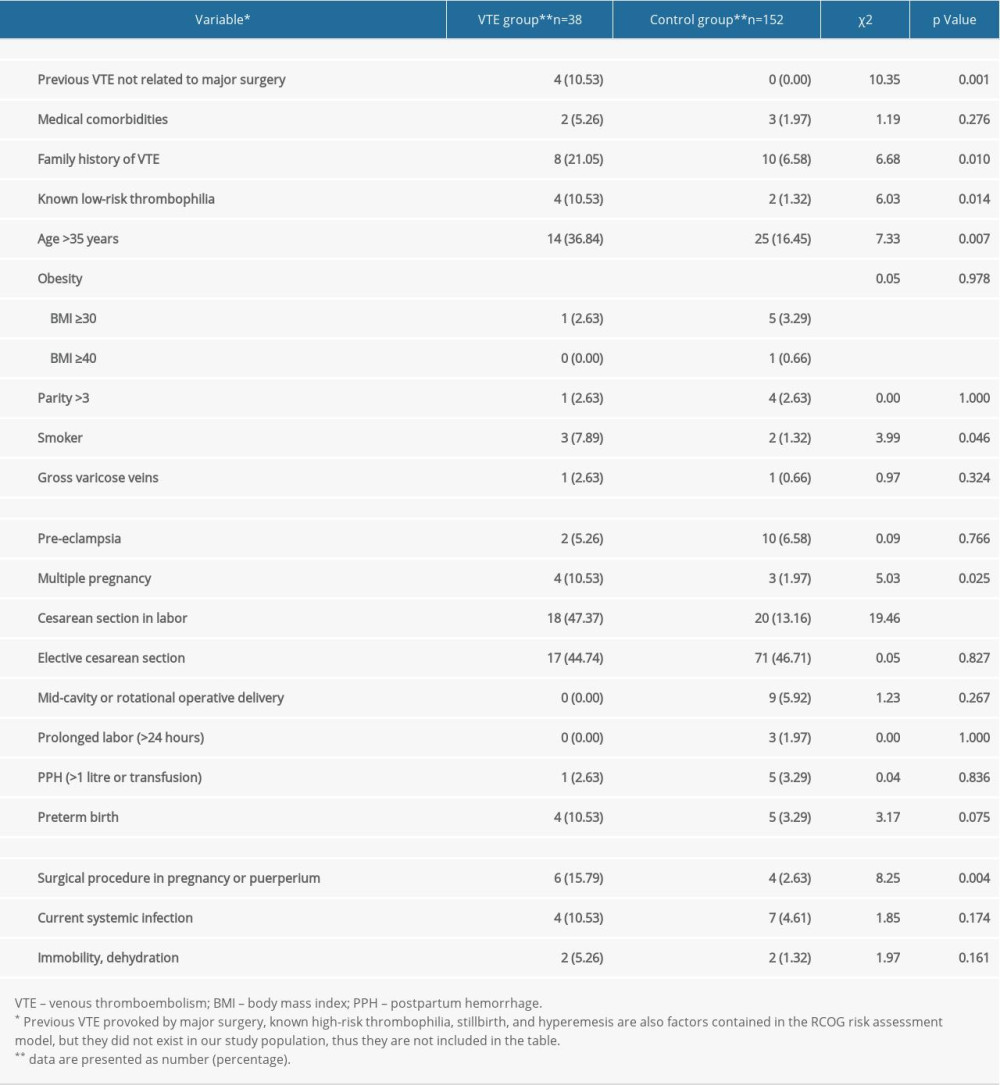 Table 3. RCOG risk score and risk level of women in the VTE group and control group.
