18 March 2022: Clinical Research
Unveiling the Differences in Biological Properties of Dental Pulp Stem Cells from Normal and Inflamed Pulp: A Comprehensive Comparative Study
Shao-Chen Nie12ABCDEF, Kun Yang3ABCD, Na-Na Luan12F, Xiao-Li Lian2B, Xiao-Hua Dai2C, Su-Xia Liang12A, Ying-Bin Yan24A*DOI: 10.12659/MSM.934511
Med Sci Monit 2022; 28:e934511
Abstract
BACKGROUND: The aims of the study were to comprehensively compare the morphology, immunophenotype, proliferation, migration, and regeneration potential of normal dental pulp stem cells (DPSCs) versus inflammatory dental pulp stem cells (iDPSCs).
MATERIAL AND METHODS: Healthy pulp or inflamed pulp tissue was used to isolate and culture DPSCs and iDPSCs, respectively. These cell populations were characterized by flow cytometry, colony formation assay, transwell assay, and multi-directional differentiation in vitro.
RESULTS: No difference was observed in the morphology, cell-surface markers, or cell migration between DPSCs and iDPSCs. DPSCs showed a higher colony-forming capacity, proliferative viability, and osteo/dentinogenesis ability compared with iDPSCs. However, iDPSCs demonstrated enhanced neurogenesis, angiogenesis, adipogenesis, and chondrogenesis capacities in comparison to DPSCs.
CONCLUSIONS: Our data revealed the differences of biological properties between DPSCs and iDPSCs. The highly angiogenic and neurogenic potential of iDPSCs indicate their possible use in the regeneration of the dentin-pulp complex and support the critical role of angiogenesis and neurogenesis in pulp regeneration.
Keywords: Cell Proliferation, Dental Pulp, Stem Cells, Adult, Cell Movement, Female, Flow Cytometry, Follow-Up Studies, Humans, Immunophenotyping, Male, Osteogenesis, young adult
Background
Dental pulp, which is surrounded by dentin, is a type of mesenchymal soft tissue derived from cranial nerve crest cells, which plays an important role in the dynamic equilibrium of teeth. Irreversible pulpitis refers to non-healing inflammatory lesions of dental pulp tissue that result in spontaneous pain, pulp necrosis, and abscess formation [1]. In the clinic, the routine treatment of teeth with irreversible pulpitis is to remove the diseased pulp tissue and perform root canal therapy (RCT). Although the success rate of RCT can be as high as 92–98% [2], the treatment process itself is expensive, technically challenging, and time-consuming. In addition, removing the pulp tissue weakens the tooth structurally, which makes it more prone to fracture, and can also cause infection [3].
In an effort to preserve root pulp vitality, minimally invasive pulpotomy has recently gained popularity for the treatment of irreversible pulpitis [4]. However, some complications, such as crown fracture, intradental resorption, and root canal calcification [5], still cannot be avoided completely. Adult stem cells, which are undifferentiated cell groups located in differentiated organs or tissues, have been used to repair or regenerate damaged tissues or organs due to their self-renewal abilities and multi-differentiation potential [6]. Therefore, stem cell-based therapy to regenerate the dentin-pulp complex is an alternative and promising approach to restore the function of pulp tissue damaged by injury or inflammation [7].
Dental pulp stem cells (DPSCs), which are one of the important sources of adult mesenchymal stem cells (MSCs), were first described by Gronthos et al [8]. They are a type of undifferentiated cell found in the dental pulp that have the potential for self-renewal and have been induced to form mineralized nodules in vitro [8]. Follow-up studies have confirmed that DPSCs can differentiate into osteoblast/odontoblast-like cells, adipocytes, chondrocytes, neurons, skeletal muscle cells, hepatocytes, and endothelial cells in vitro and form bone-like structures after transplantation in vivo [9,10]. In 2010, Wang et al [11] isolated stem cells from dental pulp tissue that had irreversible pulpitis, and named them inflammatory dental pulp stem cells (iDPSCs). Due to ethics restrictions, it is difficult to acquire DPSCs from humans. Therefore, iDPSCs are a more readily available source for stem cell-based therapy rather than simply being relegated to medical waste that is usually discarded.
Previous studies suggest that DPSCs and iDPSCs appear to have different biological properties, although the results remain controversial. In some studies, researchers have concluded that DPSCs had a significantly higher potential of proliferation and osteogenic and adipogenic differentiation than iDPSCs [11,12]. In other studies, the investigators observed that there were no significant differences between the proliferation and multi-directional differentiation potential of DPSCs and iDPSCs [13–15]. In still other publications, iDPSCs were shown to have a stronger osteogenic capacity than DPSCs [16]. This disparity might be due to the small sample sizes used in these studies and also to the heterogeneity of sex, age, and tooth position. In addition, previous studies have focused on the potential of cells to differentiate into hard tissue but have neglected their neuronal and vascular differentiation abilities, which are another important potential factor involved in regenerating the dentin-pulp complex [17]. In fact, only one prior study mentioned the neurogenic potential of the DPSCs and iDPSCs [16]. Until now, no study has compared the potential of vascular differentiation between DPSCs and iDPSCs.
