07 May 2022: Animal Study
Characterization of Inflammatory Factors and T Cell Subpopulations in a Murine Model of Kawasaki Disease Induced by Cell Wall Extracts (CAWS)
Huifang Xu1ABG*, Jing Weng1BCE, Quan Bao1CE, Fengyan NieDOI: 10.12659/MSM.936355
Med Sci Monit 2022; 28:e936355
Abstract
BACKGROUND: Kawasaki disease (KD) is an acute vasculitis in children, which ranks as the main cause of acquired heart disease in children in developed countries. The etiopathogenesis of KD remains to be clarified. Our study constructed a KD murine model and monitored the alterations of inflammatory factors and T cell subpopulations.
MATERIAL AND METHODS: Candida albicans cell wall extracts (CAWS) were utilized for inducing KD murine models. After 5 days, the mice were subcutaneously injected with rhG-CSF lasting 5 consecutive days. At 4 weeks, histopathology of hearts and arteries was observed via H&E staining. Inflammatory cytokine, chemokine, and adhesion molecule expression in serum specimens was detected via RT-qPCR and ELISA. T cell subpopulations in cardiac tissues were labeled through immunofluorescence and flow cytometry.
RESULTS: CAWS induced cardiac dysfunction (reduced fraction shortening, increased left ventricle end-diastolic diameter and heart weight/body weight ratio) and cardiac and vascular inflammation, which were ameliorated by rhG-CSF treatment. CAWS-induced mice had increased levels of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α), chemokine (RANTES), and adhesion molecules (MCP-1, VCAM-1, ICAM-1, E-selectin), as well as the decreased levels of anti-inflammatory cytokines (IL-10, G-CSF). Their levels were decreased following rhG-CSF administration. There were decreased CD3+, CD4+, CD8+, CD8+CD45+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations and an increased CD45+ T lymphocyte subpopulation in cardiac tissues of CAWS-induced mice, which were ameliorated by rhG-CSF administration.
CONCLUSIONS: Our findings reveal the characteristics of inflammatory factors and T cell subpopulations in CAWS-induced KD. Moreover, rhG-CSF might be an effective therapeutic regimen against KD.
Keywords: Candida albicans, Colony-Stimulating Factors, Inflammation, Models, Animal, Mucocutaneous Lymph Node Syndrome, T-Lymphocyte Subsets, Animals, Cell Wall, Disease Models, Animal, Mice, T-Lymphocytes
Background
Kawasaki disease (KD) is a disease that causes systemic immune vasculitis, primarily in children under 5 years of age [1]. Systemic vasculitis caused by KD mainly involves small and medium-sized blood vessels, especially coronary arteries [2]. Moreover, coronary artery damage is the most serious complication of KD, comprising coronary dilation, coronary aneurysm (CAA), and stenosis [3]. Intravenous immunoglobulin (IVIG) has been regarded as the cornerstone of KD management [4–6]. Nevertheless, about 10–20% of KD patients do not respond to IVIG treatment, and they are at risk of developing CAA [7]. Hence, it is urgent to develop novel and promising therapeutic regimens to clinically manage KD and its complications.
Material and Methods
ANIMALS:
Four-week-old male C57BL/6J mice were individually housed at 18–24°C, 40–70% relative humidity, with a 12-h light/dark cycle. During the 1-week acclimation period, the mice were allowed free access to water and food. Our experimental procedures were carried out strictly following the National Institutes of Health Guidelines for the Use of Laboratory Animals. Our study was approved by the Animal Ethics Committee of Lanxi People’s Hospital (KY-2020006).
PREPARATION OF CAWS:
CAWS was prepared from the
GROUPING AND ADMINISTRATION:
When all mice were acclimated for 1 week, then randomized into a control group, a CAWS group, and a CAWS+recombinant human granulocyte-colony stimulating factor (rhG-CSF) group (n=9 each group). For the CAWS group and CAWS+rhG-CSF group, at 24 h before administration, the mice were injected intraperitoneally with 20 μg of lipopolysaccharide. Thereafter, 0.2 mL CAWS (0.4 mg/mouse/day) was intraperitoneally administered to mice for 5 days in accordance with the approach established by Ohashi R et al [17]. In the control group, the mice were intraperitoneally injected with 0.2 mL phosphate-buffered solution (PBS) for 5 consecutive days. The mice in the CAWS+rhG-CSF group were subcutaneously injected with rhG-CSF (50 μg/kg/day) on the 5th day after intraperitoneal injection of CAWS for 5 consecutive days. CAWS-induced mice received an equal amount of PBS by subcutaneous injection during the same period. After 4 weeks, all mice were euthanized by intraperitoneally injecting an overdose of sodium pentobarbital, and peripheral blood was taken directly. First, the samples were centrifuged at 1000 rpm at 4°C for 10 min to take the supernatant for enzyme-linked immunosorbent assay (ELISA) and real-time quantitative polymerase chain reaction (RT-qPCR) detection, and the remaining blood cell pellet was used for flow cytometry. The hearts and aortas were harvested and fixed with 10% neutralized formalin.
HEMATOXYLIN AND EOSIN (H&E) STAINING:
The pathological conditions of hearts and arteries were examined by H&E staining. Briefly, paraffin-embedded sections were dewaxed in xylene at room temperature twice (20 min/time). Hydration was accomplished through a series of ethanol (5 mins/time). The sections were stained with hematoxylin for 3 min and rinsed with PBS for 4 min. Then, they were stained with eosin for 3 min, washed quickly with distilled water, and quick-washed with 70%, 80%, and 90% ethanol. Subsequently, the sections were rinsed with 95% ethanol for 30 s and 100% ethanol 2 times (3 min/time). After being treated in xylene, the slices were sealed in neutral resin.
ELISA:
Interleukin (IL)-1β (#KE1416; Immuno Way, Beijing, China), IL-6 (#KE1418; Immuno Way), IL-10 (#KE1486; Immuno Way), granulocyte-colony stimulating factor (G-CSF; #SEA042Mu; Cloud-clone, Wuhan, China), tumor necrosis factor α (TNF-α; #KE1419; Immuno Way), macrophage inflammatory protein-1 α (MIP-1α; #SEA092Mu; Cloud-clone), regulated upon activation normal T cell expressed and presumably secreted (RANTES; #H496-1; Nanjing Jiancheng, China), monocyte chemotactic protein-1 (MCP-1; #H115; Nanjing Jiancheng), vascular cellular adhesion molecule-1 (VCAM-1; #H066; Nanjing Jiancheng), intercellular adhesion molecule 1 (ICAM-1; #H065-1; Nanjing Jiancheng), and E-selectin (#H029; Nanjing Jiancheng) were detected in mouse serum specimens by the corresponding ELISA kits. Blank wells, standard wells, and test sample wells were set separately. We added 100 μL sample diluent to blank wells, and standard or 100 μL test samples were added to standard wells or test sample wells, respectively. The microtiter plate was coated and incubated at room temperature for 120 min. The liquid was discarded and the plate was washed 4 times, with soaking for 1–2 min each, with approximately 350 μL/well. Thereafter, the plate was spun dry and the liquid in the wells was patted dry on absorbent paper. We added 100 μL diluted biotinylated detection antibodies to each well, coating the membranes, and incubated for 2 h. After discarding the liquid in the well and spinning dry, 100 μL of diluted streptavidin-horseradish peroxidase (HRP) was added to each well and incubated for 60 min at room temperature. Then, 100 μL ready-to-substrate was added to each well and incubated for 15 min at room temperature for color development. We used 100-μL stop solutions for stopping the reaction, followed by quantification at an optical density of 450 immediately after mixing.