Table 3. RCOG risk score and risk level of women in the VTE group and control group.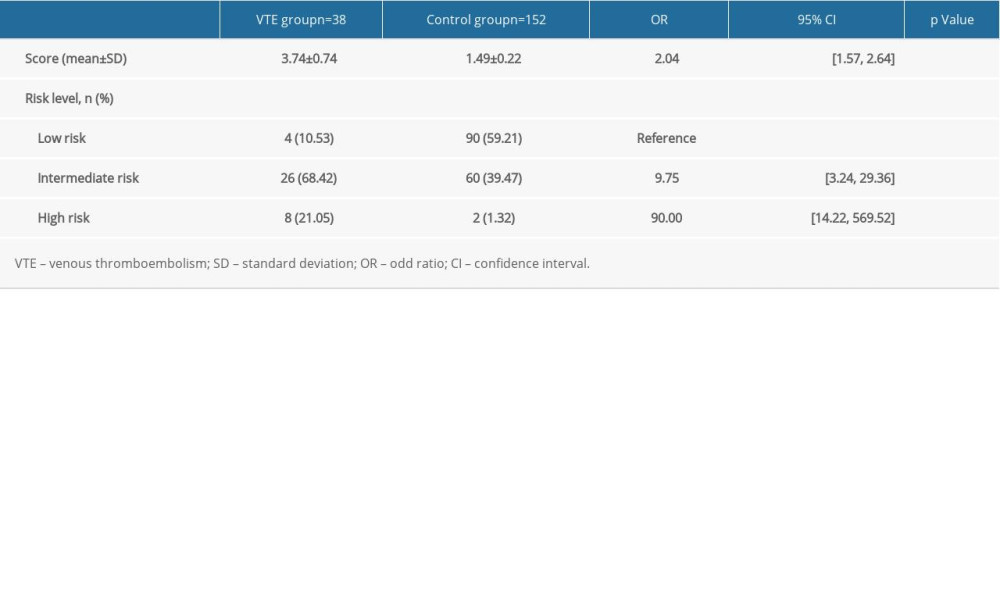 Table 4. Other clinical features and D-dimer of women in the VTE group and control group.
Table 4. Other clinical features and D-dimer of women in the VTE group and control group.