The purpose of this study was to comprehensively compare the biological properties (including proliferation and multi-lineage differentiation potentials) of DPSCs and iDPSCs using a relatively large sample size with a relatively homogeneous age and tooth position.
Material and Methods
SUBJECTS AND TISSUE HARVEST:
The teeth used for the experiment were obtained from the alveolar surgery clinic of Tianjin Stomatological Hospital. Each patient signed an informed consent form and agreed to allow us to use their teeth in our research work. The local ethics committee’s approval to conduct the study was also obtained (2015013).
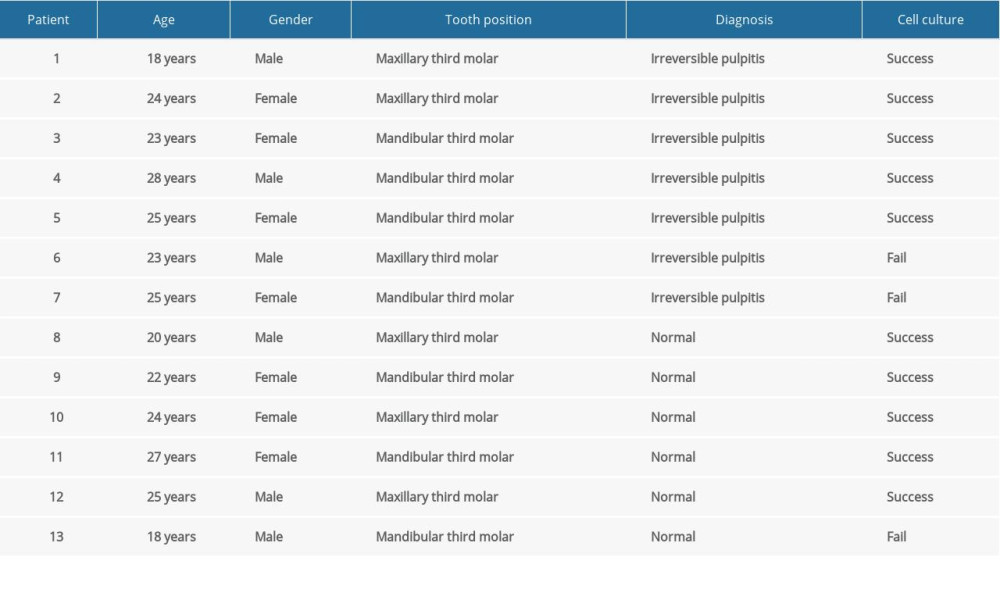
Patients were eligible for this study if they sought treatment that involved the extraction of mandibular or maxillary third molars and were between the ages of 18 and 30 years old. For each patient, only 1 tooth was requested. We collected healthy pulp tissues from caries-free, fully erupted third molars. The inflammatory pulp tissue was derived from carious wisdom teeth with irreversible pulpitis diagnosed by an endodontic specialist based on previously described criteria [13]. The pulp’s vitality was confirmed by the electronic pulp test on access. The only elimination criterion was teeth with completely necrotic pulps [13]. The status of the pulp tissue was further verified by histological examination. Between January 2019 and August 2019, 13 patients 18–28 years of age participated in this study. Detailed information regarding the patients included in the experiment is shown in Table 1.
After extraction (Figure 1A), the teeth were immediately placed in a material extraction solution, as previously described [18]. The teeth were photographed with cone-beam computed tomography within 30 min (Figure 1B) and then transferred to an ultra-clean workbench. The teeth were washed repeatedly with phosphate-buffered saline (PBS, Solaibao) containing 100 U/mL penicillin and 100 μg/mL streptomycin. After being wrapped in sterile gauze, the teeth were crushed with a hammer, and the dental pulp tissues were extracted using sterile instruments (Figure 1C). The dental pulp tissue located at the apex was cut by a sterile ophthalmology scalpel and discarded. A section of the crown pulp tissue was placed in a 4% paraformaldehyde solution for hematoxylin-eosin (HE) staining to identify the status of the pulp (Figure 1C). The remaining fresh crown pulp tissue was placed in a sterile culturing medium for isolation of cells.
ISOLATION, CULTURING, AND PURIFICATION OF DPSCS AND IDPSCS:
The primary cells were cultured using the tissue attachment method [19]. The dental pulp was cut to a size of 0.5–1 mm3 with a sterile blade and moistened with sterile sample collection medium in a 60-mm petri dish. The minced tissue was placed directly into the bottom wall of the culture flasks without any collagenase treatment. The ensuing incubation, culturing, and purification processes were the same as previously described [18]. After 2 to 3 weeks of culture, the cells were harvested for later experiments [20].