RT-QPCR:
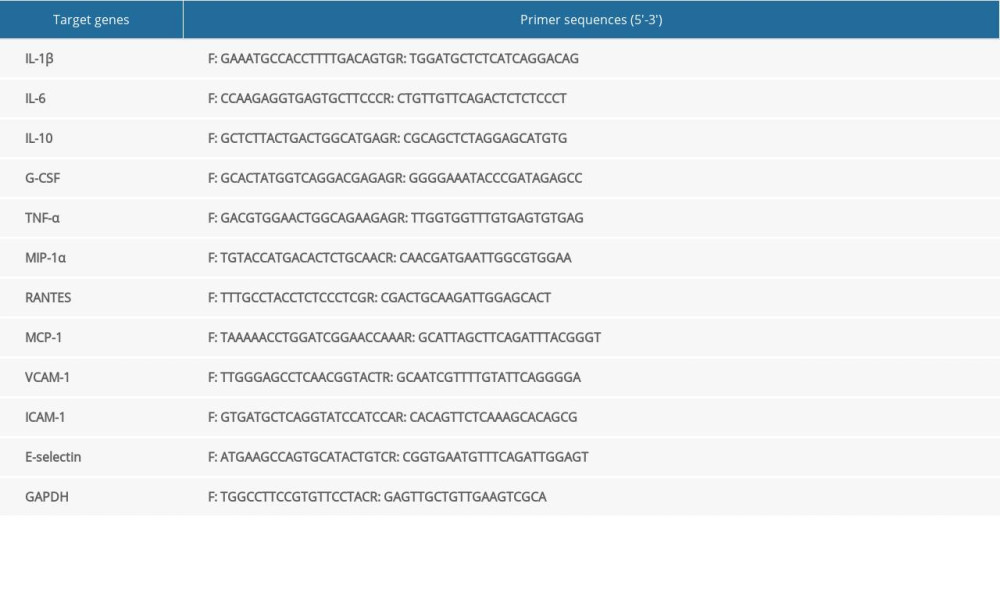
RNA extraction was implemented utilizing TRIzol reagent (Sigma, USA). RNA purity and concentration were evaluated via quantifying the absorbance at 260 and 280 nm. Using the PrimeScript™ RT reagent kit (TaKaRa, China), complementary DNA (cDNA) was acquired. RT-qPCR reactions were conducted on CFX 96™ Real-Time System (Solarbio, China) with SYBR Green. Table 1 listed the primer sequences of mouse IL-1β, IL-6, IL-10, G-CSF, TNF-α, MIP-1α, RANTES, MCP-1, VCAM-1, ICAM-1, and E-selectin. The relative expression was quantified with 2−ΔΔCt approach.
IMMUNOFLUORESCENCE:
After deparaffinization, hydration, and antigen retrieval, cardiac paraffin tissue sections were blocked with goat serum (#C0265; Beyotime, China) for 20 min at room temperature. Primary antibody against CD4 (1: 500; #ab183685; Abcam, USA), CD8 (1: 500; #ab217344; Abcam), Foxp3 (1: 200; #12653; Cell Signaling Technology, USA), F4/80 (1: 100; #70076; Cell Signaling Technology), CD11b (1: 500; #ab133357; Abcam), and Ly-6G (1: 100; #87048s; Cell Signaling Technology) was added dropwise and incubated with the sections at 37°C for 60 min. The excess liquid on the sections was removed by blotting with absorbent paper, and secondary antibody was added dropwise, followed by incubation at 37°C for 1 h. The sections were immersed with PBS 3 times, 3 min each time. Incubation with DAPI was achieved away from light, lasting 5 min, and the excess 4,6-diamidino-2-phenylindole (DAPI; #D9542; Sigma, USA) was washed off 4 times with PBST, 5 min each time. After drying the liquid on the sections with absorbent paper, the sections were sealed with anti-fluorescence quencher and observed under a fluorescence microscope.
FLOW CYTOMETRY:
The blood cell pellet was diluted with 2 volumes of PBS. The equal erythrocyte lysate was added and mixed for 5 min to completely disrupt the erythrocytes. Samples were diluted 1-fold with 5 ml of PBS and centrifuged at 1200 rpm for 5 min. After removing the supernatant, the pellet was washed twice. We used 5 ml of FACS buffer to resuspend the cells, which were then centrifuged at 1200 rpm for 5 minutes. After removing the supernatant, the remainder was used to resuspend the cells. The isolated cells received the centrifugation, followed by discarding the supernatant. We added 200 μL of surface antibody staining solution (CD4-FITC (1: 200) and CD25-APC (1: 200); CD3-FITC (1: 200), CD8-APC (1: 200) and CD45.1-PerCP (1: 200)) was added to resuspend the cells. After mixing, the cells were incubated in the dark at 4°C for 30 min, and washed with FACS buffer. We used 200 μL of fixative permeabilization working solutions for resuspending the cells, and they were then left at room temperature for 1 h. After completion, 1x perm wash buffer was used to wash the cells. The configured intracellular staining working solution included 200 μL Foxp3-PE (diluted antibody working solution with 1x perm wash buffer, 1: 100) and incubated with the cells at 4°C for 30 min in the dark. After washing with 1x perm wash buffer, FACS buffer was added to resuspend cells and stored in the dark. The percentages of CD3+, CD8+, CD45+, CD8+CD45+, CD4+, Foxp3+, CD25+, and Foxp3+CD25+ cells were detected utilizing flow cytometry.
STATISTICAL ANALYSIS:
All values are displayed as means±SEM. ANOVA and Tukey’s post hoc tests were applied to estimate differences among groups. P<0.05 was considered statistically significant.
Results
ESTABLISHMENT OF A CAWS-INDUCED MURINE MODEL OF KD WITH CARDIAC DYSFUNCTION:
This study established a CAWS-induced murine model of KD. At 4 weeks following rhG-CSF administration, our echocardiography displayed that CAWS-induced mice had a remarkable reduction in fraction shortening (Figure 1A) and an increase in left ventricle end-diastolic diameter (LVEDD; Figure 1A). This demonstrated that CAWS decreased cardiac function and elevated cardiac dimension of mice. Nevertheless, the cardiac function and dimension were remarkably maintained following rhG-CSF administration. Moreover, CAWS-induced mice had significantly elevated heart weight/body weight ratio compared with control mice, but the ratio was markedly alleviated by rhG-CSF treatment (Figure 1A).
THE CAWS-INDUCED MURINE MODEL WITH CARDIAC AND VASCULAR INFLAMMATION:
H&E staining was implemented to evaluate the pathological conditions of hearts and arteries. In the control group, the cardiomyocytes of the heart tissue were closely arranged, with normal morphology and without obvious lesions (Figure 1B). In the CAWS-induced model group, the heart tissue was seriously damaged, some myocardial cells were coagulated and necrotic, the cytoplasm was stained with eosin (black arrow), and there was focal inflammatory cell infiltration in local areas. In the rhG-CSF treatment group, the structure and morphology of the heart were basically normal, and the myocardial cells were arranged in a roughly orderly manner, but the blood vessels in some areas were congested with fibrotic lesions. As shown in Figure 1C, for control mice, the coronary intima was smooth and intact, and the endothelial cells were neatly arranged (Figure 1C). In the CAWS-induced model group, the vessel wall was partially thickened, the intima had obvious edema, thickening, and degeneration, the endothelial cells were arranged disorderly, and small vacuoles were found in the cytoplasm, surrounded by inflammatory cell infiltration (black arrow). In the rhG-CSF administration group, the thickness of the vessel wall was basically restored, the endothelial thickening and edema were significantly reduced, and it was basically restored to smoothness, and the endothelial cells were densely arranged. Above data demonstrated the CAWS-induced cardiac and vascular inflammation.
THE CAWS-INDUCED MURINE MODEL OF KD WITH THE ACTIVATION OF PRO-INFLAMMATORY CYTOKINES AND THE INACTIVATION OF ANTI-INFLAMMATORY CYTOKINES:
Through ELISA and RT-qPCR assays, we examined the serum levels of pro- and anti-inflammatory cytokines. In comparison to control mice, CAWS-induced mice presented remarkably elevated IL-1β, IL-6, and TNF-α levels (Figure 2A, 2B). Nevertheless, rhG-CSF administration significantly reduced the release of above inflammatory cytokines in CAWS-triggered murine models of KD. IL-10, and G-CSF levels were markedly reduced in CAWS-induced mice than in control mice, which were elevated after administration of rhG-CSF (Figure 2A, 2B). Hence, the CAWS-induced murine model of KD presented the activation of pro-inflammatory cytokines and the inactivation of anti-inflammatory cytokines.
