16 November 2020: Lab/In Vitro Research
Construction of a Mex3c Gene-Deficient Mouse Model to Study C-FOS Expression in Hypothalamic Nuclei and Observe Morphological Differences in Embryonic Neural Tube Development
Yong Du12ABDE*, Quan Huo3BCEF, Ting Li3BCD, Dongjun Sun3BEF, Ting Sun4BCD, Zhiguo Lu3BEFDOI: 10.12659/MSM.927334
Med Sci Monit 2020; 26:e927334
Abstract
BACKGROUND: This study utilized CRISPR/Cas9 gene editing technology to construct a Mex3c gene-deficient mouse model, and studied C-FOS expression in hypothalamic nuclei.
MATERIAL AND METHODS: Thirty Mex3c–/+ mice, 30 mice in the normal group, and 30 Mex3c–/+ mice were randomly divided into control, leptin, and ghrelin groups according to different intraperitoneal injections. HE and Nissl staining were performed to observe the morphology of hypothalamic nerve cells. The C-FOS expression in hypothalamic nuclei of each group was analyzed by immunohistochemical techniques. HE staining was used to observe neural tube morphology, and LFB staining was used to observe nerve myelin sheath morphology. TEM was used to observe neuronal ultrastructure and immunohistochemical techniques were utilized to analyze nestin expression.
RESULTS: C-FOS expression was lower in the normal control group than in the leptin and ghrelin groups. The Mex3c control group and the leptin group had higher C-FOS expression than the ghrelin group. In neural tube studies, no significant differences were found in the neural tube pathological sections of E14.5-day embryos in each group. Nestin results demonstrated lower expression in the normal group and there was little difference between the HD and Mex3c groups.
CONCLUSIONS: Mex3c appears to participate in the regulation of energy metabolism by inducing C-FOS expression in the hypothalamus. The neural tubes of the offspring of Mex3c–/+ mice had defects during development.
Keywords: clustered regularly interspaced short palindromic repeats, Energy Metabolism, Hypothalamus, Neural Tube, Cell Nucleus, Embryo, Mammalian, Genotype, Mice, Knockout, Models, Animal, Proto-Oncogene Proteins c-fos, RNA-Binding Proteins
Background
CRISPR (clustered regularly interspaced short palindromic repeats) and CRISPR-associated endonuclease Cas is a genetic editing technology that has emerged in recent years, called CRISPR/Cas9 [1]. The acquisition time of mutant mice and rats can be significantly shortened, from 1 year to several weeks, which satisfies the needs of investigators for rapid and effective editing of organism genome, modifying cations, and the construction of specific gene knockout animal models [2,3]. Through this approach, we obtained Mex3c gene-deficient mice that meet the experimental demands.
The energy balance depends upon the balance between food intake regulation and energy expenditure. The hypothalamic nucleus of the paraventricular nucleus (PVN), arcuate nucleus (Arc), medial nucleus (DMN), ventromedial nucleus (VMN), and lateral nucleus (LHA) affect appetite, which plays a vital role in energy balance regulation [4]. Since the initial study of
Neural tube defects are common and serious congenital malformations, the etiology of which is complex, and can be caused by biological genetic and environmental factors [13]. The important role of energy metabolism in neural tube closure has been validated in some animal models. For instance, Yamaguchi et al. reported that, during mammalian pregnancy, neural tube defects can be caused by malnutrition and insufficient or excessive intake of specific nutrients [14]. The Mex3c gene has an important role in energy metabolism process in the body. This study aimed to explore the mechanism by which Mex3c regulates energy balance and to assess whether it participates in neural tube formation.
Material and Methods
CONSTRUCTION OF THE MEX3C GENE-DEFICIENT MOUSE MODEL:
The Mex3c gene-deficient mouse model was constructed by Cyagen Biosciences (Guangzhou), Inc. Three male and 3 female mice were obtained and raised at the Ningxia Medical University SPF Laboratory Animal Center (12-h light/dark cycle, temperature 22±2°C, humidity 50±5%, with clean feed and water) to breed a sufficient number of experimental mouses. The Animal Experimental Ethics Committee of Ningxia Medical University approved this study, and the animal treatment process was in line with animal ethics standards. 30 wild-type (WT) and 30 Mex3c−/+ mice (8 weeks old; female) were used. Mice in each group were randomly divided into a control group, a leptin group, and a starvation hormone group. Sodium pentobarbital (1%) was intraperitoneally injected for anesthesia. We opened the chest to expose the heart, cut the left atrium, and rinsed it with 20 mL of normal saline from the right ventricle. After the left ventricle outflow was clear, we performed continuous infusion with 4% paraformaldehyde 10 mL with slow injection over a period of 10 min. The mouse brain was removed after anesthesia and we obtained paraffin-embedded preservation tissue. The WT and Mex3c−/+ mice were fed with normal feed and high-sugar and high-fat diets (experimental Animal Premix 2.0%, calcium hydrogen phosphate 2.0%, soy protein isolate 10.0%, lard 12%, sucrose 20%, cholesterol 1.0%, standard mouse maintenance feed 53%; Jiangsu Province Collaborative Medicine Bioengineering Co.). Each group had 10 mice, and the experiment was started at 8 weeks of age. After 12 weeks of continuous feeding, the WT male mice were caged with the female mice at 8: 00 pm every day. At 8: 00 am the next morning, the female mice were found have the vaginal embolism recorded at 0.5 days of pregnancy. With the feeding conditions unchanged, we anesthetized the females after 14.5 days of pregnancy. The embryos were removed and embedded in paraffin.