FLOW CYTOMETRY:
The third-passage cells were harvested and washed 3 times with PBS. Then, a single-cell suspension was obtained by digestion with 0.25% trypsin. The cells were resuspended with PBS-1% fetal bovine serum (FBS) after centrifugation. Next, 1×106 cells were incubated in the dark for 30 min at 4°C with the corresponding commercial monoclonal antibodies (CD29, cloneTS2/16, BioLegend, Ref.303003, 1: 20; CD90, clone5E10, BD Pharmingen™, Ref.555596, 1: 20; CD166, clone3A6, Ancell Corporation, Ref.393–040, 1: 50; CD45, cloneHI30, BD Pharmingen™, Ref.555483, 1: 5; CD34, clone581, BD Pharmingen™, Ref.555822, 1: 5; Isotype controls: Mouse IgG1, AbD Serotec, Ref.MCA928F, 1: 10; Mouse IgG1, κ, BD Pharmingen™, Ref.555749, 1: 5). Flow cytometry was performed on a FACSCanto device (BD Biosciences). The data were analyzed using FACSDiva (BD Biosciences) and FlowJo software (TreeStar, Inc, Ashland, OR).
IMMUNOFLUORESCENCE STAINING: The third-passage cells were inoculated in a confocal petri dish (Nest) at a density of 1×104/mL. The immunofluorescence staining was conducted as previously described [18]. The monoclonal antibodies used were phycoerythrin (PE)-conjugated anti-human CD90 (clone: 5E10, BD Pharmingen™, Ref.55559, 1: 20) and fluorescein isothiocyanate (FITC)-conjugated anti-human CD166 (clone: 3A6, Ancellular Corporation, Ref.393040, 1: 50). The fluorescently labeled cells were observed and photographed using inverted fluorescence microscopy (Nikon Ti-S).
COLONY-FORMING UNIT (CFU) ASSAY: The third-passage cells were inoculated onto a 6-well plate (Corning) at a density of 200 cells per plate. The CFU assay was performed as previously described [18]. The percentage of clone-formation efficiency was expressed as previously described [21].
CELL PROLIFERATION ASSAY: The third-passage cells were seeded in 96-well plates (Corning) at a density of 2×104/mL. The cell proliferation assay was performed with a cell counting kit-8 (CCK-8) kit as previously described [18].
TRANSWELL MIGRATION ASSAY: We used a transwell migration assay (Corning) to examine the vertical migration ability. This assay was performed as previously described [22]. The average number of migrated cells in 5 random fields was compared between the DPSCs and iDPSCs [22].
IN VITRO PENTA-LINEAGE DIFFERENTIATION EXPERIMENT:
After the third passage, DPSCs and iDPSCs (
OSTEOGENIC/ODONTOGENIC DIFFERENTIATION:
The third-passage cells were cultured under osteogenic conditions in vitro as previously described [18]. The cells were incubated at 37°C and 5% CO2, with biweekly, half-medium changes. Alizarin Red staining was used to detect the calcium deposition after induction for 14 days, as previously described [18]. The area ratio of calcium deposition in the 2 groups was measured with Image J software. Real-time quantitative polymerase chain reaction (PCR) was used to detect the mRNA expression of osteogenic/odontogenic markers, such as alkaline phosphatase (ALP), Run-related transcription factor 2 (Runx2), type I alpha 1 collagen (Col1A1), dentin sialophosphoprotein (DSPP), and dentin matrix acidic phosphoprotein 1 (DMP1), after induction for 1 or 2 weeks.
CHONDROGENIC DIFFERENTIATION:
We used 2 different induction methods (either by petri dish or by pellet), for chondrogenic differentiation that corresponded to different experimental purposes. During induction by petri dish, the third-passage cells were inoculated into a 35-mm petri dish (Corning) at a density of 1×105/mL. When cell confluence reached to approximately 70–80%, a cartilage induction solution (Cyagen, China, Ref.HUXMA-9004) was added after rinsing with PBS. The cells were induced for 28 days with biweekly, half-medium changes. The cells were then used to isolate the total RNA, the mRNA expressions of type II alpha 1 collagen (Col2A1) and sex-determining region Y (SRY)-box9 (SOX9) were detected by real-time PCR.
The induction of chondrogenic differentiation by pellet was performed as previously described [18]. The inducted pellets were fixed, cut into 4-μm-thick sections, and stained with Alcian blue (Sigma) or type II collagen (Solarbio) as previously described [18].