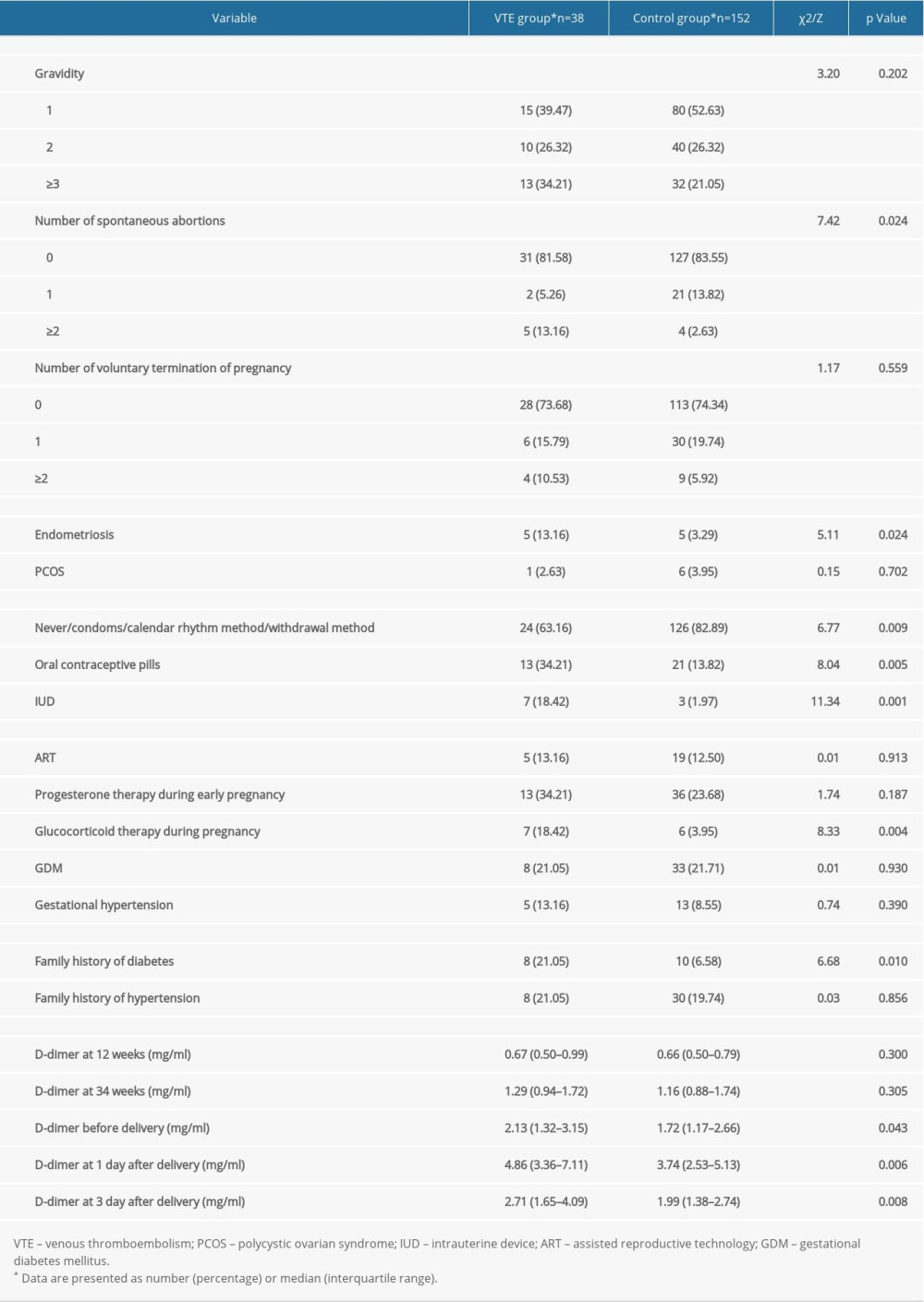 Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE.
Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE.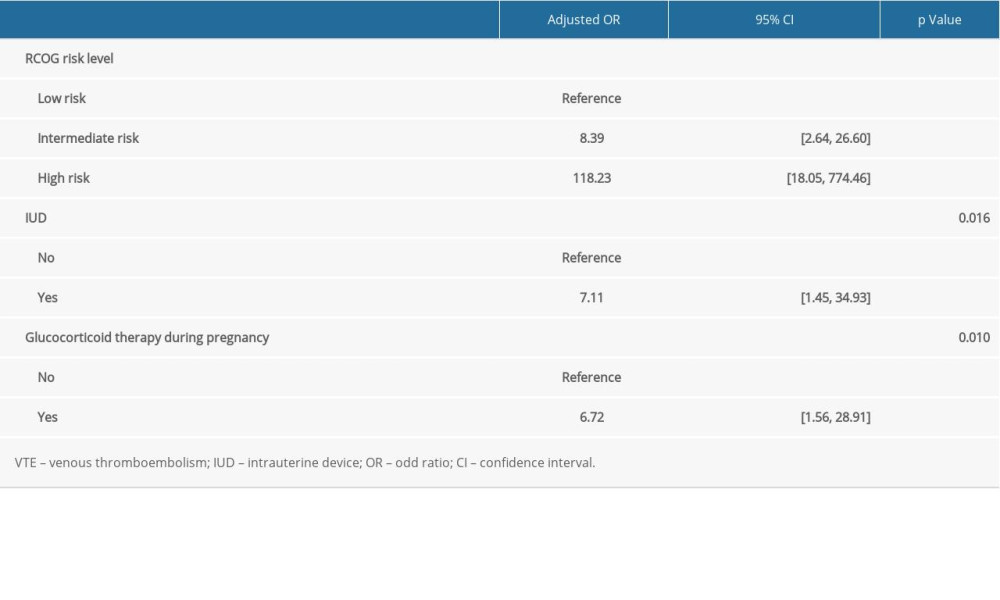 Supplementary Table 1. The RCOG risk assessment model for VTE.