MOUSE ACQUISITION AND GENETIC IDENTIFICATION:
When F0 generation mice were 8 weeks of age, males and females were bred to obtain an F1 generation. When the new female mice were 3 weeks old, after routine disinfection, we cut 3-mm-long tail pieces into 1.5-mL EP tubes and marked them for later use. We added 80 μL of Direct PCR Tail (Viagen Biotech, USA) to each EP tube and kept the tubes in a 55°C water bath overnight. On the next day, 1 μL of proteinase K was added at 80°C for 1 h to extract tissue DNA. We designed specific primers to identify Mex3c gene-deficient mice by PCR. The primer sequences were as follows:
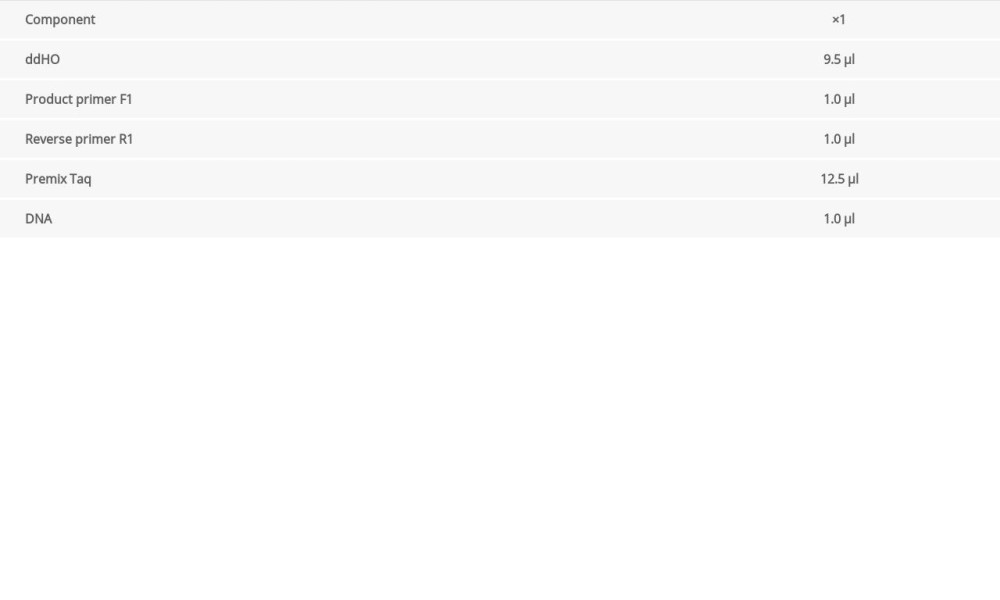
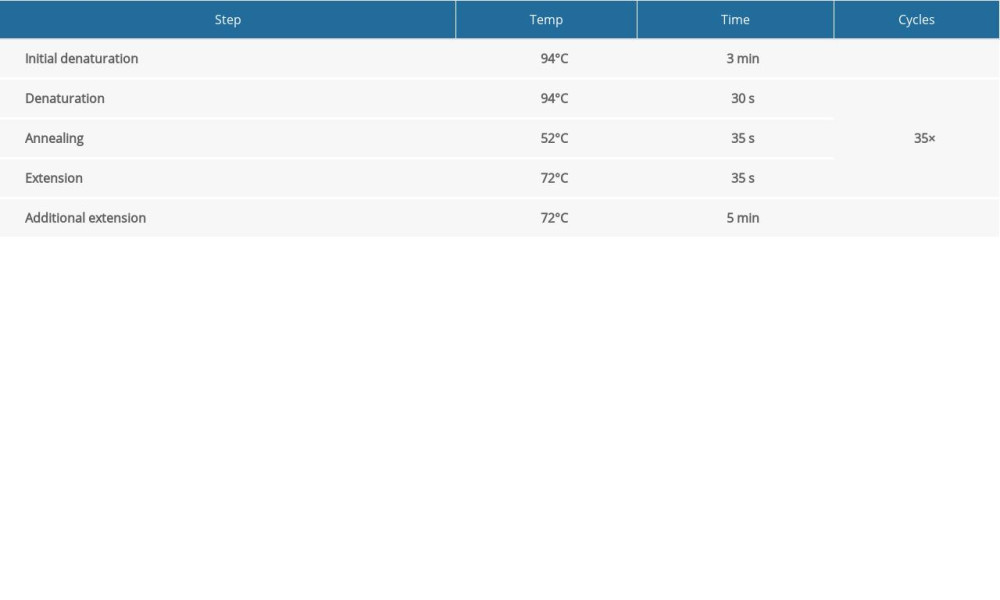
The circulation system is listed in Table 1. We configured a 1.5% agarose gel for electrophoresis and the electrophoresis system is shown in Table 2. According to the electrophoresis results, WT mice and Mex3c gene-deficient mice were identified, and they were housed in cages for use in subsequent experiments.
PARAFFIN EMBEDDING OF MOUSE BRAIN AND EMBRYO:
At 2 h after intraperitoneal injection in the saline group, leptin group, and hunger hormone group, we intraperitoneally injected anesthesia with 1% sodium pentobarbital (0.05 mg/g). After routine disinfection, the heart was opened and the left atrium was cut out and removed. We fully flushed the right atrium with normal saline. After the effluent was clear, we injected 4% paraformaldehyde for about 10 min. We carefully removed the brain tissue and placed it in freshly prepared 4% paraformaldehyde for 1 day. On the next day, we washed the brain tissue with phosphate-buffered saline (PBS), replaced PBS solution every hour 6 times, and then dehydrated with gradient alcohol (alcohol concentration was 70%, 80%, 90%, 95%, 100%, and 100%). We fully infiltrated the tissue with xylene. Paraffin waxes I and II were melted at 65°C and the tissue was immersed in the wax for 2 h for paraffin embedding. At day 14.5 of pregnancy, we routinely performed sterilization and anesthesia and found the “Y” type distribution uterus. Afterwards, we carefully separated the gestational sac, and treated the embryo tissue as previously described [1]. We then dehydrated it with gradient alcohol (alcohol concentration 30%, 50%, 70%, 80%, 90%, 95%, 100%, and 100%). Tissue was thoroughly permeated with toluene and immersed in paraffin I and II for 1 h at 65°C as paraffin embedding.
