17 March 2021: Clinical Research
Different Doses of Oxycodone for Endoscopic Injection Sclerotherapy of Esophageal Varices
Jie Yao1AEF, ChunXiao Song2BF, Chen Zhou3C, HaiLi He4AEG*, ZheFeng Quan4BDDOI: 10.12659/MSM.929111
Med Sci Monit 2021; 27:e929111
Abstract
BACKGROUND: The aim of the present study was to evaluate the effects of different doses of oxycodone during endoscopic injection sclerotherapy (EIS) for esophageal varices with painless sclerosing agents.
MATERIAL AND METHODS: A total of 119 patients were randomly divided into 3 groups: Group A, midazolam and 0.075 mg/kg oxycodone (n=40); Group B, midazolam and 0.1 mg/kg oxycodone (n=40); and Group C, midazolam and 0.125 mg/kg oxycodone (n=39). The main observation index was the incidence of body movement during the perioperative period. The secondary indices were additional propofol usage; postoperative analgesic usage; other adverse effects, such as hypoxia, myoclonus, and cough; and satisfaction scores for surgeons and patients.
RESULTS: The incidence rates for body movement during the perioperative period in groups A, B, and C were 33%, 13%, and 0, respectively (P<0.001). The satisfaction scores for surgeons and patients were highest in Group C (0.125 mg/kg oxycodone). The incidence rates for hypoxia before EIS were 15%, 8%, and 33% (P=0.026) and during EIS were 23%, 3%, and 0% (P<0.001), respectively. There were no significant between-group differences with respect to other adverse effects.
CONCLUSIONS: The ideal dose of oxycodone for perioperative analgesia during EIS for esophageal varices is 0.125 mg/kg.
Keywords: Endoscopy, Digestive System, Oxycodone, Personal Satisfaction, Dose-Response Relationship, Drug, Esophageal and Gastric Varices, Gastrointestinal Hemorrhage, Incidence, Injections, Liver Cirrhosis, Midazolam, perioperative period, Prospective Studies, Sclerosing Solutions, Sclerotherapy
Background
Esophageal varices are common in patients with cirrhosis. In early-stage disease, endoscopic injection sclerotherapy (EIS) typically is used to treat esophageal varices. Because patients with it often have impaired liver function, however, normal doses of sedatives and opioids can cause serious adverse effects [1–3]. Nevertheless, during EIS, good immobilization of patients is required, which necessitates relatively deep analgesia and sedation [4]. As a dual opioid receptor agonist, oxycodone has complete affinity for the μ-opioid receptor and a lower affinity for the κ-opioid receptor.
In one of our previous studies, oxycodone was shown to have a better analgesic effect and lower incidence of complications such as hypoxia than sufentanil during EIS in patients with esophageal varices; this indicated that oxycodone administered in combination with midazolam was a better analgesic method for EIS [5]. Other studies also have demonstrated that oxycodone can effectively attenuate visceral pain and is a sufficient analgesic for the esophagus [6,7]. However, the ideal dose of oxycodone for EIS analgesia is still unclear. In our previous study, we used 0.1 mg/kg of oxycodone for perioperative analgesia. It is not clear whether lower or higher doses of oxycodone confer more advantages. In the present study, we sought to evaluate the effects of different doses of oxycodone (0.075, 0.1, and 0.125 mg/kg) for perioperative analgesia during EIS for esophageal varices with painless sclerosing agents.
Material and Methods
SAMPLE SIZE CALCULATION:
Sample size was calculated with the Cochran-Armitage test for trend using STATA/MP software, version 16.0 (Stata Corporation, College Station, Texas, United States). In the preliminary experiment, the incidence rates for extremity movement in groups A, B, and C were 30% (12/40), 15% (6/40) and 5% (2/40), respectively. The sample size was calculated as 36 for each group with α=0.05 and power (1−β)=0.8. Considering the dropout rate (approximately 10%), the sample size was determined to be 40 patients per group.