Supplementary Table 1. The RCOG risk assessment model for VTE.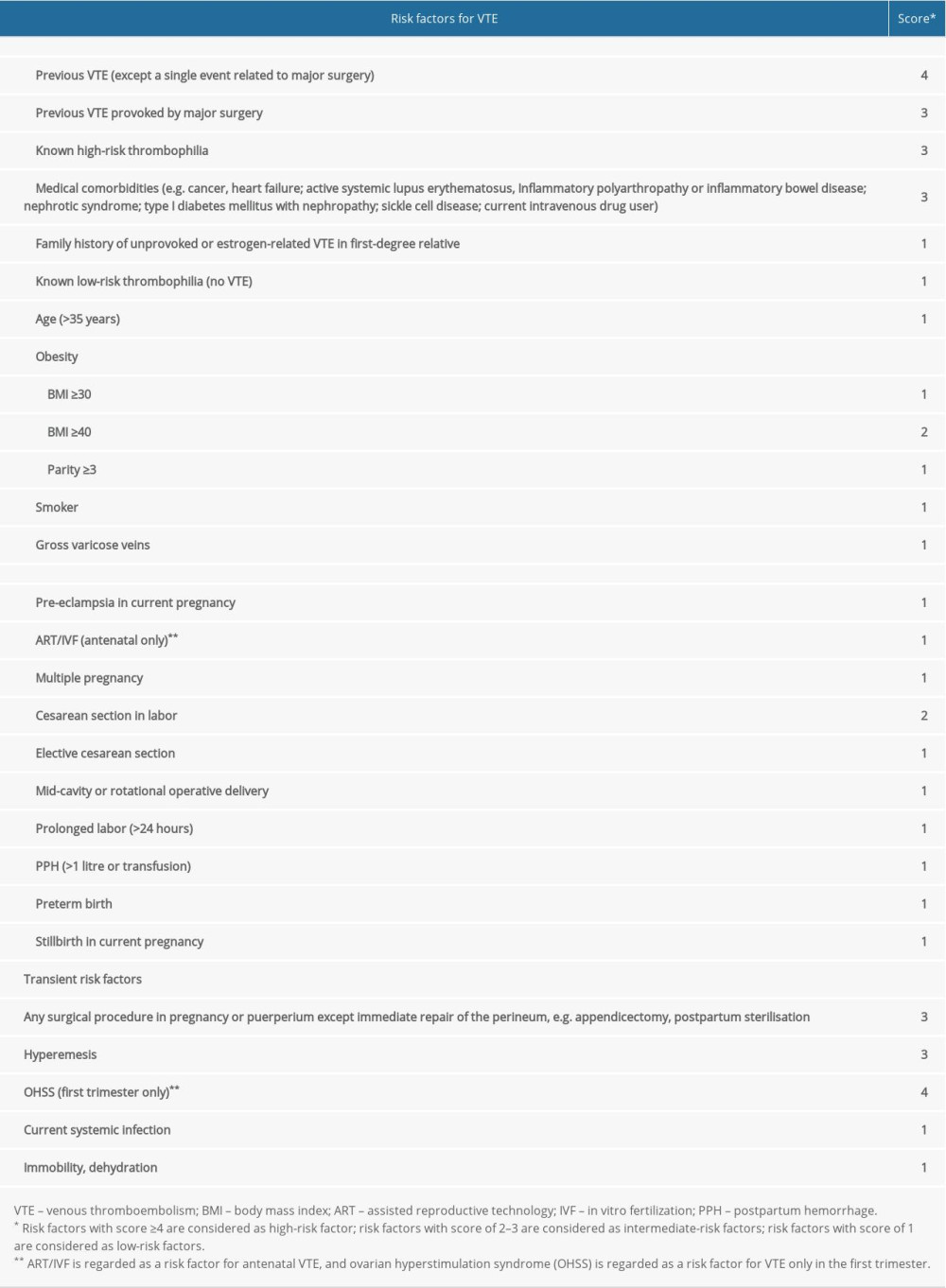 Supplementary Table 2. VTE events.