ADIPOGENIC DIFFERENTIATION:
The third-passage cells were cultured in adipogenic conditions in vitro as previously described [18]. After 28 days of induction, the cells were stained with Oil Red O (Solarbio), and the area ratio of lipid droplet formation of the 2 groups was measured using Image J software. The mRNA expression of the adipogenesis marker peroxisome proliferator activated receptor gamma (PPAR-γ) and lipoprotein lipase (LPL) were measured by real-time PCR after 14 days of induction.
NEURONAL CELLS DIFFERENTIATION:
Early neuronal marker nestin and mature neuronal marker tubulin were selected to identify the neuronal cells differentiation of the DPSCs and iDPSCs. Immunohistochemistry and immunofluorescence staining were also used to detect the expression of these 2 markers.
For the immunohistochemistry staining, the cells were prepared for cell culture with coverslips [23]. In brief, the cover glass was placed in a 35-mm petri dish (Corning). A cell suspension with a density of 1×105/mL was dripped onto the glass slides using a liquid transfer gun, with each drop having a volume of 2 mL. The inoculated cells were incubated in pre-inducing nerve induction solution (α-MEM, 20% FBS, 10 μg/L basic fibroblast growth factor [bFGF, Proteintech]) at 37°C and 5% CO2 for 24 h. Then, the medium was replaced with nerve induction solution (α-MEM medium supplemented with 1-μM hydrocortisone [BioRuler], 2% dimethyl sulfoxide [DMSO, Gibco], 200 μM butylhydroxyanisole [BHA, BioRuler], 10-μM forsklin [BioRuler], 0.1-mM β-Sulfoethanol [BioRuler]). After induction for 24 h, the cells climbing the slides were removed from the petri dish, washed with PBS, and fixed in 4% paraformaldehyde for 5 min. Next, the immunohistochemistry of nestin (clone: 1D11H2, Proteintech, Ref.66259-1-Ig, 1: 200) and tubulin (clone: 1D4A4P, Proteintech, Ref.66240-1-Ig, 1: 200) was performed according to the manufacturer’s instructions. An Olympus BX51 microscope was used to observe the cells and take photos.
The process of immunofluorescence staining for nestin (clone: 1D11H2, Proteintech, Ref.66259-1-Ig, 1: 100) and tubulin (clone: 1D4A4, Proteintech, Ref.66240-1-Ig, 1: 100) was the same as previously described [18]. We used the second antibody Alexa Fluor 488-conjugated Goat Anti-Mouse IgG (Proteintech, Ref.SA00013-1, 1: 200) or Alexa Fluor 594-conjugated Goat Anti-Mouse IgG (Proteintech, Ref.SA00013-3, 1: 200). The fluorescently labeled cells were visualized and photographed using an inverted fluorescence microscope (Nikon Ti-S).
The expression of the neurogenic genes nestin and tubulin were detected by real-time PCR following induction.
ENDOTHELIAL CELLS DIFFERENTIATION:
The third-passage cells were cultured in an angiogenic differentiation medium (α-MEM medium supplemented with 10% FBS, 100 U/mL penicillin, 100 μg/mL streptomycin, 1% L-glutamine, 50 ng/mL vascular endothelial growth factor [VEGF], 10 ng/mL bFGF, and 5 ng/mL EGF [Proteintech®]) for 14 days. The cells were observed under an inverted microscope (NikonTi-S), and their morphological characterizations were recorded. A Matrigel angiogenesis assay was carried out to observe the lumen formation as follows. The cells were diluted to 1×105/mL. The Matrigel (Corning), petri dish, and pipette tips were prechilled in a 4°C refrigerator for 1 day in advance. The Matrigel was mixed in serum-free medium at 1: 1 and evenly spread in a 35-mm petri dish with 200 μL. The cells were placed in the incubator at 37°C for 30 min and then inoculated into the Matrigel and incubated at 37°C for 4 to 6 h. The lumen formation was observed under inverted fluorescence microscopy (Nikon Ti-S). After induction for 1 or 2 weeks, real-time quantitative PCR was used to detect the mRNA expression of several markers related to blood vessel formation, such as VEGF, vascular endothelial growth factor receptor 2 (VEGFR2), stromal cell-derived factor-1 (SDF1), von Willebrand factor (vWF), CD31, and CD34. After 14 days of induction, 2 markers (VEGF and CD31) were detected by immunofluorescence staining, as previously described [18]. The anti-mouse CD31 monoclonal antibody labeled with PE (SANTA, Ref.SC-376764, 1: 200) and the anti-rabbit VEGF polyclonal antibody labeled with FITC (Beyotime, Ref.AF0312, 1: 200) were used. The second antibodies were m-IgGκBP-CFL594 (SANTA, Ref.SC-516178, 1: 200) and mouse anti-rabbit IgG-FITC (SANTA, Ref.SC-2359, 1: 200). The fluorescent-labeled cells were visualized and photographed by inverted fluorescence microscopy (Nikon Ti-S).