STUDY POPULATION AND DESIGN:
For the present study, 120 patients with liver disease who underwent elective EIS between December 2016 and September 2019 at the Beijing You’an Hospital of the Capital Medical University were recruited. Inclusion criteria were age between 18 and 65 years, body mass index between 18 and 25 kg/m2, and American Society of Anesthesiologists grades I to III. Exclusion criteria were epilepsy, adrenocortical insufficiency, psychiatric disorder, current exposure to medication affecting the central nervous system, allergy to oxycodone, receipt of general anesthesia within the preceding 7 days, and being pregnant. Before EIS, an anesthetist who did not participate in the study used a computer-generated table to randomly allocate the enrolled patients to 3 groups: 0.075 mg/kg, 0.1 mg/kg, and 0.125 mg/kg of oxycodone.
Patients underwent routine fasting and received no preoperative medication. Ten to 15 minutes before the endoscopy, 10 mL of lidocaine hydrochloride jelly (Kangye Pharmaceutical Co., Ltd., Handan, China) was applied. Heart rate, blood pressure, and SpO2 were continuously monitored. Peripheral venous access was established. All patients were given oxygen with a conventional oxygen mask (flow rate: 4 L/min). Oxycodone (Batch number BM201; Napp, UK) was diluted to a concentration of 0.5 mg/mL in a 20-mL syringe and distributed by the anesthesia nurse, who was blinded to the group identity. The anesthetist administered 0.075, 0.1, and 0.125 mg/kg oxycodone to patients in groups A, B, and C, respectively, for 1 min. After 2 min, all patients were administered 0.1 mg/kg of midazolam for 1 min. After 5 min, the Overall Assessment of Alertness/Sedation Scale (OAA/S) was used by the anesthesiologist to assess patients. A score of 5 points indicated rapid response to the normal intonation; 4 points, sluggish response to normal intonation; 3 points, response to loud or repeated calling out of the name; 2 points, response to mildly pushing the shoulder or head; and 1 point, no response to mild pushing. Patients who whose score was >1 received 10 to 30 mg of propofol, with the dose dependent on each individual’s situation. If a patient’s OAA/S score was <1, EIS was performed.
Flumazenil (0.3 mg) was administered intravenously (IV) at the end of the EIS procedure. All operations were performed by a skilled senior physician using an Olympus CV260 gastroscope (Olympus Optical Co., Ltd., Tokyo, Japan). When perioperative cough and limb movement occurred, an additional 10 to 30 mg of propofol was administered. For patients with SpO2 ≤92%, the gastroscope mask was pressurized with oxygen using the jaw thrust technique. Hypotension (defined as a >30% decrease in mean arterial pressure [MAP] from the baseline level) was treated with 5 mg of IV ephedrine. Hypertension (defined as a >30% increase in MAP from the baseline level) was treated with 5 mg of IV nimidane. Bradycardia (defined as a heart rate <50/min) was treated with 0.5 mg of IV atropine.
OBSERVATION INDICES:
The primary observation index was limb movement during the operation. The secondary observation index was the incidence of hypoxia (defined as SPO2 ≤92%), propofol bolus, remedial analgesia (intramuscular administration of meperidine hydrochloride), adverse effects (intraoperative hypertension, hypotension, postoperative myoclonus, cough, nausea, vomiting, lethargy, or dizziness), and satisfaction of surgeons and patients (poor, fair, good, very good).
STATISTICAL ANALYSIS:
Data analysis was performed with Minitab software, version 19.0 (Minitab LLC, State College, Pennsylvania, United States) and RStudio software, version 1.1.456 (RStudio Inc., Boston, Massachusetts, United States). The Anderson-Darling test was used to check the normality of data. Normally distributed variables are presented as mean±SD. Non-normally distributed variables are presented as medians with interquartile ranges. A 2-sample, independent, unpaired
Results
Of the 120 patients recruited for the present study, 1 was excluded due to a history of opioid use. The remaining 119 patients were randomly divided into 3 groups and completed the study. There were no significant between-group differences with respect to baseline characteristics (Table 1).