Supplementary Table 2. VTE events.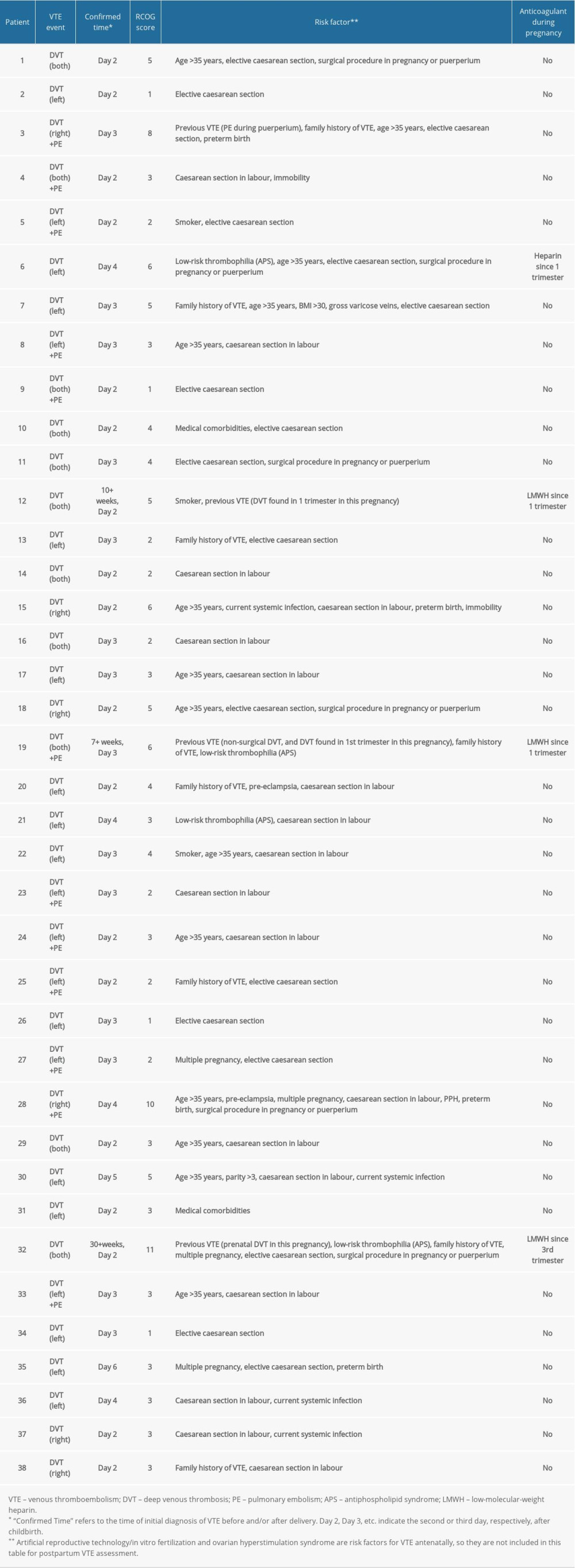
References
1. James AH, Pregnancy-associated thrombosis: Hematology Am Soc Hematol Educ Program, 2009; 277-85
2. Heit JA, Kobbervig CE, James AH, Trends in the incidence of venous thromboembolism during pregnancy or postpartum: A 30-year population-based study: Ann Intern Med, 2005; 143; 697-706
3. Pomp ER, Lenselink AM, Rosendaal FR, Doggen CJ, Pregnancy, the postpartum period and prothrombotic defects: Risk of venous thrombosis in the MEGA study: J Thromb Haemost, 2008; 6; 632-37
4. Greer IA, Thrombosis in pregnancy: Updates in diagnosis and management: Hematology Am Soc Hematol Educ Program, 2012; 2012; 203-7
5. Gherman RB, Goodwin TM, Leung B, Incidence, clinical characteristics, and timing of objectively diagnosed venous thromboembolism during pregnancy: Obstet Gynecol, 1999; 94; 730-34
6. Virkus RA, Løkkegaard EC, Lidegaard Ø, Venous thromboembolism in pregnancy and the puerperal period: A study of 1210 events: Acta Obstet Gynecol Scand, 2013; 92; 1135-42
7. James AH, Jamison MG, Brancazio LR, Myers ER, Venous thromboembolism during pregnancy and the postpartum period: incidence, risk factors, and mortality: Am J Obstet Gynecol, 2006; 194; 1311-15
8. Brennan MC, Moore LE, Pulmonary embolism and amniotic fluid embolism in pregnancy: Obstet Gynecol Clin North Am, 2013; 40; 27-35
9. Say L, Chou D, Gemmill A, Global causes of maternal death: A WHO systematic analysis: Lancet Glob Health, 2014; 2; e323-33
10. Caprini JA, Risk assessment as a guide for the prevention of the many faces of venous thromboembolism: Am J Surg, 2010; 199; S3-10
11. Royal College of Obstetricians and Gynaecologists, Reducing the Risk of Venous Thromboembolism during Pregnancy and the Puerperium (Green-top Guideline No. 37a), 2015 [EB/OL]. [2015-04-13]
12. , ACOG Practice Bulletin No. 196: Thromboembolism in pregnancy: Obstet Gynecol, 2018; 132; e1-e17
13. Yago H, Yamaki T, Sasaki Y, Application of the Caprini Risk Assessment Model for evaluating postoperative deep vein thrombosis in patients undergoing plastic and reconstructive surgery: Ann Vasc Surg, 2020; 65; 82-89
14. Hong J, Lee JH, Yhim HY, Incidence of venous thromboembolism in Korea from 2009 to 2013: PLoS One, 2018; 13; e0191897