TOTAL RNA EXTRACTION AND REAL-TIME PCR:
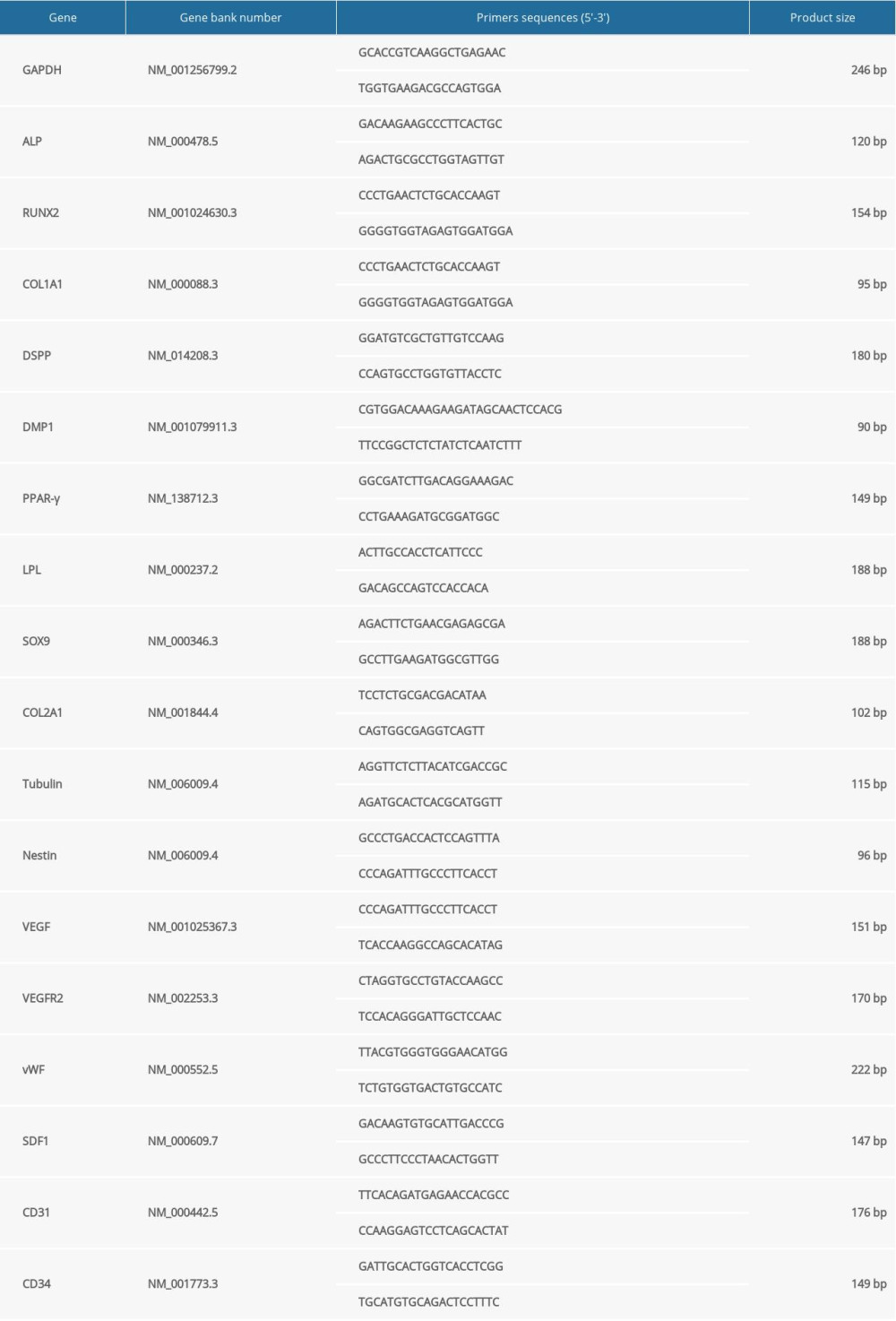
The total RNA was isolated from cells using a Mini BEST Universal RNA Extraction Kit (TaKaRa, Japan) according to the manufacturer’s instructions. cDNA was obtained using a synthesis kit (Promega, USA) in a 20-μL reaction system containing 2 μg of the total RNA [24]. Quantitative real-time PCR was performed using a LightCycler 480 II instrument (Roche, Switzerland) and FastStart Universal SYBR Green Master (Roche, Ref. 04913850001). The primers are listed in Table 2. The reaction system and PCR cycle parameters were the same as previously described [24]. The housekeeper glyceraldehyde-3-dehydrogenase (GAPDH) gene was used to standardize the expression of the target genes. The relative expression of the target genes was calculated using the 2−ΔΔct method, as previously described [24].
