07 February 2021: Review Articles
A Review of the Current Practice of Diagnosis and Treatment of Idiopathic Membranous Nephropathy in China
Lianzhong Wu12AE, Jin Lai12B, Yixin Ling12C, Yiqin Weng12B, Shujuan Zhou1D, Siqi Wu2D, Songfu Jiang1D, Xiaokai Ding3D, Xin Jin4EFG, Kang Yu1EF, Yi Chen1EF*DOI: 10.12659/MSM.930097
Med Sci Monit 2021; 27:e930097
Abstract
ABSTRACT: Idiopathic membranous nephropathy (IMN), a common pathological type of nephrotic syndrome, is one of the main causes of kidney failure. With an increasing prevalence, IMN has received considerable attention in China. Based on recent studies, we discuss advances in the diagnosis of IMN and the understanding of its genetic background. Although the pathogenesis of IMN remains unclear, our understanding has been substantially enhanced by the discovery of new antigens such as phospholipase A2 receptor, thrombospondin type-1 domain-containing 7A, exostosin1/exostosin2, neural epidermal growth factor-like 1 protein, neural cell adhesion molecule 1, semaphorin 3B, and factor H autoantibody. However, due to ethnic, environmental, economic, and lifestyle differences and other factors, a consensus has not yet been reached regarding IMN treatment. In view of the differences between Eastern and Western populations, in-depth clinical evaluations of biomarkers for IMN diagnosis are necessary. This review details the current treatment strategies for IMN in China, including renin-angiotensin system inhibitors, corticosteroid monotherapy, cyclophosphamide, calcineurin inhibitors, mycophenolate mofetil, adrenocorticotropic hormone, and traditional Chinese medicine, as well as biological preparations such as rituximab. In terms of management, the 2012 Kidney Disease Improving Global Outcomes (KDIGO) clinical practice guidelines do not fully consider the characteristics of the Chinese population. Therefore, this review aims to present the current status of IMN diagnosis and treatment in Chinese patients, and includes a discussion of new approaches and remaining clinical challenges.
Keywords: review, Diagnosis, Glomerulonephritis, Membranous, Time-to-Treatment, Adrenal Cortex Hormones, Autoantibodies, calcineurin inhibitors, Kidney, Mycophenolic Acid, nephrotic syndrome
Background
Idiopathic membranous nephropathy (IMN) is one of the main causes of pathological nephrotic syndrome and kidney failure. IMN is a common condition, but its pathogenesis is not entirely clear. The main clinical features of IMN are varying degrees of proteinuria. Approximately 60% of patients present with nephrotic syndrome, which may lead to secondary venous thrombosis. Although about 30–35% of IMN patients experience spontaneous remission, for the most part, the disease course is long and it is relatively difficult to cure. Approximately 30% to 40% of patients eventually end up in kidney failure within 5–15 years, requiring dialysis or kidney transplantation. Among Whites, IMN accounts for around 30–40% of primary nephrotic syndrome, with the peak age of onset being 40–50 years [1]. In China, the frequency of IMN has recently increased significantly among cases of renal biopsy. Hou et al reported that the frequency of membranous nephropathy (MN) has increased significantly, nearly doubling from 2003–2006 (10.4%) to 2011–2014 (24.1%) [2]. Xu et al reviewed changes in glomerular disease types in a Chinese population over the previous 11 years and found that IgA nephropathy remained the most common pathological type, with a frequency of 28.1%, followed by membranous nephropathy, with a frequency of 23.4% [3]. After data calibration, it was found that membranous nephropathy increased 13% annually, and its incidence had a tendency to exceed that of IgA nephropathy [3]. Xu et al analyzed the characteristics of Asian IMN patients and noted that treatment of these patients could not simply follow the guidelines proposed by Kidney Disease Improving Global Outcomes (KDIGO) because of differences in race, environment, economic factors, lifestyle habits, and other factors [4,5]. In view of the differences between Eastern and Western populations, in-depth clinical evaluations of biomarkers for the diagnosis of IMN is necessary. Recent research results have prompted KDIGO to review recent guidelines (post-2012) and to re-evaluate the diagnosis and treatment processes for IMN [5]. Therefore, this review aims to present the current status of the diagnosis and treatment of Chinese patients with IMN, and includes a discussion of new approaches and remaining clinical challenges.
Research Progress in the Pathogenesis of IMN
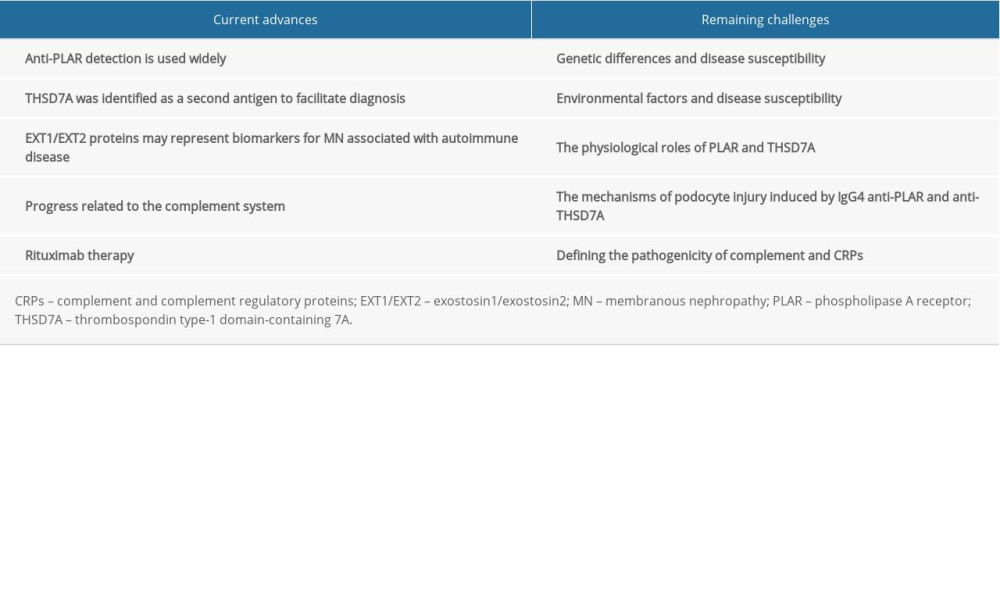
The main pathological feature of IMN is accumulation of subepithelial immune deposits, which leads to thickening of the glomerular capillary wall matrix and the formation of spikes. Immune deposits include immunoglobulin G (IgG), complement membrane attack complex, and long-term dependent known or unknown antigens. As podocytes are the main target cells in IMN, podocyte antigens are most commonly involved in immune deposits, while IgG4 is usually the most prominent subtype of IgG deposited in IMN [6]. Activation of these immune deposits and complement leads to damage of the glomerular filtration membrane and the formation of albuminuria (Table 1).