THE CAWS-INDUCED MURINE MODEL OF KD WITH THE ACTIVATION OF CHEMOKINES AND ADHESION MOLECULES:
We further examined the serum levels of chemokines and adhesion molecules in CAWS-induced murine models of KD. The data demonstrated that CAWS-induced mice displayed a reduction in chemokine MIP-1α and an increase in chemokine RANTES (Figure 3A, 3B). However, their serum levels in CAWS-induced murine models of KD were remarkably restored by rhG-CSF administration. We also found that the serum levels of adhesion molecules (MCP-1, VCAM-1, ICAM-1, and E-selectin) were remarkably elevated in CAWS-induced models (Figure 3A, 3B). Their serum levels of CAWS-induced mice were significantly decreased by rhG-CSF administration.
THE CAWS-INDUCED MURINE MODEL OF KD WITH REDUCED CD4+, CD8+, AND FOXP3+ T LYMPHOCYTE SUBPOPULATIONS IN CARDIAC TISSUES:
Through immunofluorescence, we evaluated CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues of CAWS-induced murine models. In comparison to the control mice, the levels of CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations were markedly decreased in cardiac tissues of CAWS-induced mice (Figure 4A–4C). Administration of rhG-CSF significantly enhanced the levels of CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues of CAWS-induced mice.
THE CAWS-INDUCED MURINE MODEL OF KD WITH REDUCED F4/80+ MACROPHAGES AND CD11B+ MONOCYTES AND INCREASED LY-6G+ NEUTROPHILS IN CARDIAC TISSUES:
Further analysis was conducted for examining F4/80, CD11b, and Ly-6G expressions in cardiac tissues of CAWS-induced murine models. As shown in Figure 5A, 5B, CAWS-induced mice showed reduced F4/80+ macrophages and CD11b+ monocytes in cardiac tissues in comparison to control mice. However, rhG-CSF administration enhanced the levels of F4/80+ macrophages and CD11b+ monocytes in cardiac tissues of CAWS-induced mice. Compared with control mice, higher Ly-6G+ neutrophil levels were found in cardiac tissues of CAWS-induced mice (Figure 5C). Nevertheless, Ly-6G+ neutrophil levels in cardiac tissues were decreased following rhG-CSF administration.
THE CAWS-INDUCED MURINE MODEL OF KD WITH DECREASED CD3+, CD8+, AND CD8+CD45+ T LYMPHOCYTE SUBPOPULATIONS AND INCREASED CD45+ T LYMPHOCYTE SUBPOPULATION:
Through flow cytometry, we examined the levels of CD3+, CD8+, CD45+, and CD8+CD45+ T lymphocyte subpopulations in serum of CAWS-induced murine models. In comparison to control mice, CAWS-induced mice displayed a reduction in CD3+, CD8+, and CD8+CD45+ T lymphocyte subpopulations as well as an increase in CD45+ T lymphocyte subpopulation (Figure 6A, 6B). Nevertheless, rhG-CSF administration markedly elevated the levels of CD3+, CD8+, and CD8+CD45+ T lymphocyte subpopulations and decreased the levels of CD45+ T lymphocyte subpopulation in serum of CAWS-induced mice.
THE CAWS-INDUCED MURINE MODEL OF KD WITH DECREASED CD4+, FOXP3+, CD25+, AND FOXP3+CD25+ T LYMPHOCYTE SUBPOPULATIONS:
Our further analysis showed that the levels of CD4+, Foxp3+, CD25+, and Foxp3+CD25+ T lymphocyte subpopulations were all remarkably reduced in serum of CAWS-induced murine models (Figure 7A, 7B). However, rhG-CSF administration significantly elevated the levels of CD4+, Foxp3+, CD25+, and Foxp3+CD25+ T lymphocyte subpopulations in serum of CAWS-induced mice.
Discussion
KD is a childhood systemic febrile syndrome characterized by coronary arteritis [18–20]. Although the exact cause of KD remains to be determined, epidemiological and experimental studies have shown that inflammatory factors are associated with KD pathogenesis [21–23]. Moreover, innate immune system participates in KD vasculitis development, in accordance with the findings from animal models and clinical and laboratory data of KD patients [24]. Evidence suggests that innate immune pathogen-associated molecular patterns are capable of causing vasculitis independently of acquired immunity [24]. Histologically, remarkable infiltration of macrophages and neutrophils within the arteries were found in the coronary arteries of patients with acute-phase KD [25]. Nevertheless, the limited understanding of KD-related pathogens and pathogenic cells, coupled with the unclear molecular immune mechanisms involved in the pathogenesis of KD, hinder the development of more effective therapeutic strategies against KD. Moreover, tissue specimens from patients with KD are difficult to obtain [26–28]. Herein, we established a murine model of KD induced by CAWS and revealed the characteristics of inflammatory factors and T cell subpopulations during KD.
IVIG administration reduces the occurrence and severity of coronary vasculitis, which has been often used in clinical treatment of KD. However, some children are resistant to IVIG and are prone to coronary artery damage, which has a negative impact on the prognosis of children [29]. Thus, it is important to develop more effective therapeutic strategies for KD. A previous study demonstrated that rhG-CSF treatment could remarkably increase the number of circulating endothelial progenitor cells and plasma concentration of NO and effectively improved the elastin breakdown in KD mice [30], indicating that rhG-CSF may represent a novel strategy for KD therapy. However, the protective efficacy of rhG-CSF is a key issue for future advanced experimental and clinical studies. C57BL/6J mice received 0.2 mL CAWS (0.4 mg/mouse/day) through intraperitoneal injection for 5 consecutive days to induce KD vasculitis. On the 5th day after intraperitoneal injection of CAWS, the mice were subcutaneously injected with rhG-CSF (50 μg/kg/day) for 5 consecutive days. After 4 weeks, we noticed that CAWS-induced mice displayed a remarkable cardiac dysfunction (reduced fraction shortening, increased LVEDD and heart weight/body weight ratio), as well as cardiac and vascular inflammation, which were alleviated by rhG-CSF treatment. Previous evidence demonstrated that CAWS can induce inflammatory cells to recruit into the aortic root area [31]. Moreover, we found that CAWS-induced mice had increased expression of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α), chemokine (RANTES), and adhesion molecules (MCP-1, VCAM-1, ICAM-1, E-selectin), and decreased expression of anti-inflammatory cytokines (IL-10, G-CSF), indicative of the activation of inflammation induced by CAWS. Previously, patients with KD were found to have higher serum IL-1β levels compared with healthy controls [32]. Another study demonstrated the elevated release of IL-6, IL-10, and MCP-1 in KD patients [33]. In a CAWS-induced KD murine model, GM-CSF is required for cardiac inflammatory response, which is an underlying target for therapeutic intervention in KD [34]. In clinical trials, TNF-α blockers were capable of alleviating KD in children [35]. In FK565-induced KD vasculitis, increased RANTES levels were investigated in plasma specimens [36].
Our immunofluorescence and flow cytometry demonstrated that the proportions of CD3+, CD4+, CD8+, CD8+CD45+, Foxp3+, CD25+, and Foxp3+CD25+ T lymphocyte subpopulations, F4/80+ macrophages, and CD11b+ monocytes were decreased, and the proportion of CD45+ T lymphocyte subpopulation and Ly-6G+ neutrophils were increased in cardiac tissues of CAWS-induced mice, which were ameliorated by rhG-CSF administration. Single-cell RNA sequencing (scRNA-seq) analysis of peripheral blood shows that the percentage of CD8+ T cells is reduced in acute KD, especially effector memory CD8+ T cells, in comparison to healthy controls [37]. In KD patients, the proportions of Treg cells and activated CD25+ cells were elevated following IVIG administration [38]. In comparison to healthy controls, scRNA-seq analysis demonstrated that naive CD8+ T cells, T helper cells, and B cells in KD children show reduced infiltration, especially T cells [11]. The proportions of CD25 + CD4 + regulatory T cells in subjects with acute KD are markedly reduced in comparison to healthy controls [39]. The proportion is restored to the normal level following IVIG administration.
Accordingly, our study established a CAWS-induced murine KD vasculitis model to further examine the distribution and role of T cell subpopulations and relevant molecules in the development of KD, which is of great significance to further clarify the etiology, pathogenesis, and treatment regimens of KD. Results from studies that model CAWS-induced KD and target T cell subpopulations of KD may lead to evidence for new treatments for KD.