STATISTICAL ANALYSIS:
For the digital data, a chi-square test was used. Next, a Kolmogorov-Smirnov test was carried out first to assess the normality of the measurement data. If the data conformed to a normal distribution, an independent-sample
Results
PATIENT CHARACTERISTICS:
Of the 13 total patients, wisdom teeth with healthy pulp tissue (the normal group) were extracted in 6 (46.2%), while the remaining 7 (53.8%) had irreversible pulpitis (the inflammatory group) (Table 1). There were no significant differences in proportion regarding sex or tooth position between the 2 groups (Table 1). In addition, no significant difference in the means of age (24.3±3.01 vs 23.7±3.04 years, P=0.720) was found between the normal and inflammatory groups (Table 1). The histological examination verified the status of the pulp by clinical evaluation (Figure 1C).
CELL ISOLATION AND MORPHOLOGICAL CHARACTERIZATION:
Cell cultures succeeded in 5 of 7 inflammatory samples (71.4%) and in 5 of 6 normal samples (83.3%) (Table 1). The characteristic spindle-shaped or fibroblast-like cells were observed in both inflammatory and normal samples via the modified tissue mass method (Figure 1D). The tissue mass was completely attached to the plastic, and the cells climbed out and completely adhered to the plastic in 1 to 2 days and reached about 90% confluence after 8 days (Figure 1D). The cells isolated from the normal and inflammatory dental pulp tissue samples both had similar morphological characteristics.
IDPSCS EXHIBITED A SIMILAR IMMUNOPHENOTYPE AS DPSCS:
Both DPSCs and iDPSCs highly expressed CD166 (97.90±2.42% vs 99.28±0.56%, P=0.387), CD90 (99.84±0.13% vs 99.93±0.04%, P=0.307) and CD29 (99.79±0.09% vs 99.73±0.13%, P=0.513) (Figure 2A, 2B). In addition, the immunofluorescence analysis confirmed the co-expression of CD166 and CD90 for DPSCs and iDPSCs at the single-cell level (Figure 2C). Both DPSCs and iDPSCs were also negative for hematopoietic markers CD45 and CD34 (Figure 2A, 2B)
COMPARISONS OF THE CLONE FORMING CAPACITY, PROLIFERATION, AND MIGRATION POTENTIAL OF DPSCS AND IDPSCS:
Single-cell colonies were observed by crystal violet staining after 14 days (Figure 3A). The heterogeneity in size and cell density of these colonies reflected the differences in the cell proliferation rate (Figure 3A). There was a significant difference in the average colony formation rate between DPSCs and iDPSCs (13.6±0.74% vs 9.7±2.6%, P=0.027) (Figure 3B).
The cell proliferation curve was drawn according to the absorbance of each checkpoint using the CCK-8 method (Figure 3C). The curves showed a similar proliferative potential for DPSCs and iDPSCs from Day 1 to Day 4 (Figure 3C), but there was a significantly higher viability in the DPSCs compared to that in iDPSCs from Day 5 to Day 8 (P<0.05, Figure 3C).
After 24 h of culture, the migrated cells could be seen after crystal violet staining (Figure 3D). DPSCs and iDPSCs showed a similar cell migration rate (18.35±3.04% vs 17.11±3.58%, P=0.194) (Figure 3D).
DPSCS HAD A HIGHER OSTEOGENESIS/DENTINOGENESIS ABILITY THAN IDPSCS:
The osteogenic/dentinogenic differentiation potential of the 2 groups of cells was detected by alizarin red staining (Figure 4A). Both DPSCs and iDPSCs showed extracellular calcium deposition after 14 days of induction, while the control medium was negative (Figure 4A). The area ratio of calcium deposition in DPSCs was significantly larger than that in iDPSCs (36.1±1.76% vs 15.2±1.61%, P<0.001, Figure 4B), indicating a higher osteogenesis potential in DPSCs.
The expression levels of 5 osteogenic/dentinogenic lineage-specific genes (ALP, Runx2, Col1a1, DSPP, and DMP1) were detected by real-time PCR. After 7 days of induction, DPSCs showed a significantly higher expression of Col1a1 (0.83±0.13 vs 0.50±0.24, P=0.033) and DSPP (1.63±0.6 vs 0.15±0.05, P=0.005) than iDPSCs (Figure 4C). After 2 weeks of induction, the expression of ALP (0.85±0.32 vs 0.37±0.13, P=0.014) and DSPP (1.05±0.39 vs 0.44±0.12, P=0.011) in DPSCs was significantly higher than that in iDPSCs (Figure 4D).
IDPSCS HAD HIGHER CHONDROGENIC POTENTIAL THAN DPSCS:
DPSCs and iDPSCs showed stromal mucopolysaccharide formation by Alcian blue staining after pellet cartilage induction for 28 days (Figure 5A), but the control medium was negative (Figure 5A). In addition, the immunostaining of type II collagen was more obvious in the pellets subjected to chondrogenic conditions in comparison to those that experienced control conditions (Figure 5B).