Experimental Animal Models
In the study of IMN, the Heymann nephritis model was the earliest autoimmune animal model, the target antigen of which is the podocyte membrane protein megalin, also known as low-density lipoprotein receptor-related protein 2 (LRP2) [7]. Anti-LRP2 antibodies can be detected in patients with anti-brush edge disease but not in immune deposits or the circulation in patients with IMN. Although LRP2-mediated MN is unrelated to human IMN, Heymann nephritis still contributes extensively to our understanding of MN. Researchers have also found that repeated immunization of experimental animals with cationic bovine serum albumin (C-BSA) can induce subepithelial deposition of IgG and C3 [8]. The renal perfusion test confirmed that C-BSA first binds with the anion heparin sulfate proteoglycan of the basement membrane and then forms an immune complex with an antibody, which activates complement and leads to MN [9]. Then, a new podocyte antigen, neutral endopeptidase (NEP), was detected in alloimmune neonatal nephropathy. Maternal anti-NEP antibody enters the fetus through the placenta and reacts with NEP on fetal podocytes, resulting in the formation of epithelial immune complexes, which causes MN in newborns as confirmed in an experimental rabbit model [10]. Animal models have various defects, but they all serve as useful tools for understanding IMN.
Complement Activation and IMN
The roles of complement and complement regulatory proteins (CRPs) in IMN are currently a hot topic. Complement activation is the main route by which IMN leads to renal injury. Patients with mannan binding lectin (MBL) deficiency can develop IMN, with complement activated mainly by the alternative pathway, whereas the lectin pathway is also activated in those with wild-type MBL2 promoter and coding region variations [11]. The functional evidence indicates that anti-Fx1A-induced Heymann nephritis is characterized by podocyte injury and proteinuria induced by C5b-9 [12]. C3, C4, and C5b-9 deposits were also found in human IMN. More interestingly, Leenaerts et al have shown that an active C6-deficient rat Heymann nephritis model can still develop proteinuria in the absence of C5b-9 [13]. Injection of human autoantibody against thrombospondin type-1 domain-containing 7A (THSD7A) in mice has also been reported to cause albuminuria, but no deposition of C3 was noted [14]. Therefore, further studies are needed to clarify complement-mediated damage and its mechanism before obtaining clinical data for complement inhibitors and regulators.
New Diagnostic Biomarkers
In recent years, the most important IMN research result has been the discovery of the M-type phospholipase A2 receptor (PLA2R), a specific target antigen of IMN that exists on the surface of normal podocytes [15]. This study demonstrated that anti-PLA2R antibodies could be detected in the blood of IMN patients, with a sensitivity of 78% and a specificity of more than 99%. Subsequently, IMN was further recognized as an autoimmune disease because of the discovery of THSD7A. There is no doubt that the discovery of PLA2R and THSD7A drove great progress in basic and clinical IMN research. It was proposed that the concept of IMN should be updated to autoimmune membranous nephropathy or PLA2R antibody-related membranous nephropathy, and its treatment should also consider the targeting of PLA2R antibodies [5]. The application of PLA2R and THSD7A as biomarkers for diagnosis and prognosis has been widely accepted worldwide, including in China, and their detection is also being performed in China’s third-tier cities. The current problem is how to standardize the detection of PLA2R antibodies and avoid test errors as much as possible. However, the value of renal biopsy in IMN cannot be denied because renal pathology provides necessary information for evaluating the severity of acute kidney injury (AKI) and chronic renal fibrosis. The 2019 KDIGO Guidelines suggest that for patients with positive anti-PLA2R antibodies, low-risk patients do not need to have renal biopsies, while high-risk patients do need to have renal biopsies, to carry out risk stratification assessment [5].
Despite this progress, scientists are continuing to look for other, effective ways to identify IMN. Sanjeev et al reported that exostosin1/exostosin2 might be the target antigen for secondary membranous nephropathy (SMN), especially when it is comorbid with autoimmune diseases such as lupus nephritis [16]. Thus, the detection of exostosin1/exostosin2 may be a useful supplement for the differential diagnosis of IMN and SMN, but its clinical practicability remains to be identified. However, so far, circulating autoantibodies to exostosin1/exostosin2 have not been validated. Caza et al identified neural cell adhesion molecule 1 (NCAM1) as a membranous antigen by mass spectrometry analysis [17]. Patients with NCAM1-positive IMN were followed to identify whether they developed lupus, which is important in determining whether NCAM1-positive or anti-NCAM1 antibodies can be a precursor to SLE [17]. In recent years, increasing attention has been paid to the role of the complement system and its regulatory factors in the pathogenesis of IMN. Neural epidermal growth factor-like 1 protein (NELL-1) defines a distinct third type of auto-antigen on IMN podocytes [18]. Sethi et al demonstrated that the predominant subclass of IgG deposition of NELL-1-associated MN is IgG1, whereas the subclass of IgG of PLA2R- and THSD7A-associated MN utilizes IgG4 as the principal antibody [18]. Recently, Sethi et al used mass spectrometry to identify moderate spectral counts of a unique protein, semaphorin 3B, in 3 cases of IMN, and revealed that semaphorin 3B-associated MN represents a unique type of primary MN [19]. Claudia et al pointed out that complement factor H might play a crucial role in PLA2R-related IMN, since the deterioration of proteinuria in clinical cases of IMN was closely correlated with the titer of factor H autoantibodies [20]. Therefore, the researchers suggested that factor H autoantibodies are used to assess the risk of progressive IMN in patients. Paolo et al suggested that, based on the determination of anti-PLA2R or anti-THSD7A autoantibodies, new techniques should be explored to evaluate specific B-cell immune responses and perform immune risk classification to formulate individualized treatment strategies [21]. Notably, the clinical value of these newly discovered potential biomarkers, namely, PLA2R and THSD7A autoantibodies, has not been consistently confirmed, especially in the Chinese population. Therefore, it is too early to establish an effective clinical evaluation index. For now, the PLA2R antibody is still the most valuable biomarker, and it is advocated for the detection of both circulating and tissue-bound PLA2R. In SMN, the positive rate of anti-PLA2R antibody is relatively low, but in some other diseases, hepatitis B virus and active sarcoidosis [22] for instance, their correlation with MN should be considered carefully. Thus, it is necessary to analyze each case comprehensively in addition to performing clinical analysis.