Conclusions
Collectively, our findings demonstrated the characteristics of inflammatory factors and T cell subpopulations in a murine model of KD induced by CAWS. Moreover, rhG-CSF can alleviate inflammatory response and T cell activation, which might be an effective therapeutic regimen against KD.
Figures
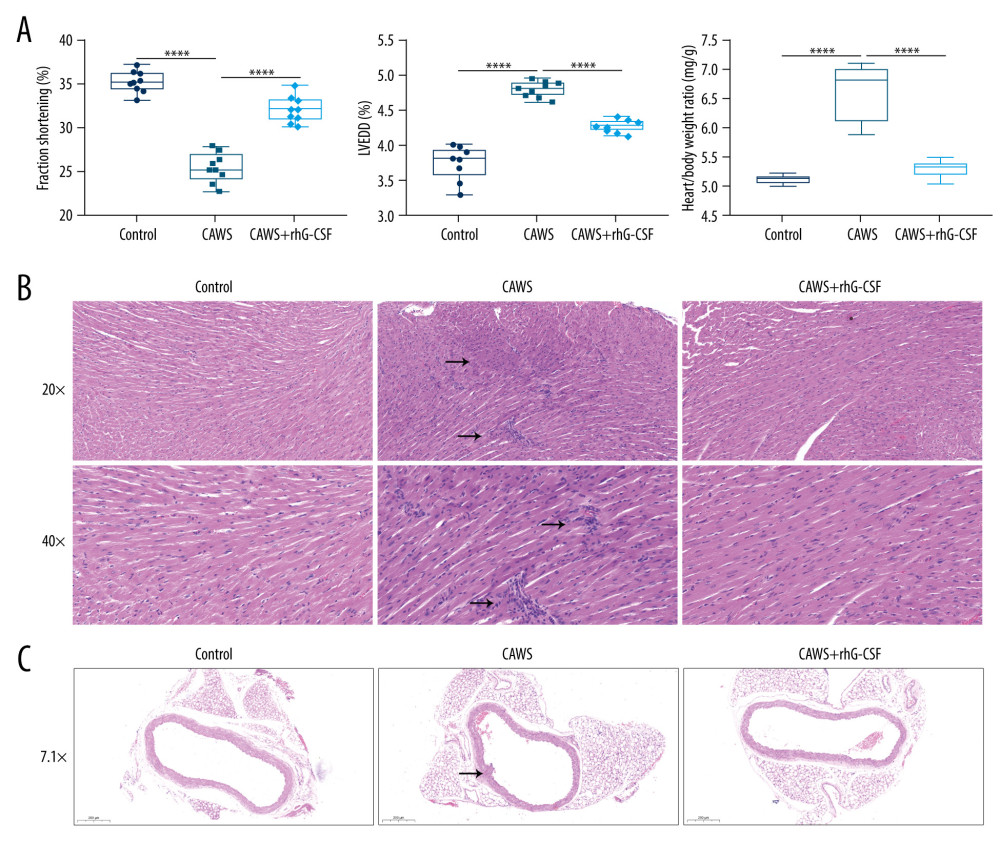 Figure 1. The CAWS-induced murine model of KD with cardiac dysfunction, cardiac and vascular inflammation. (A) Fraction shortening, left ventricle end-diastolic diameter (LVEDD) and heart weight/body weight ratio were examined at 4 weeks after rhG-CSF treatment in CAWS-induced mice. **** p<0.0001. (B, C) The sections of hearts and arteries were stained at 4 weeks following rhG-CSF administration in CAWS-induced mice. Black arrow points to the indicated lesion location. Bar=50, 20 or 200 μm. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 1. The CAWS-induced murine model of KD with cardiac dysfunction, cardiac and vascular inflammation. (A) Fraction shortening, left ventricle end-diastolic diameter (LVEDD) and heart weight/body weight ratio were examined at 4 weeks after rhG-CSF treatment in CAWS-induced mice. **** p<0.0001. (B, C) The sections of hearts and arteries were stained at 4 weeks following rhG-CSF administration in CAWS-induced mice. Black arrow points to the indicated lesion location. Bar=50, 20 or 200 μm. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. 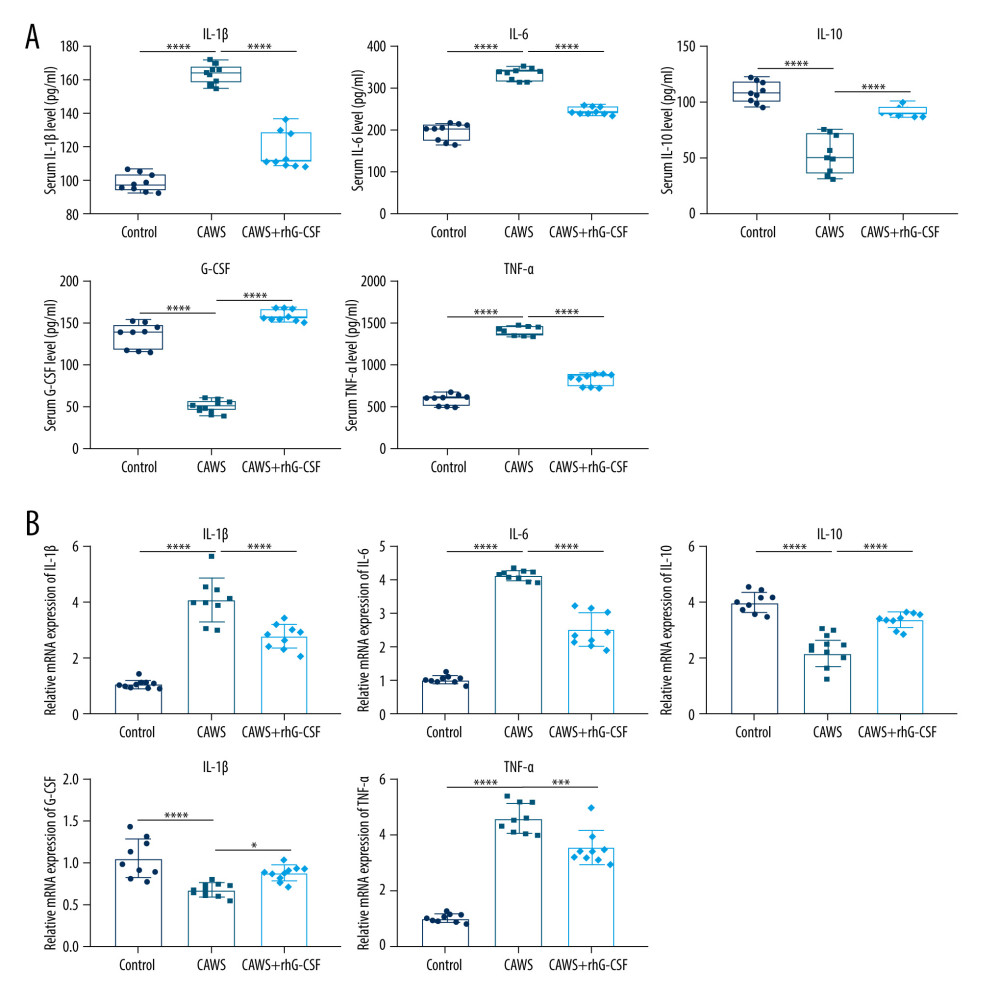 Figure 2. The CAWS-induced murine model of KD with the activation of pro-inflammatory cytokines and the inactivation of anti-inflammatory cytokines. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of inflammatory cytokines IL-1β, IL-6, IL-10, G-CSF and TNF-α at 4 weeks following rhG-CSF administration in CAWS-induced mice. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 2. The CAWS-induced murine model of KD with the activation of pro-inflammatory cytokines and the inactivation of anti-inflammatory cytokines. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of inflammatory cytokines IL-1β, IL-6, IL-10, G-CSF and TNF-α at 4 weeks following rhG-CSF administration in CAWS-induced mice. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. 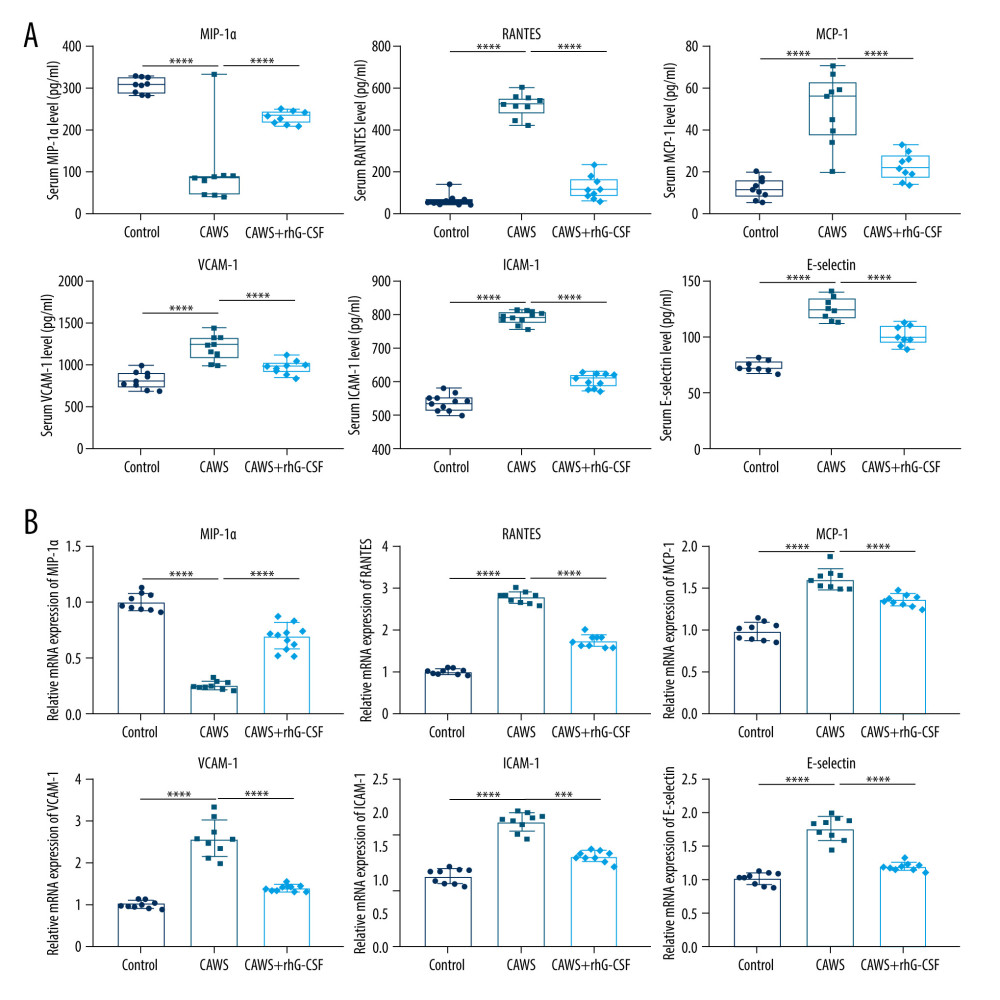 Figure 3. The CAWS-triggered murine model of KD with the activation of chemokines and adhesion molecules. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of MIP-1α, RANTES and MCP-1 at 4 weeks after rhG-CSF administration in CAWS-induced mice. VCAM-1, ICAM-1 and E-selectin expressions were examined in serum specimens from CAWS-induced mice at 4 weeks after rhG-CSF administration. *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 3. The CAWS-triggered murine model of KD with the activation of chemokines and adhesion molecules. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of MIP-1α, RANTES and MCP-1 at 4 weeks after rhG-CSF administration in CAWS-induced mice. VCAM-1, ICAM-1 and E-selectin expressions were examined in serum specimens from CAWS-induced mice at 4 weeks after rhG-CSF administration. *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. 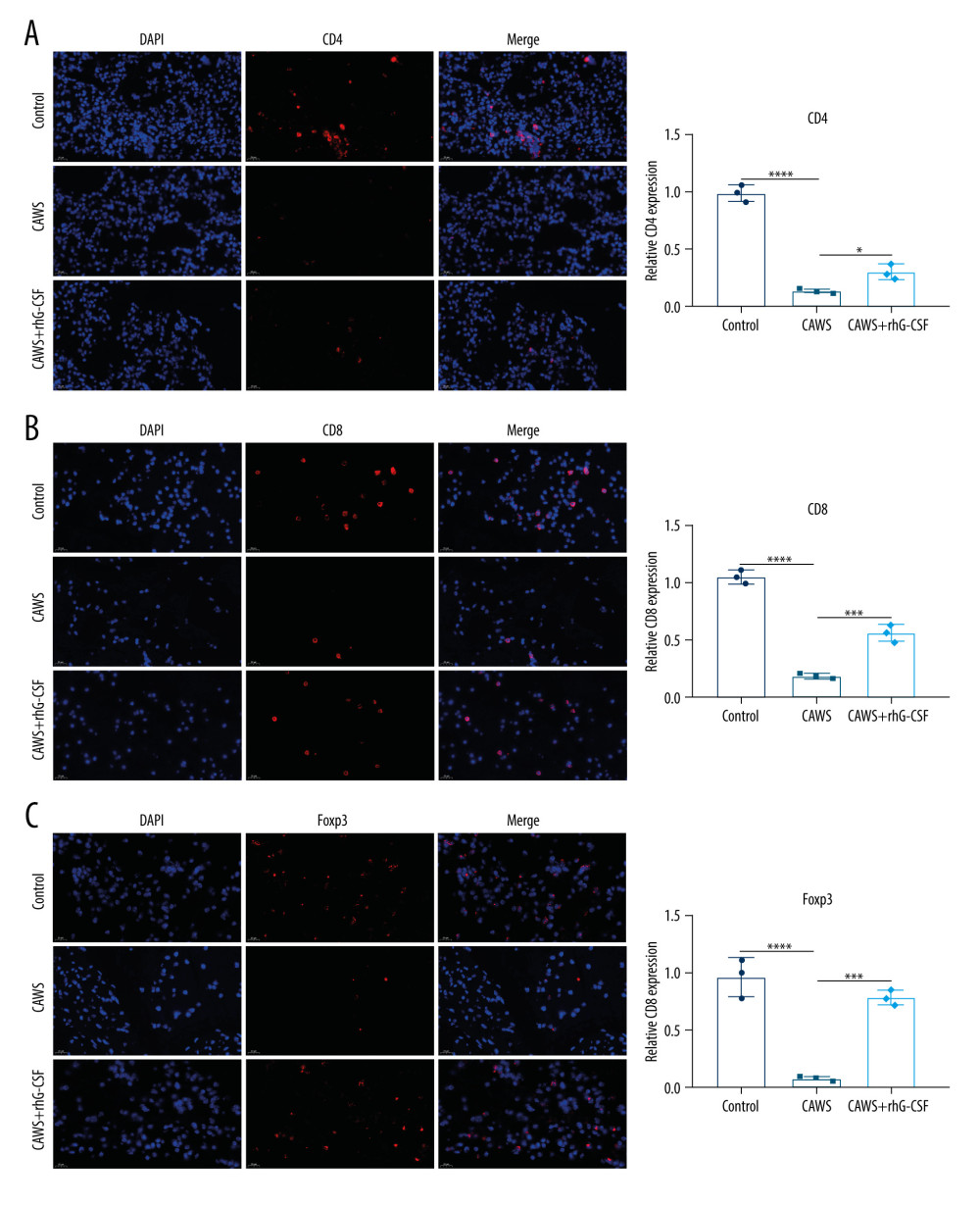 Figure 4. The CAWS-induced KD mice with reduced CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues. (A–C) Cardiac tissues were stained by (A) CD4+, (B) CD8+, and (C) Foxp3+ T lymphocyte subpopulations at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 4. The CAWS-induced KD mice with reduced CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues. (A–C) Cardiac tissues were stained by (A) CD4+, (B) CD8+, and (C) Foxp3+ T lymphocyte subpopulations at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. 