IMMUNOHISTOCHEMICAL STAINING OF MOUSE HYPOTHALAMIC NUCLEAR AND EMBRYONIC NEURAL TUBES:
Each paraffin-embedded tissue was sectioned with a vibrating slicer (Leica) to a thickness of 4 μm. We used the Mouse Brain Stereotactic Map (second edition, prepared by Keith B. J. Franklin) to locate Arc, VMN, DMN, and PV nuclei of the hypothalamus. We performed conventional dewaxing and hydration and used citrate antigen repair solution for high-pressure repair for 10 min. We used 3% H2O2 for removal of endogenous peroxidase, then goat serum blocking and addition of mouse-derived C-FOS primary antibody (1: 300, Proteintech, USA) were performed overnight at 4°C. The tissue was washed with PBS solution according to the rabbit anti-mouse secondary antibody kit instructions (ZSGB-BIO, China). Positive expression was detected by analyzing the percentage of unit area using ImageJ software. We used the nestin (1: 300, Bioss, China) expression to assess the development of the mouse embryonic neural tube. The experimental steps used were similar to those previously described [1].
ELECTRON MICROSCOPIC OBSERVATION OF ULTRASTRUCTURE:
Mouse embryos obtained at 14.5 days of pregnancy were quickly placed in glutaraldehyde for 2 h. We then washed them 3 times in buffer for 2 h. They were processed with osmium tetroxide fixation, then dehydrated, embedded, and sectioned under a Hitachi H-7650 transmission electron microscope (Tokyo, Japan). We observed the development of synapses and mitochondria in mouse embryonic neural tubes.
STATISTICAL ANALYSIS:
We used SPSS 17.0 statistical software (SPSS, Inc., Chicago, IL, USA) to analyze the experimental data. Measurement data are denoted by mean±SEM (X±S). One-way analysis of variance and SNK-q test were utilized for comparison between groups.
Results
:
The F1 mice obtained by breeding were subjected to Mex3c knockout identification, in which M stands for marker band and 3 consecutive wells represent 1 mouse. F1 primer 640 bp shows a bright band to indicate gene knockout success. F2 primer 499bp-positive means wild-type, combined with F1. The F2-positive result determines pure mouse zygote, heterozygous, wild-type. The F primer 335 bp is the internal reference primer, and both the wild-type and the KO-positive have a strip which ensures that the sample DNA was successfully extracted during the experiment and the PCR was successful. Finally, 30 Mex3c gene-deficient mice were obtained according to the electrophoresis results (Figure 1, PCR results indicated).
BRAIN HISTOMORPHOLOGY STAINING:
The hypothalamic partial nuclei of mice were observed by HE staining and Nissl staining technique. HE staining showed that the nerve cells in each nucleus were densely packed and densely clustered, with normal morphology and structure of the nucleus tissue. The cytoplasmic staining of the nucleus was dense, with no damage or degeneration, necrotic neurons were observed, and the tissue staining was uniform, anatomical, and clear. Nissl staining showed that the nucleus of the neurons was obviously stained blue, and the cytoplasm was light blue. It can be seen that the Nissl body is evenly distributed in the perinuclear cytoplasm, dyeing is deep or shallow, found in the cytoplasm or in the periphery of the cell, and no staining becomes shallow and blurred. The neuronal cells of each group were large and round and were located in Arc, VMN, DM, and PV nuclei. There was no nuclear dissolution or nuclear pyknosis, and no obvious vacuoles (Figures 2, 3).
C-FOS EXPRESSION IN THE HYPOTHALAMUS:
Immunohistochemistry results showed that the 3 intervention treatments of WT mice and Mex3c−/+ mice were intraperitoneal injection of normal saline, leptin (causing a negative energy balance effect), and ghrelin (causing a positive energy balance effect). The expression trends of C-FOS in each group were similar in the nuclei of the hypothalamus. We found that expression of C-FOS in the normal control group was very low, but after intervention with leptin and ghrelin, the expression of C-FOS was greatly increased (p<0.001). In the Mex3c−/+ mice, the expression of C-FOS was significantly higher in the control group, and the expression of C-FOS was not significantly changed after leptin treatment (P>0.05). However, the expression of C-FOS was significantly decreased after injection of ghrelin compared with the Mex3c−/+ control group (P<0.001), and the difference was significant. Finally, we found that the normal control group and the Mex3c−/+ ghrelin group had similar C-FOS expression levels and they had no differences in DM and PV nuclei (P>0.05) (Figure 4).