Genetic Background
Current studies suggest that PLA2R antibody-associated MN is likely caused by genetic susceptibility, loss of tolerance, changes in antigen expression, and other factors, which are closely related to environmental factors and the human leukocyte antigen (HLA) gene [23]. However, the HLA alleles involved may differ in diverse ethnic groups. There is evidence that HLA-DRB1*15: 01 and HLA-DRB3*02: 02 are independent and closely bound alleles of PLA2R-related MN in the Chinese population [24]. Cui et al revealed that DRB1*15: 01 and DRB1*03: 01 are independent risk alleles in patients with IMN, which is associated with circulating anti-PLA2R antibodies [25]. Recently, 2 other studies used candidate gene methods; their results showed that HLA-DRB1*15: 01 is an HLA risk allele in Chinese and Japanese populations [26,27]. A genome-wide association study (GWAS) of MN indicated that the HLA-DRB1*03: 01-DQA1*05: 01 haplotype associated with HLA-DRB3*02: 02 was higher in the European white population, and there was no evidence of HLA-DRB1*15: 01 in this population [23]. It can be concluded that there may be a difference in HLA alleles between MN patients in Asia and Europe. Approximately 10% of PLA2R-negative MN patients in Europe and North America possess THSD7A target antigens. Jia et al screened 578 Chinese patients with IMN and found that 12 cases (2%) were positive for THSD7A, suggesting that the incidence of THSD7A-associated MN in Chinese patients is lower than that in European and American populations [28]. Accordingly, race and genes are responsible for different incidences, clinical manifestations, and drug responses between Eastern and Western IMN patients. Therefore, as Japan has done [29], the formulation of diagnosis and treatment guidelines should be adjusted such that they are suitable for each country’s own national conditions.
Treatment of IMN
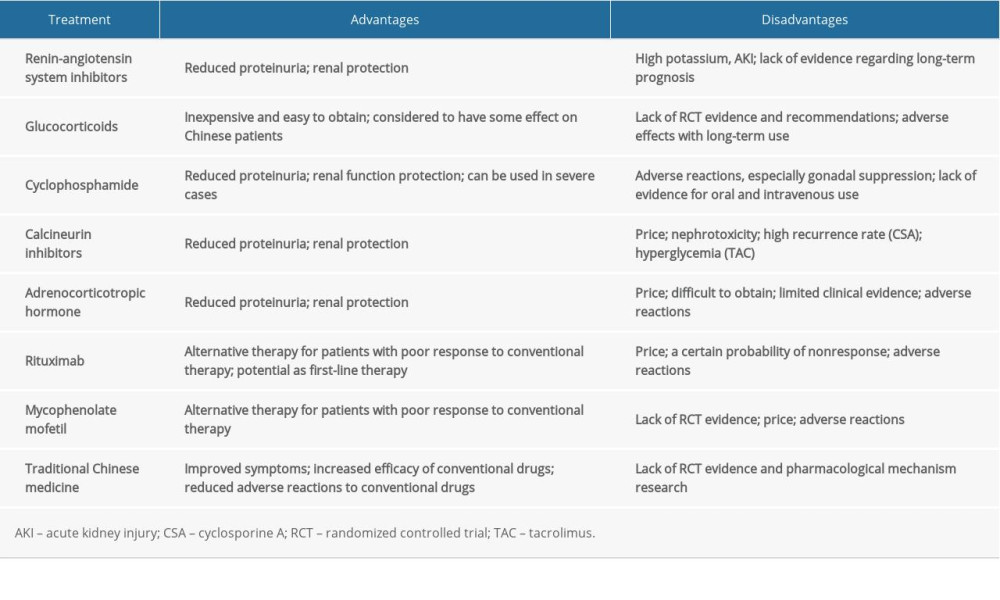
At present, treatment recommendations following the KDIGO guidelines for IMN mainly derive from evidence-based medicine for Whites. However, due to ethnic, environmental, lifestyle, and economic differences and other factors as mentioned above, the guidelines should not be strictly applied to Chinese people. For example, the KDIGO guidelines do not recommend hormone monotherapy for IMN, but a number of observational studies from Asia suggest that Asian IMN patients respond better to hormone monotherapy than Western patients [29–31]. In addition, the timing of the use of immunosuppressants is controversial. The principles of treatment based on risk assessment proposed at a KDIGO meeting indicate that for patients with mild symptoms and good renal function, suspending the use of immunosuppressive therapy within 3 years after precise control of albuminuria, hypertension, hyperlipidemia, and other common symptoms, is acceptable [5]. As a result, risks still exist for patients lacking proper immunosuppressive therapy. In China, traditional Chinese medicine has always been applied in clinical practice for IMN, a practice that requires additional, objective evaluation. In summary, there has been no consensus on the treatment of IMN in Asia, and China specifically, so far (Table 2), and additional evidence-based data are required to build a treatment system suitable for the Chinese population.