The primary observation index was the incidence of limb movement. The incidence rates in groups A, B, and C were 32.5%, 12.5%, and 0, respectively (
The proportions of patients who required propofol bolus before the EIS in groups A, B, and C were 35%, 22.5%, and 2.6%, respectively (
The proportions of surgeons in groups A, B, and C who rated their satisfaction “very good” were 52.5%, 77.5%, and 97%, respectively (
The percentages of patients in groups A, B, and C who rated their satisfaction “fair” were 37.5%, 10%, and 5.1%, respectively (
Discussion
In the present study, midazolam was the main sedative used and propofol was the auxiliary sedative. The use of propofol was avoided or minimized owing to its adverse effects on the respiratory and circulatory systems. Administration of midazolam in combination with propofol has been shown to decrease the incidence of respiratory depression as compared to that induced by use of propofol alone [8]. The combination also can be used with flumazenil to induce quick recovery from sedation.
The incidence rates for limb movement in groups A, B, and C were 33%, 13%, and 0, respectively. The best analgesic effect was observed in Group C. However, the incidence of hypoxia in Group C before EIS was up to 33%, which was significantly higher than that in groups A (15%) and B (7.5%); this may be related to the higher dose of oxycodone used in Group C. The incidence of complications was also increased in Group C [9–11]. However, there was no hypoxia in Group C during EIS at 8 min after the administration of oxycodone, which may be related to the 1- to 3-min and 4- to 6-min times for onset and peak effect of oxycodone, respectively [12–16]. In previous studies, the incidence of hypoxia induced by oxycodone was highest within 8 min after IV infusion [17]. The onset time for midazolam is 30 to 60 s, and it reaches a peak in 4 min. This is also the main reason that the surgeons in Group C had the highest satisfaction scores. The incidence of hypoxia in Group A was 23%, which may be related to the addition of propofol during EIS. However, higher doses of oxycodone did not cause more complications than lower doses of the drug [18].
Currently, oxycodone is widely used in day surgery because of its unique advantages [19,20], especially for painless gastroscopy [5,21]. However, there is a paucity of studies on the use of oxycodone during EIS.
In the present study, the rate of propofol use before EIS in Group C was significantly lower than that in the other 2 groups, which may be related to the significant increase in sedative effect of oxycodone in Group C. During EIS, the dosage of propofol was the lowest in Group C and the highest in Group A, which was related to the less effective perioperative analgesia with low-dose oxycodone and need for the addition of propofol. In addition, the patients in Group A required significantly more postoperative analgesia than those in groups B and C, which is why the satisfaction scores for postoperative analgesia were significantly higher in groups B and C than in Group A. There was no significant difference between groups B and C in postoperative analgesia requirement. There was also no difference between the 2 groups in patient satisfaction (fair+poor) (Group B, 7/40, 18%; Group C, 4/39, 10%), which may be due to the fact that a single dose of oxycodone before EIS did not provide adequate postoperative analgesia in patients undergoing EIS.
The limitations of the present study are that the injection sites in each patient differed and the sclerotherapy doses varied, depending upon the location and severity of the esophageal varices. In addition, although all of the endoscopic procedures were performed by skilled senior surgeons, there were still differences in their individual levels of manual dexterity.
In conclusion, midazolam administered in combination with 0.125 mg/kg of oxycodone was the dosage most conducive to meeting surgeons’ needs for adequate patient immobilization during EIS for esophageal varices and was not associated with an increased incidence of perioperative adverse effects.
Conclusions
The present study suggests that 0.125 mg/kg of oxycodone is the effective dose for providing perioperative analgesia for EIS of esophageal varices.
Tables
Table 1. Baseline characteristics of the patients. Table 2. Comparison of adverse effects among the 3 groups.