15. Liang WZ, Huang QT, Zhong MComparison of the predictive performance of different risk assessment model in patients with deep vein thrombosis during pregnancy and postpartum: Progress in Obstetrics and Gynecology, 2014(8); 593-97 [in Chinese]
16. Zhang LX, Xie ZX, Liu CY, The forecast value of RCOG thrombosis risk assessment model in maternal venous thromboembolism: Medical Innovation of China, 2016; 13(12); 25-27 [in Chinese]
17. Chen Y, Dai Y, Song J, Establishment of a risk assessment tool for pregnancy-associated venous thromboembolism and its clinical application: Protocol for a prospective observational study in Beijing: BMC Pregnancy Childbirth, 2019; 19; 294
18. Voon HY, Chai MC, Hii LY, Postpartum thromboprophylaxis in a multireligious cohort: A retrospective review of indications and uptake: J Obstet Gynaecol, 2018; 38; 493-97
19. Robertson L, Jones LE, Fixed dose subcutaneous low molecular weight heparins versus adjusted dose unfractionated heparin for the initial treatment of venous thromboembolism: Cochrane Database Syst Rev, 2017; 2; CD001100
20. Andersen AS, Berthelsen JG, Bergholt T, Venous thromboembolism in pregnancy: Prophylaxis and treatment with low molecular weight heparin: Acta Obstet Gynecol Scand, 2010; 89; 15-21
21. Roshani S, Cohn DM, Stehouwer AC, Incidence of postpartum haemorrhage in women receiving therapeutic doses of low-molecular-weight heparin: Results of a retrospective cohort study: BMJ Open, 2011; 1; e000257
22. Jean S, Isolated pulmonary embolism in a patient with progestin intrauterine device and factor V Leiden: J Community Hosp Intern Med Perspect, 2019; 9; 264-66
23. , Intrauterine devices: An effective alternative to oral hormonal contraception: Prescrire Int, 2009; 18; 125-30
24. Khanna A, Biswas AK, Dubey B, Khanna AK, Fibrinolytic activity in bleeding associated with intrauterine contraceptive devices: Indian J Med Res, 1992; 96; 147-49
25. Sheppard BL, Bonnar J, The effects of intrauterine contraceptive devices on the ultrastructure of the endometrium in relation to bleeding complications: Am J Obstet Gynecol, 1983; 146; 829-39
26. Brotman DJ, Girod JP, Posch A, Effects of short-term glucocorticoids on hemostatic factors in healthy volunteers: Thromb Res, 2006; 118; 247-52
27. Johannesdottir SA, Horváth-Puhó E, Dekkers OM, Use of glucocorticoids and risk of venous thromboembolism: A nationwide population-based case-control study: JAMA Intern Med, 2013; 173; 743-52
Figures
 Figure 1. Flow chart.