Renin-Angiotensin System Inhibitors
Renin-angiotensin system (RAS) inhibitors, including angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs), can reduce proteinuria and protect the kidney by inhibiting the RAS system, reducing glomerular filtration pressure and restoring glomerular barrier dysfunction. These compounds are also clearly defined as the first-line drug treatment approach for general IMN according to the KDIGO guidelines. Although most IMN patients from Asia have massive albuminuria [32], there are few randomized controlled trials (RCTs) on the efficacy of ACEIs/ARBs in Asia. Previous studies have shown that RAS inhibitors can reduce proteinuria by 40% to 50% and delay the development of renal failure [33]. Thus, the maximum tolerable dose of ACEIs/ARBs is recommended as treatment. In fact, the use of RAS inhibitors stemmed from experience in the treatment of other glomerular diseases. According to current evidence-based medicine and treatment experience, RAS inhibitors are still essential drugs for the treatment of IMN; however, we need to pay attention to individual dose adjustments to avoid complications such as hyperkalemia, AKI, and hypotension. The strategy of dual RAS inhibition has some supporting evidence in Chinese patients with IgA nephropathy, and the curative effect in IMN is also worth exploring. Serum anti-PLA2R antibody levels are positively correlated with the severity of IMN and decreased renal function, and negatively correlated with spontaneous remission rate and induced remission rate [34]. The change in the severity of IMN usually lags behind the change in the level of anti-PLA2R antibody. If anti-PLA2R antibody levels decrease progressively and renal function is stable, non-immunotherapy can be extended for 3 to 6 months, after which the necessity of immunosuppressive therapy can be re-evaluated [35].
Glucocorticoids
Glucocorticoids are widely used in the treatment of glomerular diseases because of their immunosuppressive, anti-inflammatory, and podocyte-protective effects. Since corticosteroid monotherapy has shown no significant benefit in RCTs in Western populations, KDIGO does not recommend it for the treatment of IMN. However, the situation in Asia is drastically different. A retrospective study of the Chinese population showed that initial hormone therapy was effective in young IMN patients with nephrotic syndrome [30]. A small-sample study from Hong Kong demonstrated that glucocorticoid therapy (0.5 mg/kg/day, orally) used for 9 months (median 7 months) induced remission in 71% of IMN patients [31]. Japanese guidelines on the treatment of nephrotic syndrome also suggested using glucocorticoid monotherapy to prevent a decline in renal function, although it was not clear whether glucocorticoid monotherapy can effectively reduce proteinuria [29]. In some areas of China, sufficient glucocorticoids are still sometimes used as the initial treatment in IMN patients. Due to the lack of effective evaluation methods and the characteristic of spontaneous remission of IMN, it is difficult to form reliable conclusions. Although some clinical studies have indicated that glucocorticoid monotherapy works well in Asian patients with IMN, and Japanese guidelines also affirm the value of glucocorticoids, there is no prospective RCT involving Asian patients with a high risk of progression, so it is difficult to evaluate the actual efficacy. Since the current KDIGO guidelines do not recommend hormone monotherapy for IMN, additional convincing research data are indispensable before hormone monotherapy becomes a recognized strategy.
Cyclophosphamide
Cyclophosphamide (CTX) is the most commonly used cytotoxic drug in Asia, Europe, and the United States, and is the only drug that has been shown to prevent kidney failure and death [5]. The modified Ponticelli regimen – CTX combined with methylprednisolone (MP) for 6 months every other month – is a treatment scheme recommended by KDIGO for the treatment of IMN. This therapy has been shown to alleviate nephrotic syndrome and delay the deterioration of renal function [5]. Compared with oral administration, the intravenous CTX regimen is safe and effective. To be clear, intravenous CTX also has serious adverse effects; however, the intravenous regimen shows fewer adverse effects than the oral regimen, because the accumulated dose is lower. There is no significant difference in the remission of membranous nephrotic syndrome between oral administration and intravenous CTX. So far, there has been no direct comparison research between intravenous CTX and oral CTX. At present, many doctors in China administer monthly intravenous CTX. Meta-analyses involving Asian populations compared intravenous CTX and cyclosporine A (CSA) and showed that monthly intravenous CTX and oral corticosteroids, for 6 months, improved the remission rate and delayed the progression of renal insufficiency [36,37]. Therefore, the evidence indirectly shows that intravenous CTX is useful and monthly intravenous CTX plus an oral hormone may be an alternative treatment for Chinese patients with IMN. In China, there are some limitations to the use of CTX owing to fertility issues. Some studies have adopted an improved CTX dosage regimen to treat Chinese patients with IMN, and this improved regimen has reduced adverse reactions [38]. In Japan, the guidelines recommend that clinical use should be considered with caution because patients often have adverse reactions after using alkylating agents, and their effectiveness is relatively low [29]. Thus, it is clear that due to ethnic differences, social factors, and other reasons, the drug treatment dose for the Asian population may be different from that for European and American populations to some extent. To explore the appropriate dose for the Chinese population and to improve the scientific scheme, large-scale clinical studies are needed.
Calcineurin Inhibitors
Calcineurin inhibitors (CNIs) can not only directly stabilize the podocyte actin cytoskeleton but can also reduce the level of PLA2R, thereby reducing proteinuria [39]. In one CNI study, only the reduction in albuminuria was used as evidence of the curative effect, and the high recurrence rate was the main defect. For now, CNIs mainly include tacrolimus (TAC) and CSA. In North America, the prescription of CSA is quite common and deemed a first-line treatment [40]. In recent years, the use of CNIs in China has increased greatly [4]. Based on a favorable response to hormones in the Chinese population, medium-dose hormones (0.3–0.5 mg/kg/day) combined with CNI therapy is often adopted by Chinese clinicians, even though this dose choice is not supported by evidence-based medicine. In China, CNIs are often used for cases in which hormone monotherapy is not effective, or in patients with severe osteoporosis or fertility requirements or intolerance to cytotoxic drugs. Thus, it is a promising substitute for CTX.