Table 2. Comparison of adverse effects among the 3 groups.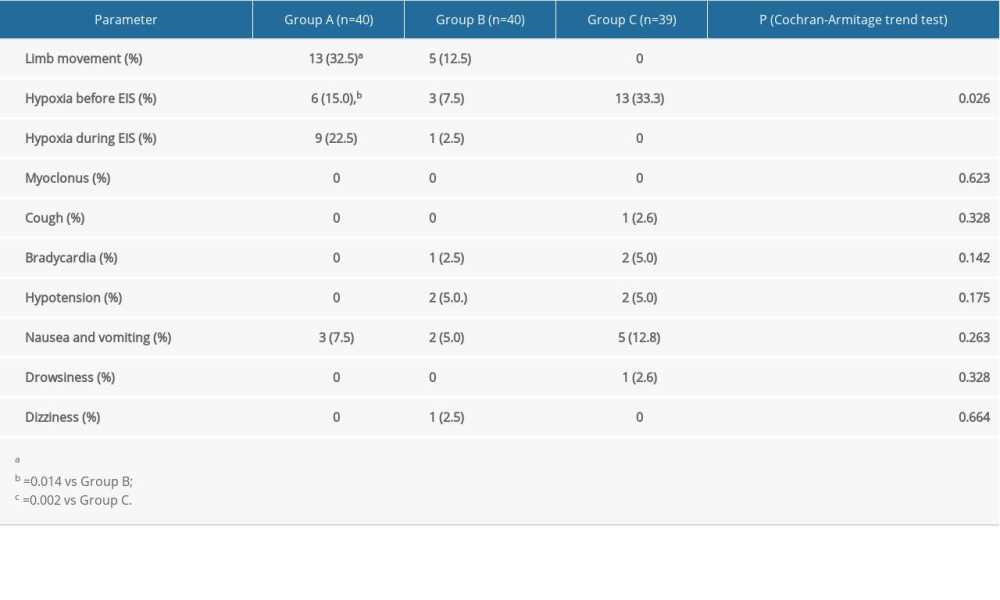 Table 3. Comparison of propofol bolus among the 3 groups.
Table 3. Comparison of propofol bolus among the 3 groups.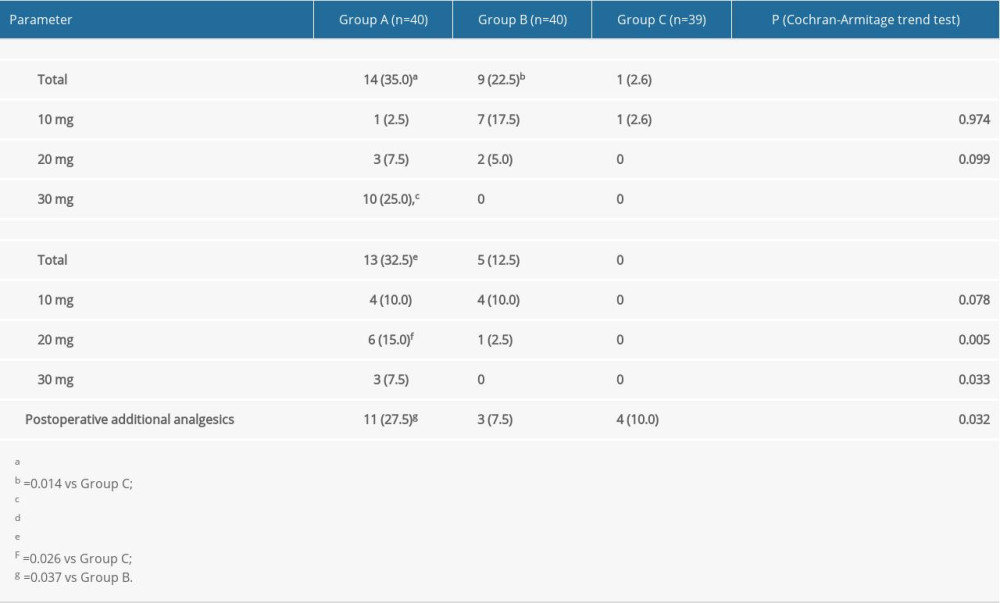 Table 4. Comparison of physician satisfaction among the 3 groups.