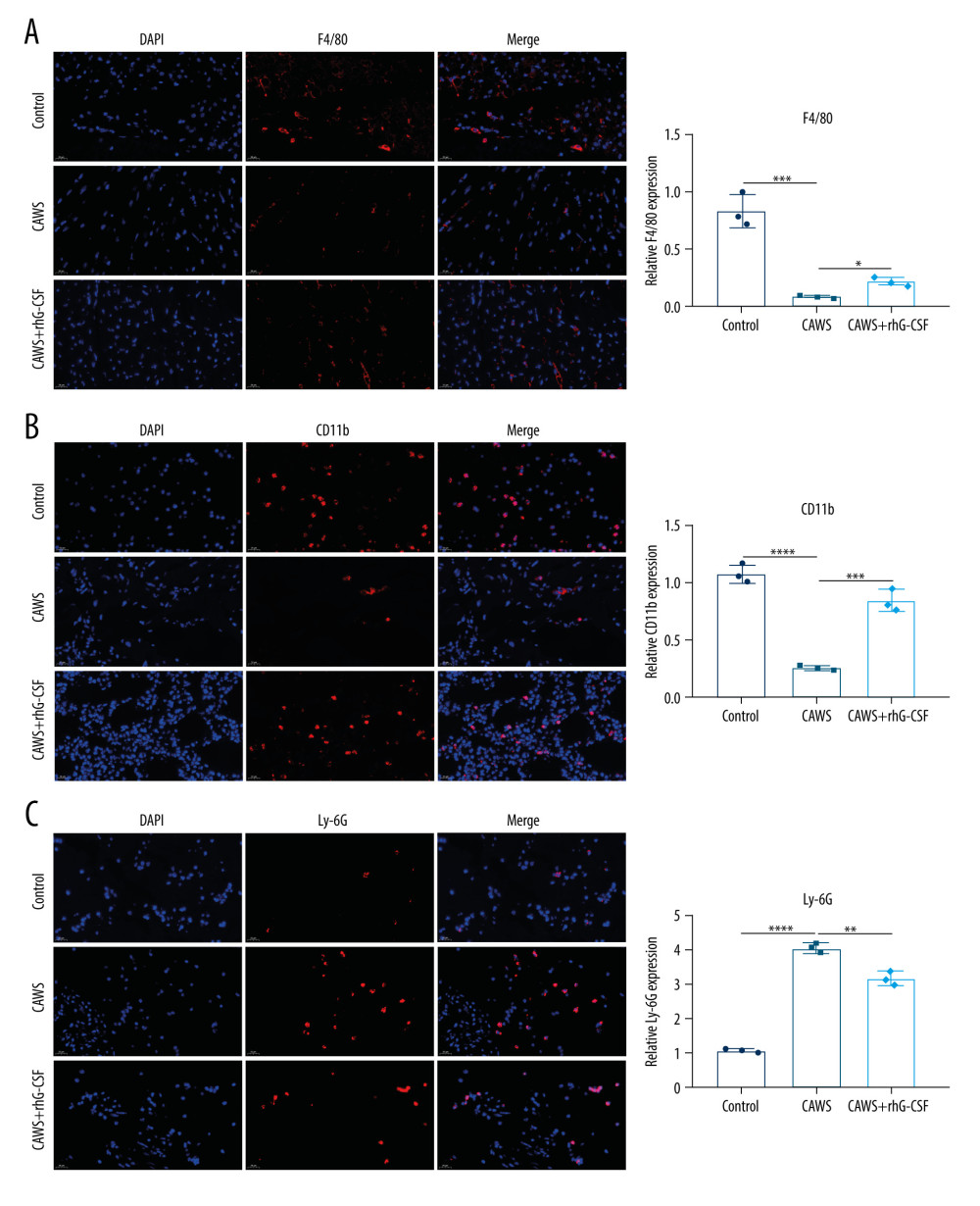 Figure 5. The CAWS-induced murine model of KD with reduced F4/80+ macrophages, CD11b+ monocytes and increased Ly-6G+ neutrophils in cardiac tissues. (A–C) Cardiac tissues were stained by (A) F4/80+ macrophages, (B) CD11b+ monocytes, and (C) Ly-6G+ neutrophils at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; ** p<0.01; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 5. The CAWS-induced murine model of KD with reduced F4/80+ macrophages, CD11b+ monocytes and increased Ly-6G+ neutrophils in cardiac tissues. (A–C) Cardiac tissues were stained by (A) F4/80+ macrophages, (B) CD11b+ monocytes, and (C) Ly-6G+ neutrophils at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; ** p<0.01; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. 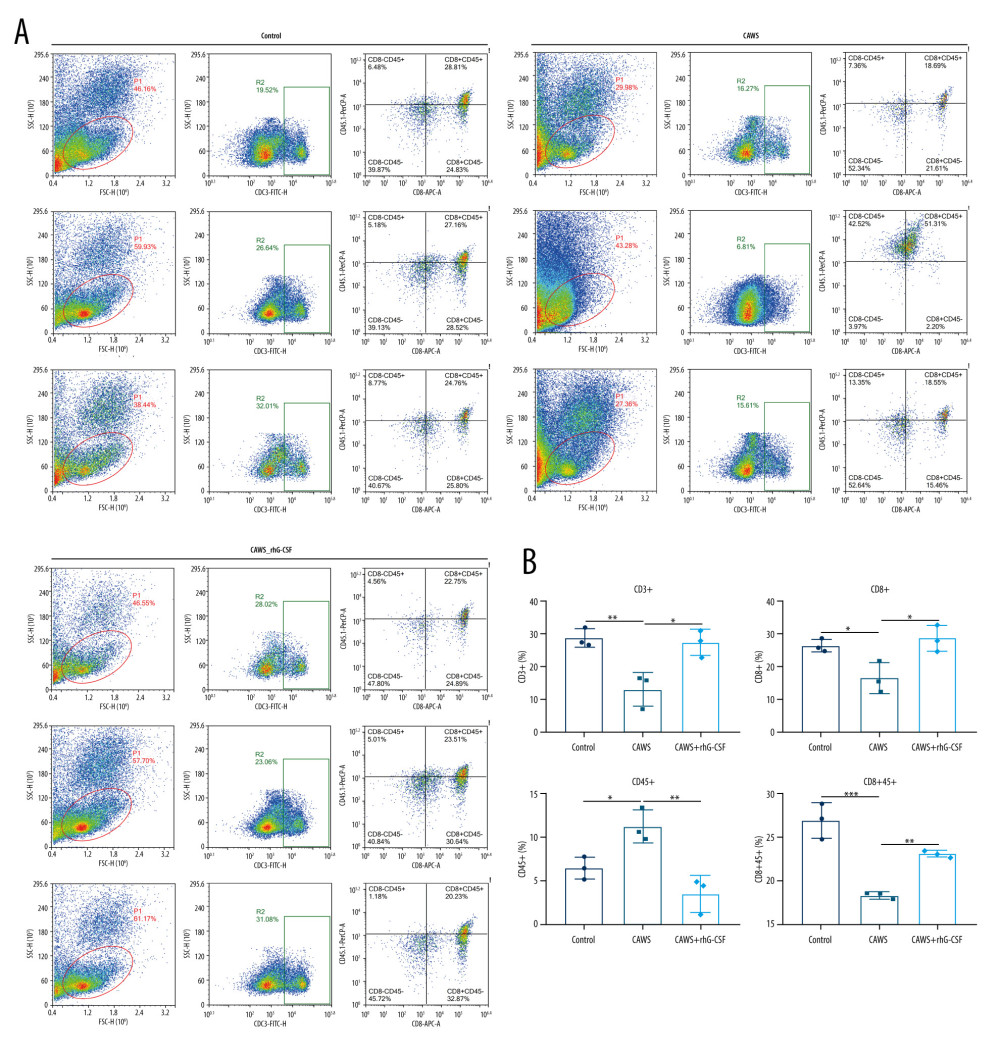 Figure 6. The CAWS-induced murine model of KD with decreased CD3+, CD8+ and CD8+CD45+ T lymphocyte subpopulations and increased CD45+ T lymphocyte subpopulation. (A, B) The levels of CD3+, CD8+, CD45+ and CD8+CD45+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 6. The CAWS-induced murine model of KD with decreased CD3+, CD8+ and CD8+CD45+ T lymphocyte subpopulations and increased CD45+ T lymphocyte subpopulation. (A, B) The levels of CD3+, CD8+, CD45+ and CD8+CD45+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. 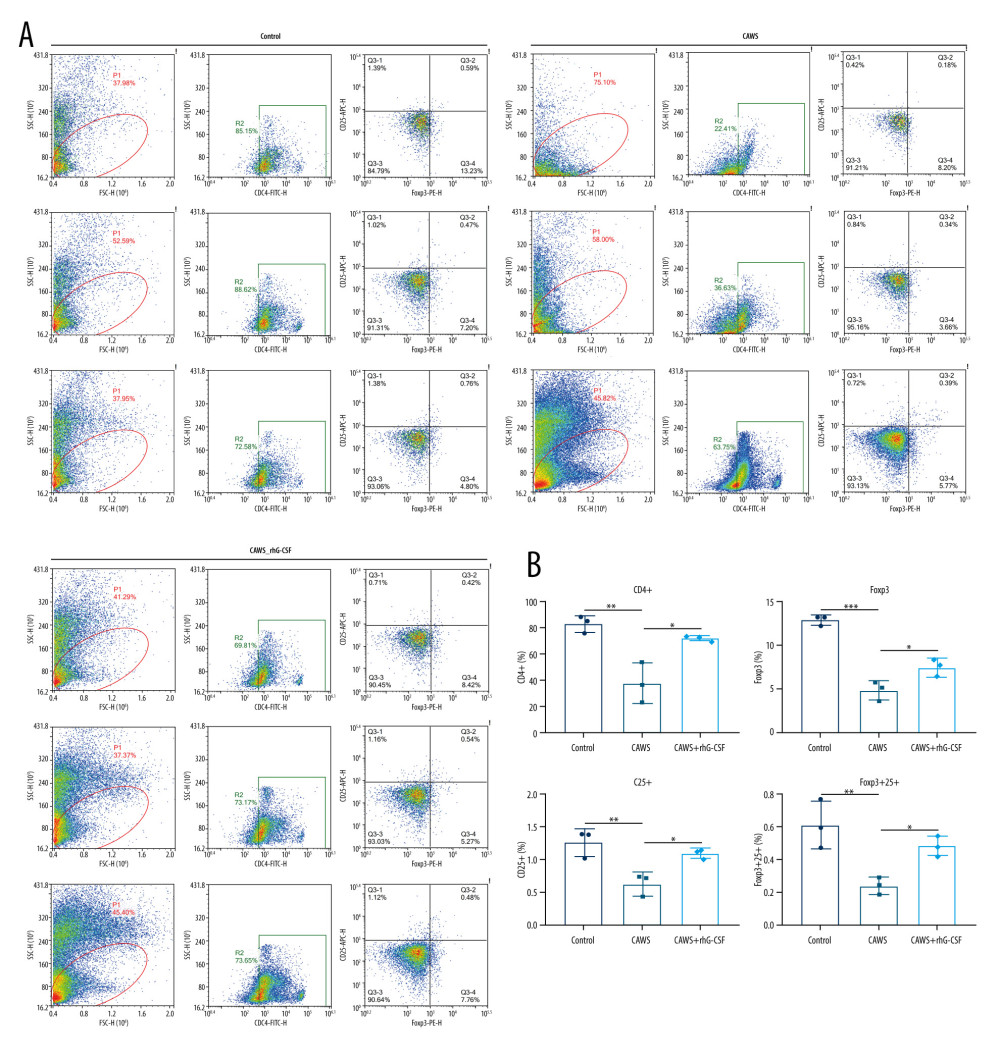 Figure 7. The CAWS-induced murine model of KD with decreased CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations. (A, B) The levels of CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 7. The CAWS-induced murine model of KD with decreased CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations. (A, B) The levels of CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. References
1. Lun Y, Borjini N, Miura NN, CDCP1 on dendritic cells contributes to the development of a model of Kawasaki disease: J Immunol, 2021; 206; 2819-27
2. Noval Rivas M, Arditi M, Kawasaki disease: Pathophysiology and insights from mouse models: Nat Rev Rheumatol, 2020; 16; 391-405
3. Jia C, Zhuge Y, Zhang S, IL-37b alleviates endothelial cell apoptosis and inflammation in Kawasaki disease through IL-1R8 pathway: Cell Death Dis, 2021; 12; 575
4. Koné-Paut I, Tellier S, Belot A, Phase II open label study of anakinra in intravenous immunoglobulin-resistant Kawasaki disease: Arthritis Rheumatol, 2021; 73; 151-61
5. Liu E, Gonzalez J, Siu A, Use of premedication with intravenous immune globulin in Kawasaki disease: A retrospective review: Pediatr Allergy Immunol, 2021; 32; 750-55
6. Zhu YP, Shamie I, Lee JC, Immune response to intravenous immunoglobulin in patients with Kawasaki disease and MIS-C: J Clin Invest, 2021; 131; e147076
7. Anzai F, Watanabe S, Kimura H, Crucial role of NLRP3 inflammasome in a murine model of Kawasaki disease: J Mol Cell Cardiol, 2020; 138; 185-96
8. Nakamura J, Watanabe S, Kimura H, Adeno-associated virus vector-mediated interleukin-10 induction prevents vascular inflammation in a murine model of Kawasaki disease: Sci Rep, 2018; 8; 7601
9. Yanai C, Tanaka H, Miura NN: Biol Pharm Bull, 2020; 43; 848-58
10. Ishibashi K, Fukazawa R, Miura NN, Diagnostic potential of antibody titres against Candida cell wall β-glucan in Kawasaki disease: Clin Exp Immunol, 2014; 177; 161-67