CSA is a cyclic polypeptide, composed of 11 amino acids, that selectively inhibits helper T cells and cytotoxic T cells. In some small-sample RCTs, it was confirmed that long-term low-dose CSA combined with medium-dose prednisone is safe and effective in most Chinese patients with IMN nephropathy (including those who are unable to tolerate other immunosuppressive regimens) and has better efficacy with fewer adverse effects than traditional CTX regimens [41–44]. Lin et al performed a meta-analysis to evaluate the efficacy and safety of CSA for the treatment of IMN of Asian patients [37]. This study found that the efficacy of CSA plus glucocorticoid in the Asian population was better than that of CTX plus glucocorticoid and similar to that of TAC, with mild adverse reactions but a high recurrence rate after short-term treatment [37]. CSA is a more widely used CNI than TAC in China because of its price advantage and its characteristic of not raising blood sugar.
As a CNI, TAC also inhibits T cells, but has the disadvantage of neurotoxicity. A meta-analysis of European and American populations showed that TAC had more advantages in terms of short-term efficacy and safety than CTX, and no significant difference was found in the long-term remission rate [45]. However, it has also been reported that the recurrence rate after TAC withdrawal is much higher than that after withdrawal from oral CTX [46]. Furthermore, the function of TAC in reducing the development of kidney failure in the long term has not been directly confirmed by RCTs. Most comparative studies of TAC and CSA demonstrate that they have equivalent efficacy [47,48]. In existing clinical studies involving Chinese populations, TAC combined with corticosteroids induced IMN remission effectively and quickly [49,50]. Further, it also worked effectively for IMN with refractory nephropathy syndrome [51]. Compared with European and American populations, for Chinese populations, even low-dose TAC combined with prednisone was sufficient to induce remission in most IMN patients [52–54]. An RCT study also showed that TAC monotherapy was an effective and safe treatment for Chinese IMN patients [55]. However, the long-term efficacy of this treatment strategy requires additional long-term followup studies.
Adrenocorticotropic Hormone
Adrenocorticotropic hormone (ACTH), a trace polypeptide hormone secreted by the adenohypophysis, is the main regulator of adrenocortical activity. Berg et al first reported the efficacy of synthetic ACTH in 14 patients with IMN-related nephrotic syndrome and found that it not only reduced blood lipids but also reduced urinary albumin excretion by 90% [56]. In a subsequent RCT, 32 patients with IMN received synthetic ACTH for 12 months or standard periodic treatment for 6 months. The results indicated that the remission rates of the 2 groups were similar (the rates in the ACTH and alkylating agent groups were 87% and 75%, respectively). However, ACTH treatment caused only mild adverse effects [57]. The positive results of the above trials have led to new interest in ACTH as a potential drug for the treatment of IMN, and additional small-scale studies have recently been reported [58–60]. However, a prospective open-cohort study evaluated the safety and efficacy of synthetic ACTH for the treatment of IMN patients at risk of renal failure [61], and these results showed that synthetic ACTH was not as effective as CTX in inducing remission in high-risk patients with IMN. Moreover, it had many adverse events. Therefore, the study concluded that synthetic ACTH is not recommended as a standard treatment for IMN. The clinical use of ACTH remains relatively low.
Rituximab Therapy
In recent years, with the successive discoveries of the anti-PLA2R and THSD7A antibodies, it has been suggested that IMN is associated with the deposition of immune complexes on the epithelial side of the glomerular basement membrane caused by humoral immunity mediated by B lymphocytes [62]. Rituximab (RTX) is a chimeric mouse/human monoclonal antibody that induces B-cell apoptosis and can be used to treat MN by eliminating B cells. According to the KDIGO guidelines, RTX should be considered for patients with severe nephrotic syndrome or for whom traditional immunosuppressive therapy is ineffective. Research from Peking University indicated that for IMN patients with high risk of progression of renal insufficiency, who were unresponsive to other immunosuppressive therapy, RTX could relieve albuminuria and stabilize renal function [63]. The cumulative incidence of partial remission with RTX was lower than that with CTX, but there were no significant differences in the probability of complete remission or the incidence of endpoint events [62]. Recent reports by Fervenza et al showed that RTX was not inferior to cyclosporine in inducing remission of MN [64]. After cyclosporine treatment, albuminuria was significantly relieved, but the recurrence rate was high, while the anti-albuminuria effect of RTX lasted longer than that of cyclosporine [65]. Therefore, at present, the call for RTX to become a first-line treatment for IMN is strong [65,66]. Recently, published membranous nephropathy trial of rituximab (MENTOR) studies have shown that rituximab may replace cyclosporine as a first-line immunosuppressive drug [64]. Some studies also evaluated the combination of RTX and other immunosuppressants that had achieved solid results [67]. Although some encouraging results have been obtained in the study of RTX, the nonresponse rate of RTX treatment is approximately 35%. Evidence suggests that its long-term safety and efficacy is superior to that of other immunosuppressants for the treatment of IMN [35,68]. However, this evidence is not yet convincing enough and needs to be further evaluated in clinical studies with large samples and long-term followup.
Mycophenolate Mofetil
Mycophenolate mofetil (MMF) is often used for the treatment of glomerular diseases, as it inhibits T and B lymphocytes by inhibiting the activity of hypoxanthine nucleotide dehydrogenase in the ab initio synthesis pathway. However, for the initial treatment of IMN, the KDIGO guidelines do not recommend MMF because of the low power of the evidence indicating that the effect of MMF plus corticosteroids is similar to that of CTX plus corticosteroids [65]. There are also studies on the treatment of IMN patients with severe albuminuria with MMF combined with low-dose corticosteroids; the results of these studies show efficacy similar to that of CSA [69]. MMF monotherapy has almost no effect on the treatment of IMN [70]. The data from an RCT in Hong Kong showed that MMF combined with hormones was effective in the treatment of IMN patients with nephrotic syndrome [71]. Compared with other commonly used cytotoxic drugs, MMF has the potential benefit of a reduced hormone dosage and reduced corresponding adverse reactions. Yet, due to the high price of MMF, some adverse reactions, the difficulty of concentration detection, and other factors, Chinese clinicians do not regard it as a first-choice treatment. Nevertheless, for patients with poor responses to conventional CTX and CNIs and patients who cannot tolerate RTX, MMF can be considered as an alternative choice.