Table 4. Comparison of physician satisfaction among the 3 groups.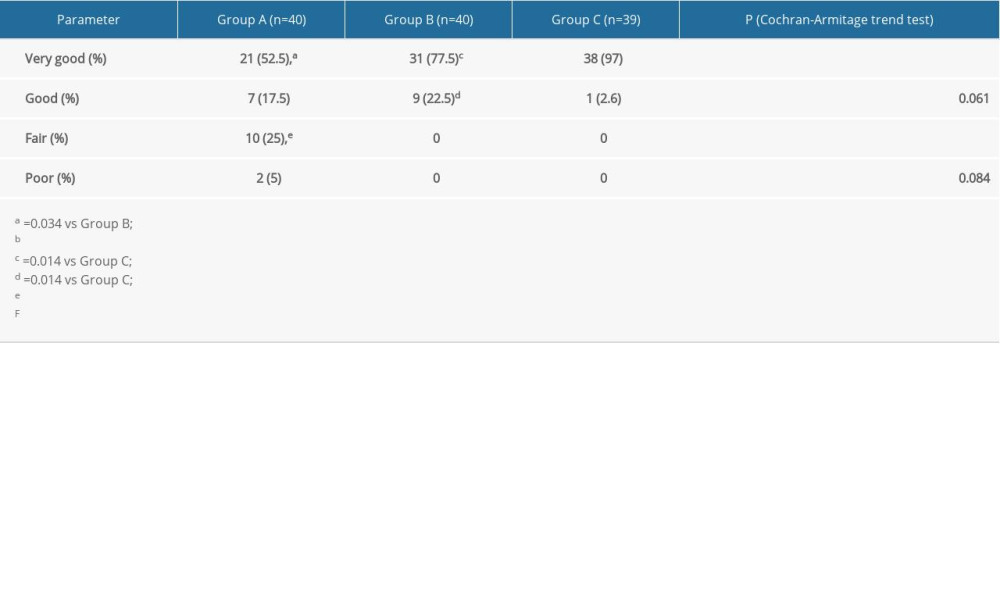 Table 5. Comparison of patient satisfaction among the 3 groups.
Table 5. Comparison of patient satisfaction among the 3 groups.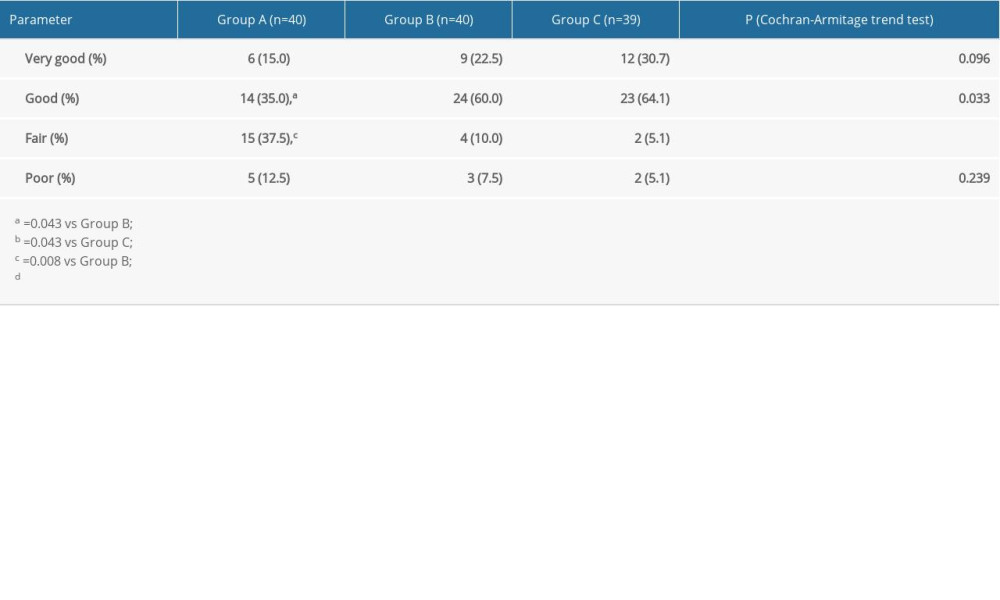
References
1. Xiaoqing Z, Na L, Lili M, Endoscopic cyanoacrylate injection with lauromacrogol for gastric varices: Long-term outcomes and predictors in a retrospective cohort study: J Laparoendosc Adv Surg Tech A, 2019; 29; 1135-43
2. Ali SM, Wu S, Xu H, A prospective study of endoscopic injection sclerotherapy and endoscopic variceal ligation in the treatment of esophageal varices: J Laparoendosc Adv Surg Tech A, 2017; 27; 333-41
3. Wang L, Xu X, Hou Y, Acute mesenteric vein thrombosis after endoscopic injection sclerotherapy for esophageal varices in a patient with liver cirrhosis: Drug Discov Ther, 2019; 13; 118-21
4. Yu Y, Qi S-L, Zhang Y, Role of combined propofol and sufentanil anesthesia in endoscopic injection sclerotherapy for esophageal varices: World J Gastroenterol, 2017; 23; 7875-80
5. Quan Z, Luo C, Chi P, Analgesic effects of oxycodone relative to those of sufentanil, in the presence of midazolam, during endoscopic injection sclerotherapy for patients with cirrhosis and esophageal varices: Anesth Analg, 2018; 127; 382-86
6. Han L, Su Y, Xiong H, Oxycodone versus sufentanil in adult patient-controlled intravenous analgesia after abdominal surgery: A prospective, randomized, double-blinded, multiple-center clinical trial: Medicine (Baltimore), 2018; 97(31); e11552
7. Pergolizzi JV, Seow-Choen F, Wexner SD, Perspectives on intravenous oxycodone for control of postoperative pain: Pain Pract, 2016; 16(7); 924-34
8. Kang R, Shin YH, Gil NS, A comparison of the use of propofol alone and propofol with midazolam for pediatric magnetic resonance imaging sedation: A retrospective cohort study: BMC Anesthesiol, 2017; 11(1); 138
9. Cuiping Y, Fan T, Wang P, Effects of different doses of oxycodone hydrochloride on spontaneous breathing and consciousness level of patients: Herald Med, 2017; 36; 905-8