MORPHOLOGICAL DIFFERENCES IN NEURAL TUBE DEVELOPMENT:
We used HE staining, Luxol fast blue (LFB), and immunohistochemical detection to analyze nestin expression in neural tubes. We observed the neural tube ultrastructure by electron microscopy to explore the morphological differences in neural tube development in offspring of the 3 groups of embryonic mice. HE staining showed no significant differences in the morphology of neural tube cells in the control group, Mex3c−/+, and high-sugar and high-fat diet (HD) groups. In the LFB staining results, we observed that myelin sheaths in the control group had a deeper color than those in the other 2 groups. Neural tube transmission electron microscopy (TEM) showed that, compared with the control group, the mitochondrial edema of neuron cells in the neural tube of the embryonic mice of Mex3c−/+ and HD groups showed larger numbers of vacuoles. The mitochondrial cristae were broken and the structure was clearly abnormal in the neural tubes of embryonic mice of the Mex3c−/+ and HD groups. The presynaptic membrane and postsynaptic membrane were clear but the synaptic space disappeared. The synapses had a certain degree of fusion and the typical structure was difficult to find. We observed that neuronal cells were damaged, and their nuclear chromatin was lost. Also, the nuclei had pyknosis. According to TEM results, Mex3c−/+ and HD group mouse embryos had this particular defect during neural tube development. Using immunohistochemical staining technology to detect nestin expression in mouse embryonic neural tubes in each group, we found that the expression of this factor in the Mex3c−/+ and HD groups were not significantly different (P>0.05) and its expression was significantly increased compared with the control group (P<0.05) (Figure 5).
Discussion
In this study, we successfully constructed a Mex3c gene-deficient mouse model. The new CRISPR/Cas9 technology was used to powerfully suppress or activate the target gene. The operation makes this technology useful in biology and clinical research [15]. Using this technology, we obtained the Mex3c gene-deficient mice and found that Mex3c−/− mice were much weaker than normal mice and Mex3c−/+ mice, which had slower weight gain and lower activity after birth. At 1 month of age or after cutting off mouse tails for genotyping, some mice died. Lu et al. reported that Mex3c-deficient mice died due to poor respiration after birth, similar to IGF1-deficient mice [8]. Therefore, in the present study, we decided to use Mex3c−/+ mice instead of Mex3c−/− mice, which had better physiological conditions than homozygotes. The mice could survive to adulthood and met the requirements of reproductive offspring. The Mex-3 protein was discovered during the study of
The generation of neural tubes is a major event in the development of all animals, and is closely related to subsequent generation of the central and peripheral nervous systems. During neural tube development, the neural plate must be correctly closed accurately to form the neural tube. Otani and Kappen et al. found that during pregnancy of diabetic and hyperglycemic mice, the embryos were not closed at E10.5 days, but the neural tubes of mice of the same strain without diabetes were completely closed at this time [22]. Also, the neural tube closure time in the avian embryos in a hyperglycemic environment was significantly delayed [23]. Disturbances in maternal nutrition and metabolism have significant effects on the development of neural tubes in offspring. Numerous studies confirmed that supplementation with folic acid during pregnancy can significantly reduce the risk of NTDs [24,25]. We chose to use E14.5-day mouse embryos as the in the present study. Compared with mouse embryos in the normal group, the high-fat-diet group and Mex3c mouse embryos not only had difficult pregnancy, but also had fewer offspring. There were different numbers of stillbirths and absorption fetuses, but no neural tube-malformed embryos were observed. Observing the surviving mice in each group, there were no obvious differences in appearance in the 3 groups of mouse embryos. Regularly arranged neurons were seen in pathological sections, and nerve fibers were obvious. The myelin sheath is made of sphingomyelin, which is a tubular sheath wrapped around an axon. LFB staining is a commonly used and reliable staining method for myelin sheaths [26,27]. When the myelin sheath of immature mice is immature, the sensitivity to LFB staining is higher. LFB staining showed that the normal group was significantly darker, possibly because the degree of myelin sheath development was different between the groups. TEM showed that mitochondria and synapses in the microsystem were impaired or functionally impaired in the HD and Mex3c−/+ groups. Nestin is encoded by the nestin gene, which is a unique marker of neural stem cells. Nestin begins to be expressed when neural stem cells start to differentiate into neuronal cells. Accompanying the differentiation of neural cells, nestin expression gradually weakens [28]. Immunohistochemistry results showed that the highest expression of nestin per unit area in the neural tube was in the Mex3c−/+ group and the HD group was slightly lower. The normal group had a significantly lower expression percentage than the other 2 groups (
Conclusions
We constructed a Mex3c gene-deficient mouse model using CRISPR/Cas9 technology. Through reproduction and gene identification, we obtained genetically stable and heterozygous mice. We found that Mex3c was involved in the process of energy metabolism in the body by increasing C-FOS expression in the hypothalamus. In addition, there were some defects in the neural tube of Mex3c−/+ female embryos during development, but the specific mechanism is unclear and needs further investigation.
Figures
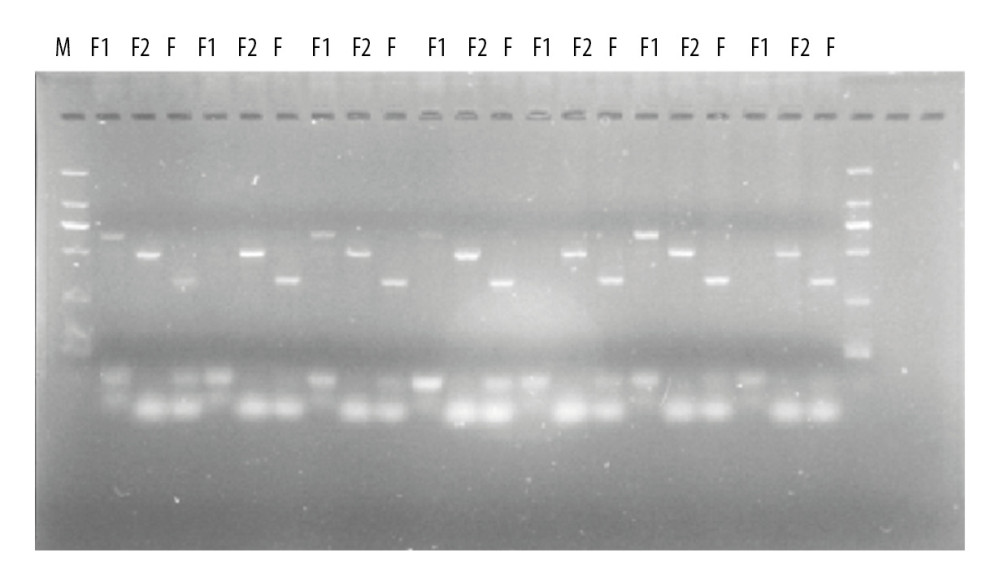 Figure 1. PCR identification of mouse gene gel plate results.