Traditional Chinese Medicine
In China, traditional Chinese medicine is widely used for the treatment of IMN. Tripterygium wilfordii multiglycosides (TWGs) and Shenqi are the most widely used traditional Chinese medicines. A prospective cohort study of the Chinese population showed that the remission rate of the TWG plus hormone group was comparable to that of the TAC plus hormone group [72]. A Chinese multicenter RCT showed that the improvement in albuminuria in the 2 groups was similar to that of the traditional Ponticelli strategy, while Shenqi had the advantage of restoring the estimated glomerular filtration rates (eGFR) in patients with IMN. More importantly, Shenqi treatment did not cause serious adverse reactions [73]. Han et al studied the efficacy of Shenjiu prescription (SR) and Qingre Moshen granules (QMG) in the treatment of IMN, and found that SR plus QMG was effective in patients with IMN who failed to respond to a variety of Western medicines [74]. This strategy improves the level of cellular immunity and modifies the abnormal expression of IL-2. However, the main obstacle in the use of traditional Chinese medicine is obtaining medical evidence through high-level basic and clinical research to obtain international acceptance.
New Approaches and Remaining Clinical Challenges
Overall, IMN is not only a common pathological type of primary nephrotic syndrome, but also one of the main causes of kidney failure in China, and its treatment is still a major challenge in clinical practice. In view of the differences between Eastern and Western populations, in-depth clinical evaluations of biomarkers for the diagnosis of IMN is necessary. Further study is warranted in PLA2R and THSD7A antibodies, exostosin1/exostosin2, neural epidermal growth factor-like 1 protein, neural cell adhesion molecule 1, semaphorin 3B, factor H autoantibody, and the response of the Chinese population to these treatments. In terms of treatment, the current KDIGO guidelines, especially those regarding hormone and immunosuppression treatment timing, program selection, and other controversial aspects, do not fully take into account the characteristics of the Asian population. Therefore, a prominent problem that needs to be solved urgently is how to optimize the diagnosis and treatment of Chinese patients with IMN and improve their prognosis. The alternative use of CTX combined with glucocorticoids (CTX plus MP), which was once advocated, is not widely used in China, and the usage of CTX is not uniform in China. Moreover, due to outstanding adverse reactions caused by CTX, its use is subject to many restrictions. The use of CNIs has gradually increased in China over recent years. Although CNIs help improve the overall remission rate in IMN patients, the recurrence rate is high, and long-term use leads to nephrotoxicity, so its use should be considered on a case-by-case basis. In recent years, some new drugs, such as RTX and ACTH, have been tested worldwide. However, the sample sizes in the preclinical trials of these drugs were small, with short average followup times. In addition, the long-term kidney survival rate has not been evaluated. Therefore, continuous observation is still required. Small-sample observation of RTX in clinical practice has yielded results indicating that RTX could be a promising alternative for refractory IMN. RTX-based or other B-cell targeted regimens represent the future direction of IMN treatment. The traditional Chinese medicine TWG has shown a degree of efficacy in the clinic, but clarifying its mechanism and carrying out effective RCTs is still a problem to be considered.
Conclusions
IMN is a disease with different diagnostic and therapeutic challenges in China compared with other countries. The recommendations from the 2012 KDIGO clinical practice guidelines may not apply to the Chinese population, due to ethnic, environmental, economic, and lifestyle differences. Although Chinese clinical guidelines are awaited, treatment strategies for patients with IMN in China include renin-angiotensin inhibitors, corticosteroid monotherapy, cyclophosphamide, calcineurin inhibitors, mycophenolate mofetil, adrenocorticotropic hormone, biologics such as rituximab, and also some traditional Chinese medicines.
References
1. Keri KC, Blumenthal S, Kulkarni V, Comprehensive review and historical perspective: Postgrad Med J, 2019; 95; 23-31
2. Hou JH, Zhu HX, Zhou ML, An analysis of 40,759 biopsy-proven cases from 2003 to 2014 in China: Kidney Dis (Basel), 2018; 4; 10-19
3. Xu X, Wang G, Chen N, Long-term exposure to air pollution and increased risk of membranous nephropathy in China: J Am Soc Nephrol, 2016; 27; 3739-46
4. Xu J, Hu X, Xie J, Management of membranous nephropathy in Asia: Kidney Dis (Basel), 2015; 1; 119-25
5. Floege J, Barbour SJ, Cattran DC, Management and treatment of glomerular diseases (part 1): Conclusions from a Kidney Disease: Improving Global Outcomes (KDIGO) Controversies Conference: Kidney Int, 2019; 95; 268-80
6. Segawa Y, Hisano S, Matsushita M, IgG subclasses and complement pathway in segmental and global membranous nephropathy: Pediatr Nephrol, 2010; 25; 1091-9
7. Kerjaschki D, Farquhar MG, The pathogenic antigen of Heymann nephritis is a membrane glycoprotein of the renal proximal tubule brush border: Proc Natl Acad Sci USA, 1982; 79; 5557-61
8. Kobayashi M, Muro K, Yoh K, Effects of FK506 on experimental membranous glomerulonephritis induced by cationized bovine serum albumin in rats: Nephrol Dial Transplant, 1998; 13; 2501-8