10. Hill R, Dewey WL, Kelly E, Oxycodone-induced tolerance to respiratory depression: Reversal by ethanol, pregabalin and protein kinase C inhibition: Br J Pharmacol, 2018; 175; 2492-503
11. van der Schrier R, Roozekrans M, Olofsen E, Influence of ethanol on oxycodone-induced respiratory depression: A dose-escalating study in young and elderly individuals: Anesthesiology, 2017; 26; 534-42
12. Kinnunen M, Piirainen P, Kokki H, Updated clinical pharmacokinetics and pharmacodynamics of oxycodone: Clin Pharmacokinet, 2019; 58(6); 705-25
13. Kim N-S, Kang KS, Yoo SH, A comparison of oxycodone and fentanyl in intravenous patient-controlled analgesia after laparoscopic hysterectomy: Korean J Anesthesiol, 2015; 68; 261-66
14. Choi BM, Lee YH, An SM, Population pharmacokinetics and analgesic potency of oxycodone: Br J Clin Pharmacol, 2017; 83(2); 314-25
15. Wang N, Zhou H, Song X, Comparison of oxycodone and sufentanil for patient-controlled intravenous analgesia after laparoscopic radical gastrectomy: A randomized double-blind clinical trial: Anesth Essays Res, 2016; 10; 557-60
16. Ruan X, Mancuso KF, Kaye AD, Revisiting oxycodone analgesia: A review and hypothesis: Anesthesiol Clin, 2017; 35(2); e163-74
17. Kalso E, Pharmacokinetics and ventilatory effects of intravenous oxycodone in postoperative children: Br J Clin Pharmacol, 1995; 39; 214-15
18. Koh GH, Jung KT, So KY, Effect of different doses of intravenous oxycodone and fentanyl on intubation-related hemodynamic responses: A prospective double-blind randomized controlled trial: Medicine (Baltimore), 2019; 98; e15509
19. Choi BM, A new therapeutic option for postoperative pain management with oxycodone HCI injection: Korean J Anesthesiol, 2016; 69; 211-18
20. Wu J, Gui Q, Wang J, Oxycodone preemptive analgesia after endoscopic plasma total adenotonsillectomy in children: A randomized controlled trial: Medicine (Baltimore), 2020; 99; e19004
21. Yin N, Xia J, Cao YZ, Effect of propofol combined with opioids on cough reflex suppression in gastroscopy: Study protocol for a double-blind randomized controlled trial: BMJ Open, 2017; 7; e014881
Tables
In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