Figure 1. PCR identification of mouse gene gel plate results. 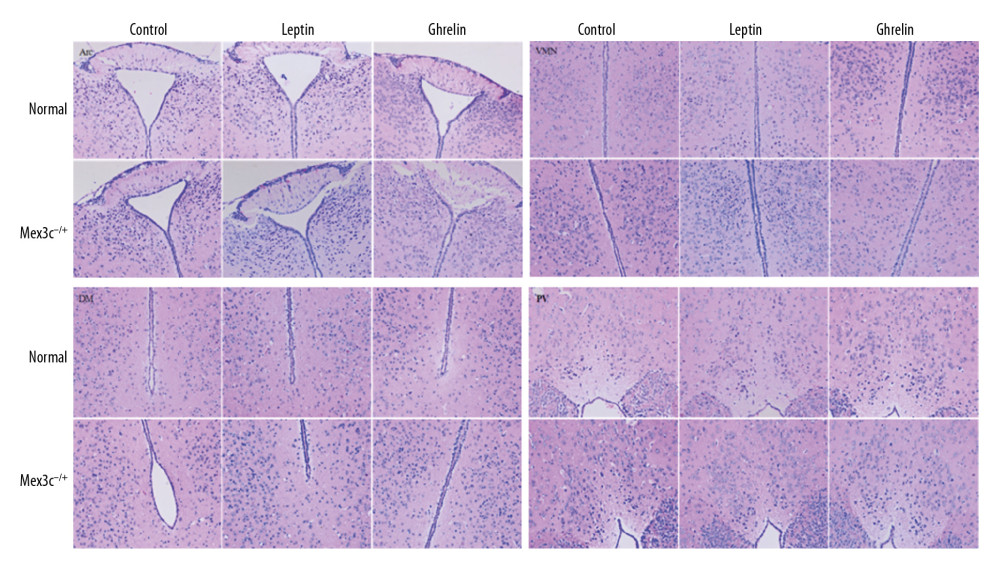 Figure 2. H&E staining results of hypothalamic Arc, VMN, DM, and PV nuclei in each group of mice (×200). Scale bar=100 μm.
Figure 2. H&E staining results of hypothalamic Arc, VMN, DM, and PV nuclei in each group of mice (×200). Scale bar=100 μm. 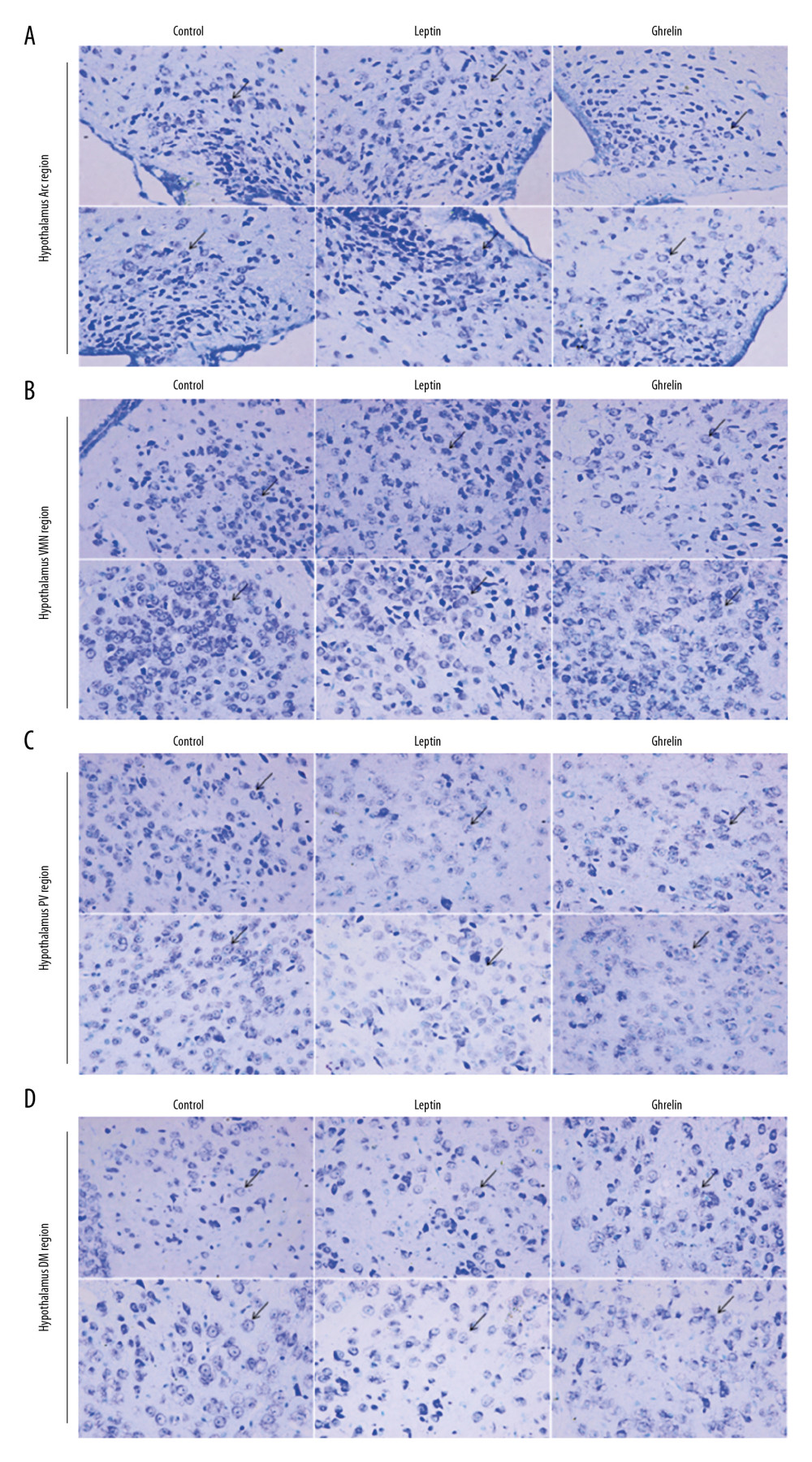 Figure 3. (A) Nissl staining results of hypothalamic Arc nuclei in each group of mice (×400). (A) Arc nuclei (×400). (B) VMN nuclei (×400). (C) DM nuclei (×400). (D) PV nuclei (×400). Scale bar=50 μm. Black arrow indicates neuron cells.