9. Koyama A, Inage H, Kobayashi M, Effect of chemical cationization of antigen on glomerular localization of immune complexes in active models of serum sickness nephritis in rabbits: Immunology, 1986; 58; 529-34
10. Debiec H, Guigonis V, Mougenot B, Antenatal membranous glomerulonephritis due to anti-neutral endopeptidase antibodies: N Engl J Med, 2002; 346; 2053-60
11. Bally S, Debiec H, Ponard D, Phospholipase A2 receptor-related membranous nephropathy and mannan-binding lectin deficiency: J Am Soc Nephrol, 2016; 27; 3539-44
12. Cybulsky AV, Rennke HG, Feintzeig ID, Complement-induced glomerular epithelial cell injury. Role of the membrane attack complex in rat membranous nephropathy: J Clin Invest, 1986; 77; 1096-107
13. Leenaerts PL, Hall BM, Van Damme BJ, Active Heymann nephritis in complement component C6 deficient rats: Kidney Int, 1995; 47; 1604-14
14. Tomas NM, Hoxha E, Reinicke AT, Autoantibodies against thrombospondin type 1 domain-containing 7A induce membranous nephropathy: J Clin Invest, 2016; 126; 2519-32
15. Garcia-Vives E, Sole C, Moline T, Antibodies to M-type phospholipase A2 receptor (PLA2R) in membranous lupus nephritis: Lupus, 2019; 28; 396-405
16. Sethi S, Madden BJ, Debiec H, Exostosin 1/exostosin 2-associated membranous nephropathy: J Am Soc Nephrol, 2019; 30; 1123-36
17. Caza T, Hassen S, Kuperman M, Neural cell adhesion molecule 1 is a novel autoantigen in membranous lupus nephritis: Kidney Int, 2020 [Online ahead of print]
18. Sethi S, Debiec H, Madden B, Neural epidermal growth factor-like 1 protein (NELL-1) associated membranous nephropathy: Kidney Int, 2020; 97; 163-74
19. Sethi S, Debiec H, Madden B, Semaphorin 3B-associated membranous nephropathy is a distinct type of disease predominantly present in pediatric patients: Kidney Int, 2020; 98; 1253-64
20. Seikrit C, Ronco P, Debiec H, Factor H autoantibodies and membranous nephropathy: N Engl J Med, 2018; 379; 2479-81
21. Cravedi P, Jarque M, Angeletti A, Immune-monitoring disease activity in primary membranous nephropathy: Front Med (Lausanne), 2019; 6; 241
22. Xie Q, Li Y, Xue J, Renal phospholipase A2 receptor in hepatitis B virus-associated membranous nephropathy: Am J Nephrol, 2015; 41; 345-53
23. Sekula P, Li Y, Stanescu HC, Extension of and association with other chronic kidney disease aetiologies: Nephrol Dial Transplant, 2017; 32; 325-32
24. Le WB, Shi JS, Zhang T, HLA-DRB1*15: 01 and HLA-DRB3*02: 02 in PLA2R-related membranous nephropathy: J Am Soc Nephrol, 2017; 28; 1642-50
25. Cui Z, Xie LJ, Chen FJ, MHC class II risk alleles and amino acid residues in idiopathic membranous nephropathy: J Am Soc Nephrol, 2017; 28; 1651-64
26. Xie J, Liu L, Mladkova N, The genetic architecture of membranous nephropathy and its potential to improve non-invasive diagnosis: Nat Commun, 2020; 11; 1600
27. Lv J, Hou W, Zhou X, Interaction between PLA2R1 and HLA-DQA1 variants associates with anti-PLA2R antibodies and membranous nephropathy: J Am Soc Nephrol, 2013; 24; 1323-29
28. Wang J, Cui Z, Lu J, Circulating antibodies against thrombospondin type-I domain-containing 7A in Chinese patients with idiopathic membranous nephropathy: Clin J Am Soc Nephrol, 2017; 12; 1642-51
29. Nishi S, Ubara Y, Utsunomiya Y, Evidence-based clinical practice guidelines for nephrotic syndrome 2014: Clin Exp Nephrol, 2016; 20; 342-70
30. Wang Y, Wang GP, Li BM, Clinicopathological analysis of idiopathic membranous nephropathy in young adults: Genet Mol Res, 2015; 14; 4541-48
31. Tang S, Chan TM, Cheng IK, Clinical features and treatment outcome of idiopathic membranous nephropathy in Chinese patients: QJM, 1999; 92; 401-6
32. Yamaguchi M, Ando M, Yamamoto R, Patient age and the prognosis of idiopathic membranous nephropathy: PLoS One, 2014; 9; e110376
33. Wolf G, Ritz E, Combination therapy with ACE inhibitors and angiotensin II receptor blockers to halt progression of chronic renal disease: Pathophysiology and indications: Kidney Int, 2005; 67; 799-812
34. Hoxha E, Harendza S, Pinnschmidt H, PLA2R antibody levels and clinical outcome in patients with membranous nephropathy and non-nephrotic range proteinuria under treatment with inhibitors of the renin-angiotensin system: PLoS One, 2014; 9; e110681
35. Ruggenenti P, Debiec H, Ruggiero B, Anti-phospholipase A2 receptor antibody titer predicts post-rituximab outcome of membranous nephropathy: J Am Soc Nephrol, 2015; 26; 2545-58
36. Yuan J, Fang W, Zhang W, Treatment of nephrotic idiopathic membranous nephropathy with monthly i.v. pulse cyclophosphamide and oral steroids: A single centre’s retrospective study: Nephrology (Carlton), 2011; 16; 440-45
37. Lin S, Li HY, Zhou T, Efficacy and safety of cyclosporine A in the treatment of idiopathic membranous nephropathy in an Asian population: Drug Des Devel Ther, 2019; 13; 2305-30
38. Ren S, Wang Y, Xian L, Comparative effectiveness and tolerance of immunosuppressive treatments for idiopathic membranous nephropathy: A network meta-analysis: PLoS One, 2017; 12; e0184398
39. Bech AP, Hofstra JM, Brenchley PE, Association of anti-PLA(2)R antibodies with outcomes after immunosuppressive therapy in idiopathic membranous nephropathy: Clin J Am Soc Nephrol, 2014; 9; 1386-92
40. Alfaadhel T, Cattran D, Management of membranous nephropathy in western countries: Kidney Dis (Basel), 2015; 1; 126-37
41. Li MX, Yu YW, Zhang ZY, Administration of low-dose cyclosporine alone for the treatment of elderly patients with membranous nephropathy: Genet Mol Res, 2015; 14; 2665-73