11. Fan X, Zhou Y, Guo X, Xu M, Utilizing single-cell RNA sequencing for analyzing the characteristics of PBMC in patients with Kawasaki disease: BMC Pediatr, 2021; 21; 277
12. Yilmaz A, Rowley A, Schulte DJ, Activated myeloid dendritic cells accumulate and co-localize with CD3+ T cells in coronary artery lesions in patients with Kawasaki disease: Exp Mol Pathol, 2007; 83; 93-103
13. Jia S, Li C, Wang G, Yang J, Zu Y, The T helper type 17/regulatory T cell imbalance in patients with acute Kawasaki disease: Clin Exp Immunol, 2010; 162; 131-37
14. Sun M, Xing H, Interleukin-35 regulates peripheral T cell activity in patients with Kawasaki disease: Int Immunopharmacol, 2021; 96; 107642
15. Noval Rivas M, Lee Y, Wakita D: Arthritis Rheumatol, 2017; 69; 410-21
16. Ye Q, Gong FQ, Shang SQ, Hu J, Intravenous immunoglobulin treatment responsiveness depends on the degree of CD8+ T cell activation in Kawasaki disease: Clin Immunol, 2016; 171; 25-31
17. Ohashi R, Fukazawa R, Watanabe M, Etanercept suppresses arteritis in a murine model of kawasaki disease: A comparative study involving different biological agents: Int J Vasc Med, 2013; 2013; 543141
18. Chen DT, Chang JP, Cheng SW, Kawasaki disease in childhood and psychiatric disorders: A population-based case-control prospective study in Taiwan: Brain Behav Immun, 2022; 100; 105-11
19. Hsieh LE, Grifoni A, Sidney J, Characterization of SARS-CoV-2 and common cold coronavirus-specific T-cell responses in MIS-C and Kawasaki disease children: Eur J Immunol, 2022; 52; 123-37
20. Marzano AV, Cassano N, Moltrasio C, Multisystem inflammatory syndrome in children associated with COVID-19: A review with an emphasis on mucocutaneous and Kawasaki disease-like findings: Dermatology, 2022; 238; 35-43
21. Cavalli G, Colafrancesco S, Emmi G, Interleukin 1α: A comprehensive review on the role of IL-1α in the pathogenesis and treatment of autoimmune and inflammatory diseases: Autoimmun Rev, 2021; 20; 102763
22. Porritt RA, Markman JL, Maruyama D, Interleukin-1 beta-mediated sex differences in Kawasaki disease vasculitis development and response to treatment: Arterioscler Thromb Vasc Biol, 2020; 40; 802-18
23. Weng KP, Hsieh KS, Huang SH, Interleukin-18 and coronary artery lesions in patients with Kawasaki disease: J Chin Med Assoc, 2013; 76; 438-45
24. Hara T, Nakashima Y, Sakai Y, Nishio H, Kawasaki disease: A matter of innate immunity: Clin Exp Immunol, 2016; 186; 134-43
25. Kobayashi M, Matsumoto Y, Ohya M, Histologic and immunohistochemical evaluation of infiltrating inflammatory cells in Kawasaki disease arteritis lesions: Appl Immunohistochem Mol Morphol, 2021; 29; 62-67
26. Alsaied T, Tremoulet AH, Burns JC, Review of cardiac involvement in multisystem inflammatory syndrome in children: Circulation, 2021; 143; 78-88
27. Nakashima Y, Sakai Y, Mizuno Y, Lipidomics links oxidized phosphatidylcholines and coronary arteritis in Kawasaki disease: Cardiovasc Res, 2021; 117; 96-108
28. Nielsen TM, Andersen NH, Torp-Pedersen C, Kawasaki disease, autoimmune disorders, and cancer: A register-based study: Eur J Pediatr, 2021; 180; 717-23
29. Zhang RL, Lo HH, Lei C, Current pharmacological intervention and development of targeting IVIG resistance in Kawasaki disease: Curr Opin Pharmacol, 2020; 54; 72-81
30. Liu J, Chen Z, Du Z, Lu D, Granulocyte colony-stimulating factor ameliorates coronary artery elastin breakdown in a mouse model of Kawasaki disease: Chin Med J (Engl), 2014; 127; 3712-17