Figure 3. (A) Nissl staining results of hypothalamic Arc nuclei in each group of mice (×400). (A) Arc nuclei (×400). (B) VMN nuclei (×400). (C) DM nuclei (×400). (D) PV nuclei (×400). Scale bar=50 μm. Black arrow indicates neuron cells. 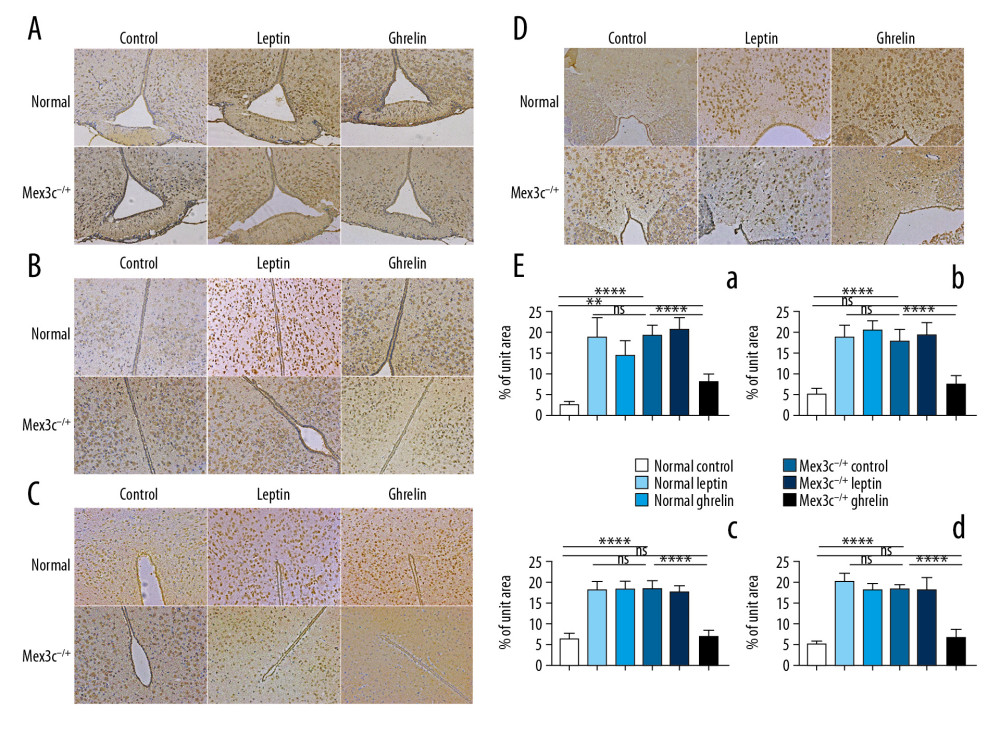 Figure 4. C-FOS expression of hypothalamic nuclei. (A) Arc nuclei (×200). (B) VMN nuclei (×200). (C) DM nuclei (×200). (D) PV nuclei (×200). Scale bar=100 μm. (E) The 2 ends of the horizontal line indicate a comparison between the 2 groups. a–d is a bar chart comparing the C-FOS expression levels of the hypothalamic A–D nucleus (* P<0.05, ** P<0.01, **** P<0.001, ns: no significance).