42. Chen Y, Schieppati A, Chen X, Immunosuppressive treatment for idiopathic membranous nephropathy in adults with nephrotic syndrome: Cochrane Database Syst Rev, 2014; 18; CD004293
43. Yu X, Ruan L, Qu Z, Effectiveness and renal safety: Ren Fail, 2017; 39; 688-97
44. Li J, Zhang YM, Qu Z, An uncontrolled study with prospective follow-up: Am J Med Sci, 2010; 339; 532-36
45. Qiu TT, Zhang C, Zhao HW, A systematic review and meta-analysis of 21 clinical trials: Autoimmun Rev, 2017; 16; 136-45
46. Ramachandran R, Yadav AK, Kumar V, Two-year follow-up study of membranous nephropathy treated with tacrolimus and corticosteroids versus cyclical corticosteroids and cyclophosphamide: Kidney Int Rep, 2017; 2; 610-16
47. Peng L, Wei SY, Li LT, Comparison of different therapies in high-risk patients with idiopathic membranous nephropathy: J Formos Med Assoc, 2016; 115; 11-18
48. Cattran DC, Brenchley PE, Membranous nephropathy: Integrating basic science into improved clinical management: Kidney Int, 2017; 91; 566-74
49. Chen M, Li H, Li XY, A multicenter randomized controlled trial: Am J Med Sci, 2010; 339; 233-38
50. Xu J, Zhang W, Xu Y, A randomized, prospective, controlled trial: Contrib Nephrol, 2013; 181; 152-62
51. Chen W, Liu Q, Liao Y, A prospective, multicenter clinical trial: Am J Med Sci, 2013; 345; 81-87
52. He L, Peng Y, Liu H, Treatment of idiopathic membranous nephropathy with combination of low-dose tacrolimus and corticosteroids: J Nephrol, 2013; 26; 564-71
53. Zhang XJ, Ji CF, Yuan JZ, Efficacy and safety of tacrolimus-based treatment for nephrotic idiopathic membranous nephropathy in young adults: A retrospective study: Kaohsiung J Med Sci, 2019; 35; 633-39
54. Yuan H, Liu N, Sun GD, Effect of prolonged tacrolimus treatment in idiopathic membranous nephropathy with nephrotic syndrome: Pharmacology, 2013; 91; 259-66
55. Liang Q, Li H, Xie X, The efficacy and safety of tacrolimus monotherapy in adult-onset nephrotic syndrome caused by idiopathic membranous nephropathy: Ren Fail, 2017; 39; 512-18
56. Berg AL, Nilsson-Ehle P, Arnadottir M, Beneficial effects of ACTH on the serum lipoprotein profile and glomerular function in patients with membranous nephropathy: Kidney Int, 1999; 56; 1534-43
57. Ponticelli C, Passerini P, Salvadori M, A randomized pilot trial comparing methylprednisolone plus a cytotoxic agent versus synthetic adrenocorticotropic hormone in idiopathic membranous nephropathy: Am J Kidney Dis, 2006; 47; 233-40
58. Bomback AS, Tumlin JA, Baranski J, Treatment of nephrotic syndrome with adrenocorticotropic hormone (ACTH) gel: Drug Des Devel Ther, 2011; 5; 147-53
59. Bomback AS, Canetta PA, Beck LH, A prospective trial: Am J Nephrol, 2012; 36; 58-67
60. Hladunewich MA, Cattran D, Beck LH, A pilot study to determine the dose and effectiveness of adrenocorticotrophic hormone (H.P. Acthar(R) Gel) in nephrotic syndrome due to idiopathic membranous nephropathy: Nephrol Dial Transplant, 2014; 29; 1570-77
61. van de Logt AE, Beerenhout CH, Brink HS, Synthetic ACTH in high risk patients with idiopathic membranous nephropathy: A prospective, open label cohort study: PLoS One, 2015; 10; e0142033
62. van de Logt AE, Dahan K, Rousseau A, Immunological remission in PLA2R-antibody-associated membranous nephropathy: Cyclophosphamide versus rituximab: Kidney Int, 2018; 93; 1016-17
63. Wang X, Cui Z, Zhang YM, Rituximab for non-responsive idiopathic membranous nephropathy in a Chinese cohort: Nephrol Dial Transplant, 2018; 33; 1558-63
64. Fervenza FC, Appel GB, Barbour SJ, Rituximab or cyclosporine in the treatment of membranous nephropathy: N Engl J Med, 2019; 381; 36-46
65. Rojas-Rivera JE, Carriazo S, Ortiz A, Treatment of idiopathic membranous nephropathy in adults: KDIGO 2012, cyclophosphamide and cyclosporine A are out, rituximab is the new normal: Clin Kidney J, 2019; 12; 629-38
66. Ruggenenti P, Fervenza FC, Remuzzi G, Treatment of membranous nephropathy: Time for a paradigm shift: Nat Rev Nephrol, 2017; 13; 563-79
67. Rojas-Rivera J, Fernandez-Juarez G, Ortiz A, A European multicentre and open-label controlled randomized trial to evaluate the efficacy of Sequential treatment with TAcrolimus-Rituximab versus steroids plus cyclophosphamide in patients with primary MEmbranous Nephropathy: The STARMEN study: Clin Kidney J, 2015; 8; 503-10
68. Dahan K, Debiec H, Plaisier E, A 6-month trial with extended follow-up: J Am Soc Nephrol, 2017; 28; 348-58
69. Choi JY, Kim DK, Kim YW, A multicenter randomized trial: J Korean Med Sci, 2018; 33; e74
70. Dussol B, Morange S, Burtey S, A 1-year randomized controlled trial: Am J Kidney Dis, 2008; 52; 699-705
71. Chan TM, Lin AW, Tang SC, Prospective controlled study on mycophenolate mofetil and prednisolone in the treatment of membranous nephropathy with nephrotic syndrome: Nephrology (Carlton), 2007; 12; 576-81
72. Liu S, Li X, Li H, Comparison of tripterygium wilfordii multiglycosides and tacrolimus in the treatment of idiopathic membranous nephropathy. A prospective cohort study: BMC Nephrol, 2015; 16; 200
73. Chen Y, Deng Y, Ni Z, Efficacy and safety of traditional chinese medicine (Shenqi particle) for patients with idiopathic membranous nephropathy: A multicenter randomized controlled clinical trial: Am J Kidney Dis, 2013; 62; 1068-76
74. Han L, Deng YY, Chen YPClinical observation of Shen No. 9 Recipe with Qingre Moshen Granule on the treatment of idiopathic membranous nephropathy after treated by multiple Western drugs with no efficacy: Zhongguo Zhong Xi Yi Jie He Za Zhi, 2011; 31; 1496-501 [in Chinese]
In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952