The expressions of Col2a1 and SOX9 were examined by real-time PCR after 4 weeks of chondrogenic induction. The expression of Col2a1 (2.05±0.45 vs 0.92±0.14, P=0.01) and SOX9 (6.77±2.72 vs 2.30±1.21, P=0.017) in iDPSCs was significantly higher than that in DPSCs (Figure 5C).
IDPSCS HAD HIGHER ADIPOGENESIS POTENTIAL THAN DPSCS:
After 4 weeks of adipogenic induction, the Oil Red O staining of DPSCs and iDPSCs was positive, indicating the existence of adipocyte polyvesicular cells secreting lipid droplets (Figure 5D). However, 28-day non-stimulated cells demonstrated the absence or much weaker dyeing of lipids than the stimulated MSCs (Figure 5D). The area ratio of lipid droplet formation in iDPSCs was significantly larger than that in DPSCs (3.72±3.45% vs 0.47±0.38%, P<0.01) (Figure 5D).
The expression levels of PPARγ and LPL were tested by real-time PCR after 4 weeks of adipogenic induction. The iDPSCs showed a higher expression of PPARγ (4.78±2.08 vs 1.89±1.12, P<0.05) and LPL (3.46±1.29 vs 1.40±0.29, P<0.05) than DPSCs (Figure 5D).
IDPSCS HAD STRONGER NEUROGENIC POTENTIAL THAN DPSCS:
After 48 h of neurogenic induction, both DPSCs and iDPSCs contained neuron-like cells with slender cytoplasmic processes and a star-shaped appearance (Figure 6A), while the cells in the control medium presented with a spindle-forming fiber-like appearance (Figure 6A).
DPSCs and iDPSCs were positive in the immunostaining of nestin (Figure 6A, 6B) and tubulin (Figure 6A, 6B) after induction. The immunostaining intensity of nestin and tubulin seemed to be more prominent in iDPSCs than that in DPSCs (Figure 6A). Non-stimulated cells were characterized by the absence or much weaker staining for nestin and tubulin than the stimulated MSCs (Figure 6A, 6B).
The expression levels of the neuronal marker genes nestin and tubulin were also tested by real-time PCR after 48 h of induction. The iDPSCs showed a significantly higher expression of tubulin than the DPSCs (1.64±0.41 vs 0.87±0.39, P=0.016, Figure 6C). The expression of nestin in iDPSCs tended to be higher than that in DPSCs, but there was no statistically significant difference (Figure 6C).
IDPSCS HAD STRONGER ANGIOGENESIS POTENTIAL THAN DPSCS:
After 14 days of angiogenic induction, DPSCs and iDPSCs both demonstrated a flat, paving-stone-like shape that was common in vascular endothelial cells (Figure 7A), while the cells in the control medium presented with a long fusiform and vortex arrangement (Figure 7A). The induced cells were seeded in Matrigel for 6 h. Both the DPSCs and iDPSCs could form a lumen-like interconnected structure (Figure 7B). The luminal structure observed in iDPSCs seemed to be more obvious than that seen in DPSCs (Figure 7B).
The expression levels of 6 angiogenic lineage-specific genes were assessed by real-time PCR. The iDPSCs showed significantly higher expression levels of VEGF (1.39±0.3 vs 0.90±0.24, P=0.022), VEGFR2 (1.73±0.92 vs 0.68±0.18, P=0.038), and CD34 (6.35±1.13 vs 2.24±1.33, P=0.001) than DPSCs after 1 week of angiogenic induction (Figure 7C). No significant difference was observed in the expression levels of vWF, SDF1, and CD31 between the iDPSCs and DPSCs after 1 week of angiogenic induction (Figure 7C). After 2 weeks of induction, however, the iDPSCs showed significantly higher expressions of VEGF (1.53±0.17 vs 1.13±0.27, P=0.025), SDF1 (2.87±0.97 vs 0.89±0.37, P=0.008), CD31 (9.86±6.25 vs 2.31±1.67, P=0.031), and CD34 (2.41±0.43 vs 1.56±0.64, P=0.04) than DPSCs (Figure 7C). There was no significant difference in the expression of vWF and VEGFR2 between the iDPSCs and DPSCs (Figure 7C).
After 14 days of induction, DPSCs and iDPSCs demonstrated the co-expression of CD31 and VEGF by immunofluorescence staining, but the control medium remained negative (Figure 7D).