31. Miyabe C, Miyabe Y, Bricio-Moreno L, Dectin-2-induced CCL2 production in tissue-resident macrophages ignites cardiac arteritis: J Clin Invest, 2019; 129; 3610-24
32. Inoue T, Murakami S, Matsumoto K, Matsuda A, Functional benefits of corticosteroid and IVIG combination therapy in a coronary artery endothelial cell model of Kawasaki disease: Pediatr Rheumatol Online J, 2020; 18; 76
33. Su Y, Feng S, Luo L, Liu R, Yi Q, Association between IL-35 and coronary arterial lesions in children with Kawasaki disease: Clin Exp Med, 2019; 19; 87-92
34. Stock AT, Hansen JA, Sleeman MA, GM-CSF primes cardiac inflammation in a mouse model of Kawasaki disease: J Exp Med, 2016; 213; 1983-98
35. Yamaji N, da Silva Lopes K, Shoda T, TNF-α blockers for the treatment of Kawasaki disease in children: Cochrane Database Syst Rev, 2019; 8; CD012448
36. Ohashi R, Fukazawa R, Watanabe M, Characterization of a murine model with arteritis induced by Nod1 ligand, FK565: A comparative study with a CAWS-induced model: Mod Rheumatol, 2017; 27; 1024-30
37. Wang Z, Xie L, Ding G, Single-cell RNA sequencing of peripheral blood mononuclear cells from acute Kawasaki disease patients: Nat Commun, 2021; 12; 5444
38. McAlpine SM, Roberts SE, Heath JJ, High dose intravenous IgG therapy modulates multiple NK cell and T cell functions in patients with immune dysregulation: Front Immunol, 2021; 12; 660506
39. Furuno K, Yuge T, Kusuhara K, CD25+CD4+ regulatory T cells in patients with Kawasaki disease: J Pediatr, 2004; 145; 385-90
Figures
 Figure 1. The CAWS-induced murine model of KD with cardiac dysfunction, cardiac and vascular inflammation. (A) Fraction shortening, left ventricle end-diastolic diameter (LVEDD) and heart weight/body weight ratio were examined at 4 weeks after rhG-CSF treatment in CAWS-induced mice. **** p<0.0001. (B, C) The sections of hearts and arteries were stained at 4 weeks following rhG-CSF administration in CAWS-induced mice. Black arrow points to the indicated lesion location. Bar=50, 20 or 200 μm. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 1. The CAWS-induced murine model of KD with cardiac dysfunction, cardiac and vascular inflammation. (A) Fraction shortening, left ventricle end-diastolic diameter (LVEDD) and heart weight/body weight ratio were examined at 4 weeks after rhG-CSF treatment in CAWS-induced mice. **** p<0.0001. (B, C) The sections of hearts and arteries were stained at 4 weeks following rhG-CSF administration in CAWS-induced mice. Black arrow points to the indicated lesion location. Bar=50, 20 or 200 μm. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. Figure 2. The CAWS-induced murine model of KD with the activation of pro-inflammatory cytokines and the inactivation of anti-inflammatory cytokines. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of inflammatory cytokines IL-1β, IL-6, IL-10, G-CSF and TNF-α at 4 weeks following rhG-CSF administration in CAWS-induced mice. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 2. The CAWS-induced murine model of KD with the activation of pro-inflammatory cytokines and the inactivation of anti-inflammatory cytokines. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of inflammatory cytokines IL-1β, IL-6, IL-10, G-CSF and TNF-α at 4 weeks following rhG-CSF administration in CAWS-induced mice. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. Figure 3. The CAWS-triggered murine model of KD with the activation of chemokines and adhesion molecules. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of MIP-1α, RANTES and MCP-1 at 4 weeks after rhG-CSF administration in CAWS-induced mice. VCAM-1, ICAM-1 and E-selectin expressions were examined in serum specimens from CAWS-induced mice at 4 weeks after rhG-CSF administration. *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 3. The CAWS-triggered murine model of KD with the activation of chemokines and adhesion molecules. (A) ELISA and (B) RT-qPCR were utilized for quantifying the serum levels of MIP-1α, RANTES and MCP-1 at 4 weeks after rhG-CSF administration in CAWS-induced mice. VCAM-1, ICAM-1 and E-selectin expressions were examined in serum specimens from CAWS-induced mice at 4 weeks after rhG-CSF administration. *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. Figure 4. The CAWS-induced KD mice with reduced CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues. (A–C) Cardiac tissues were stained by (A) CD4+, (B) CD8+, and (C) Foxp3+ T lymphocyte subpopulations at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 4. The CAWS-induced KD mice with reduced CD4+, CD8+, and Foxp3+ T lymphocyte subpopulations in cardiac tissues. (A–C) Cardiac tissues were stained by (A) CD4+, (B) CD8+, and (C) Foxp3+ T lymphocyte subpopulations at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. Figure 5. The CAWS-induced murine model of KD with reduced F4/80+ macrophages, CD11b+ monocytes and increased Ly-6G+ neutrophils in cardiac tissues. (A–C) Cardiac tissues were stained by (A) F4/80+ macrophages, (B) CD11b+ monocytes, and (C) Ly-6G+ neutrophils at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; ** p<0.01; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures.
Figure 5. The CAWS-induced murine model of KD with reduced F4/80+ macrophages, CD11b+ monocytes and increased Ly-6G+ neutrophils in cardiac tissues. (A–C) Cardiac tissues were stained by (A) F4/80+ macrophages, (B) CD11b+ monocytes, and (C) Ly-6G+ neutrophils at 4 weeks after rhG-CSF administration in CAWS-induced mice. Bar=20 μm. * p<0.05; ** p<0.01; *** p<0.001; and **** p<0.0001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) and Olympus software (version 2.2; Olympus, Japan) were used to create the pictures. Figure 6. The CAWS-induced murine model of KD with decreased CD3+, CD8+ and CD8+CD45+ T lymphocyte subpopulations and increased CD45+ T lymphocyte subpopulation. (A, B) The levels of CD3+, CD8+, CD45+ and CD8+CD45+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 6. The CAWS-induced murine model of KD with decreased CD3+, CD8+ and CD8+CD45+ T lymphocyte subpopulations and increased CD45+ T lymphocyte subpopulation. (A, B) The levels of CD3+, CD8+, CD45+ and CD8+CD45+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. Figure 7. The CAWS-induced murine model of KD with decreased CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations. (A, B) The levels of CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures.
Figure 7. The CAWS-induced murine model of KD with decreased CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations. (A, B) The levels of CD4+, Foxp3+, CD25+ and Foxp3+CD25+ T lymphocyte subpopulations were examined in serum of CAWS-induced mice at 4 weeks after rhG-CSF administration. * p<0.05; ** p<0.01; *** p<0.001. GraphPad Prism software (version 8.0.1; Graph Pad; San Diego, CA, USA) was used to create the pictures. In Press
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952