Figure 4. C-FOS expression of hypothalamic nuclei. (A) Arc nuclei (×200). (B) VMN nuclei (×200). (C) DM nuclei (×200). (D) PV nuclei (×200). Scale bar=100 μm. (E) The 2 ends of the horizontal line indicate a comparison between the 2 groups. a–d is a bar chart comparing the C-FOS expression levels of the hypothalamic A–D nucleus (* P<0.05, ** P<0.01, **** P<0.001, ns: no significance). 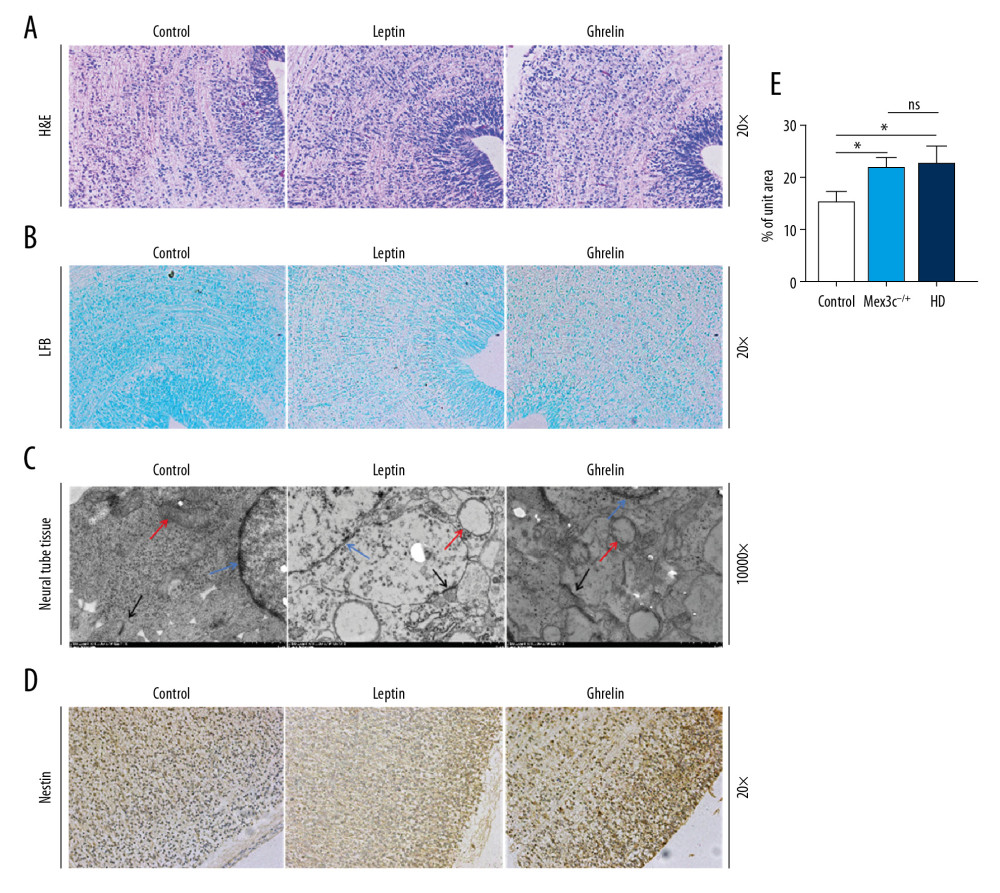 Figure 5. Morphological differences in mouse embryonic neural tube at 14.5 days. (A) H&E staining (200×). (B) LFB staining (200×). (C) Transmission electron microscopy (TEM) result (10 000×). Red arrows represent mitochondria, black arrows represent synapses, blue arrows represent neuron nuclei. (D) Nestin expression in each group as determined by immunohistochemistry (200×). (E) Bar chart of nestin expression (* P<0.05). Scale bar =100 μm.
Figure 5. Morphological differences in mouse embryonic neural tube at 14.5 days. (A) H&E staining (200×). (B) LFB staining (200×). (C) Transmission electron microscopy (TEM) result (10 000×). Red arrows represent mitochondria, black arrows represent synapses, blue arrows represent neuron nuclei. (D) Nestin expression in each group as determined by immunohistochemistry (200×). (E) Bar chart of nestin expression (* P<0.05). Scale bar =100 μm. References
1. Hsu PD, Lander ES, Zhang F, Development and applications of CRISPR-Cas9 for genome engineering: Cell, 2014; 157(6); 1262-78
2. Sander JD, Joung JK, CRISPR-Cas systems for editing, regulating and targeting genomes: Nat Biotechnol, 2014; 32(4); 347-55
3. Hryhorowicz M, Lipiński D, Zeyland J, Słomski R, CRISPR/Cas9 Immune System as a Tool for Genome Engineering: Arch Immunol Ther Exp (Warsz), 2017; 65(3); 233-40
4. Berthoud HR, Multiple neural systems controlling food intake and body weight: Neurosci Biobehav Rev, 2002; 26(4); 393-428
5. Greene LA, Tischler AS, Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor: Proc Natl Acad Sci USA, 1976; 73(7); 2424-28
6. Burrell RA, McClelland SE, Endesfelder D, Replication stress links structural and numerical cancer chromosomal instability [published correction appears in Nature, 2013; 500(7463): 490]: Nature, 2013; 494(7438); 492-96
7. Han C, Jiao Y, Zhao Q, Lu B, Mex3c mutation reduces adiposity partially through increasing physical activity: J Endocrinol, 2014; 221(3); 457-68
8. Jiao Y, Bishop CE, Lu B, Mex3c regulates insulin-like growth factor 1 (IGF1) expression and promotes postnatal growth: Mol Biol Cell, 2012; 23(8); 1404-13
9. Watts AG, Khan AM, Sanchez-Watts G, Activation in neural networks controlling ingestive behaviors: What does it mean, and how do we map and measure it?: Physiol Behav, 2006; 89(4); 501-10
10. Kovács KJ, Measurement of immediate-early gene activation – c-fos and beyond: J Neuroendocrinol, 2008; 20(6); 665-72
11. Qiu J, Zhang C, Borgquist A, Insulin excites anorexigenic proopiomelanocortin neurons via activation of canonical transient receptor potential channels: Cell Metab, 2014; 19(4); 682-93
12. Dhillon H, Zigman JM, Ye C, Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis: Neuron, 2006; 49(2); 191-203
13. Xue J, Gu H, Liu D, Mitochondrial dysfunction is implicated in retinoic acid-induced spina bifida aperta in rat fetuses: Int J Dev Neurosci, 2018; 68; 39-44
14. Yamaguchi Y, Miyazawa H, Miura M, Neural tube closure and embryonic metabolism: Congenit Anom (Kyoto), 2017; 57(5); 134-37
15. Burstein D, Harrington LB, Strutt SC, New CRISPR-Cas systems from uncultivated microbes: Nature, 2017; 542(7640); 237-41
16. Buchet-Poyau K, Courchet J, Le Hir H, Identification and characterization of human Mex-3 proteins, a novel family of evolutionarily conserved RNA-binding proteins differentially localized to processing bodies: Nucleic Acids Res, 2007; 35(4); 1289-300
17. Nestler EJ, ΔFosB: A transcriptional regulator of stress and antidepressant responses: Eur J Pharmacol, 2015; 753; 66-72
18. Pedroso JA, Buonfiglio DC, Cardinali LI, Inactivation of SOCS3 in leptin receptor-expressing cells protects mice from diet-induced insulin resistance but does not prevent obesity: Mol Metab, 2014; 3(6); 608-18