Figure 1. Flow chart. Figure 2. The distribution of the women in each score segment according to the RCOG assessment model. (A) Histogram of RCOG score (n=190). (B) Rates of VTE in each RCOG score segment.
Figure 2. The distribution of the women in each score segment according to the RCOG assessment model. (A) Histogram of RCOG score (n=190). (B) Rates of VTE in each RCOG score segment. Figure 3. ROC curve, calibration plot and DCA of the RCOG risk assessment model. (A) ROC curve of the RCOG RAM. The AUC was 0.828 (95% CI: 0.762–0.894), and the Youden index was 0.50. (B) Calibration plot of the RCOG RAM. The apparent (thin dotted line) and bias-corrected (black solid line) nonparametric smoothed calibration curves are shown. (C) DCA of the RCOG RAM. The gray and black solid lines represent the net benefit of the strategy of treating all patients and no patients, respectively.
Figure 3. ROC curve, calibration plot and DCA of the RCOG risk assessment model. (A) ROC curve of the RCOG RAM. The AUC was 0.828 (95% CI: 0.762–0.894), and the Youden index was 0.50. (B) Calibration plot of the RCOG RAM. The apparent (thin dotted line) and bias-corrected (black solid line) nonparametric smoothed calibration curves are shown. (C) DCA of the RCOG RAM. The gray and black solid lines represent the net benefit of the strategy of treating all patients and no patients, respectively. Tables
 Table 1. Socio-demographic characteristics of women in the VTE group and control group.
Table 1. Socio-demographic characteristics of women in the VTE group and control group. Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model.
Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model. Table 3. RCOG risk score and risk level of women in the VTE group and control group.
Table 3. RCOG risk score and risk level of women in the VTE group and control group. Table 4. Other clinical features and D-dimer of women in the VTE group and control group.
Table 4. Other clinical features and D-dimer of women in the VTE group and control group. Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE.
Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE. Table 1. Socio-demographic characteristics of women in the VTE group and control group.
Table 1. Socio-demographic characteristics of women in the VTE group and control group. Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model.
Table 2. Risk factors for postpartum VTE contained in the RCOG risk assessment model. Table 3. RCOG risk score and risk level of women in the VTE group and control group.
Table 3. RCOG risk score and risk level of women in the VTE group and control group. Table 4. Other clinical features and D-dimer of women in the VTE group and control group.
Table 4. Other clinical features and D-dimer of women in the VTE group and control group. Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE.
Table 5. Multivariable logistic regression analysis of risk factors for postpartum VTE. Supplementary Table 1. The RCOG risk assessment model for VTE.
Supplementary Table 1. The RCOG risk assessment model for VTE. Supplementary Table 2. VTE events.
Supplementary Table 2. VTE events. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