19. Cowley MA, Smart JL, Rubinstein M, Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus: Nature, 2001; 411(6836); 480-84
20. Okuno H, Regulation and function of immediate-early genes in the brain: Beyond neuronal activity markers: Neurosci Res, 2011; 69(3); 175-86
21. Li X, Li Y, Liu C, Oocyte-specific expression of mouse MEX3C652AA in the ovary and its potential role in regulating maternal fos mRNA: Biol Reprod, 2016; 94(5); 115
22. Otani H, Tanaka O, Tatewaki R, Diabetic environment and genetic predisposition as causes of congenital malformations in NOD mouse embryos: Diabetes, 1991; 40(10); 1245-50
23. Kappen C, Modeling anterior development in mice: diet as modulator of risk for neural tube defects: Am J Med Genet C Semin Med Genet, 2013; 163C(4); 333-56
24. Greene ND, Leung KY, Copp AJ, Inositol, neural tube closure and the prevention of neural tube defects: Birth Defects Res, 2017; 109(2); 68-80
25. Miyazawa H, Yamamoto M, Yamaguchi Y, Miura M, Mammalian embryos show metabolic plasticity toward the surrounding environment during neural tube closure: Genes Cells, 2018; 23(9); 794-802
26. Khodanovich MY, Sorokina IV, Glazacheva VY, Histological validation of fast macromolecular proton fraction mapping as a quantitative myelin imaging method in the cuprizone demyelination model: Sci Rep, 2017; 7; 46686
27. Wang C, Sun C, Hu Z, Improved neural regeneration with olfactory ensheathing cell inoculated PLGA scaffolds in spinal cord injury adult rats: Neurosignals, 2017; 25(1); 1-14
28. Lendahl U, Zimmerman LB, McKay RD, CNS stem cells express a new class of intermediate filament protein: Cell, 1990; 60(4); 585-95
Figures
 Figure 1. PCR identification of mouse gene gel plate results.
Figure 1. PCR identification of mouse gene gel plate results. Figure 2. H&E staining results of hypothalamic Arc, VMN, DM, and PV nuclei in each group of mice (×200). Scale bar=100 μm.
Figure 2. H&E staining results of hypothalamic Arc, VMN, DM, and PV nuclei in each group of mice (×200). Scale bar=100 μm. Figure 3. (A) Nissl staining results of hypothalamic Arc nuclei in each group of mice (×400). (A) Arc nuclei (×400). (B) VMN nuclei (×400). (C) DM nuclei (×400). (D) PV nuclei (×400). Scale bar=50 μm. Black arrow indicates neuron cells.
Figure 3. (A) Nissl staining results of hypothalamic Arc nuclei in each group of mice (×400). (A) Arc nuclei (×400). (B) VMN nuclei (×400). (C) DM nuclei (×400). (D) PV nuclei (×400). Scale bar=50 μm. Black arrow indicates neuron cells. Figure 4. C-FOS expression of hypothalamic nuclei. (A) Arc nuclei (×200). (B) VMN nuclei (×200). (C) DM nuclei (×200). (D) PV nuclei (×200). Scale bar=100 μm. (E) The 2 ends of the horizontal line indicate a comparison between the 2 groups. a–d is a bar chart comparing the C-FOS expression levels of the hypothalamic A–D nucleus (* P<0.05, ** P<0.01, **** P<0.001, ns: no significance).
Figure 4. C-FOS expression of hypothalamic nuclei. (A) Arc nuclei (×200). (B) VMN nuclei (×200). (C) DM nuclei (×200). (D) PV nuclei (×200). Scale bar=100 μm. (E) The 2 ends of the horizontal line indicate a comparison between the 2 groups. a–d is a bar chart comparing the C-FOS expression levels of the hypothalamic A–D nucleus (* P<0.05, ** P<0.01, **** P<0.001, ns: no significance). Figure 5. Morphological differences in mouse embryonic neural tube at 14.5 days. (A) H&E staining (200×). (B) LFB staining (200×). (C) Transmission electron microscopy (TEM) result (10 000×). Red arrows represent mitochondria, black arrows represent synapses, blue arrows represent neuron nuclei. (D) Nestin expression in each group as determined by immunohistochemistry (200×). (E) Bar chart of nestin expression (* P<0.05). Scale bar =100 μm.
Figure 5. Morphological differences in mouse embryonic neural tube at 14.5 days. (A) H&E staining (200×). (B) LFB staining (200×). (C) Transmission electron microscopy (TEM) result (10 000×). Red arrows represent mitochondria, black arrows represent synapses, blue arrows represent neuron nuclei. (D) Nestin expression in each group as determined by immunohistochemistry (200×). (E) Bar chart of nestin expression (* P<0.05). Scale bar =100 μm. In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952