Discussion
Angiogenesis, which has often been neglected by previous studies, is critical for pulp regeneration [25]. The dental pulp vascular system not only helps establish nutrition and byproduct transport but also promotes the inflammatory response and tissue regeneration [26]. To the best of our knowledge, no study has compared the difference in angiogenesis between DPSCs and iDPSCs until now. In the present study, we showed for the first time that iDPSCs had stronger angiogenic differentiation potential than DPSCs. The inflamed dental pulp tissue is in a state of hypoxia due to the change in vascular permeability and the release of inflammatory mediators. Under hypoxic conditions, dental pulp cells quickly increase the expression of hypoxia-inducible transcription factor-1 [27], which mediates the transcription of angiogenic genes, such as VEGF [28,29]. As an angiogenic factor, VEGF plays an important role in tissue repair, inducing the survival of endothelial cells and the promotion of chemotaxis, proliferation, and differentiation of dental pulp cells [30,31]. Therefore, we suggest that hypoxic stress promoted the angiogenic differentiation of iDPSCs.
Neurogenesis plays an important role in the regeneration of the dental pulp-dentin complex. Previous studies have shown that DPSCs and iDPSCs can differentiate into mature neurons [32,33]. In the present study, we found that iDPSCs demonstrated increased neurogenesis compared with DPSCs, which indicated that the former might be more suitable as a source of stem cells to regenerate pulp nerves. However, Park et al [16] reported that the neurogenic differentiation potential of DPSCs was stronger than that of iDPSCs, which is contrary to our findings. The effects of inflammatory processes on the neuronal differentiation of MSCs is still a controversial topic. It seems that acute inflammation along with a relatively high level of proinflammatory cytokines would prevent the neuronal differentiation of neural precursor cells, while chronic inflammation accompanied by a relatively low level of proinflammatory cytokines would support their neuronal differentiation [34]. Therefore, we anticipated that the inflammatory level of the collected pulp tissue might have caused a discrepancy in different studies. Grading the level of inflamed pulp tissue on the basis of molecular markers should help to clarify the precise role of inflammation in the neurogenic differentiation of DPSCs.
Regarding the function of DPSCs in repairing dental tissue damage, these cells’ ability to form hard tissue has attracted the most attention so far. Accordingly, the osteogenic/dentinogenic differentiation potential naturally became a research focus when DPSCs were compared with iDPSCs, although the outcomes remained controversial. In the present study, we found that the odonto/osteogenic differentiation abilities of DPSCs were stronger than those of iDPSCs, which was consistent with the findings of Alongi et al [12] and Kim et al [35]. Clinically diagnosed irreversible pulpitis has also been associated with increased expression of the tumor necrosis factor-alpha (TNF-α) gene in human pulp [36]. It has also been shown that the osteogenic differentiation of DPSCs is impaired when they are exposed to TNF-α and interleukin-1β [37,38]. Similar outcomes have also been observed for periodontal ligament stem cells [39]. Therefore, the osteogenic differentiation of iDPSCs might be inhibited due to these increased inflammatory factors.
In contrast to our results, Park et al [16] reported that iDPSCs had a stronger osteogenic efficacy than DPSCs. However, only 2 samples were used for each group in their study, so an effect of individual differences on the outcomes could not be avoided. Interestingly, some other studies have found no significant difference in odonto/osteoblast differentiation between DPSCs and iDPSCs [11,13]. This disparity might be due to the different cohorts studied, since the ages of the subjects enrolled in the study of Wang et al [11] ranged from 6 to 40 years old, while we collected samples from patients 18–28 years of age. Additionally, the sample sizes might have affected the results of these different studies. Pereira et al [13] only enrolled 3 samples in each group, while we used 5 samples. Meaningful effects were likely to be missed due to the small sample sizes. In short, we tried to guarantee the heterogeneity of sex, age, and tooth position and used a relatively large sample, which minimized the influence of individual differences on the experimental results.
Although adipogenesis and chondrogenesis were used to verify the differentiation potential of DPSCs and iDPSCs, few studies have explored their differences, which is partly due to the irrelevant role of these processes played in the regeneration of pulp-dentin complex. The present study demonstrated that the adipogenic and chondrogenic differentiation abilities of iDPSCs were significantly stronger than those of DPSCs. Our results were supported by the study of Ramenzoni et al [39], who observed enhanced chondrogenesis and adipogenesis of the periodontal ligament stem cells under the stimulation of inflammatory cytokines IL-6, IL-8, and IL-1β. In contrast to our results, Pereira et al [13] reported no difference regarding their chondrogenic or adipogenic differentiation potential. This disparity might be due to the sample sizes, as only 3 samples in each group were used in the study of Pereira et al [13], and the meaningful effects were therefore likely to be missed. Further studies are needed to identify the underlying mechanism by which inflammation exerts its effect on the adipogenic and chondrogenic differentiation of iDPSCs.
We found that DPSCs showed a higher colony-forming capacity and proliferative viability than iDPSCs, which was consistent with the findings of previous studies [11,12,40]. Prior studies have demonstrated that mild inflammation of dental pulp, such as the pulp tissue extracted from teeth with deep caries, could stimulate the proliferation of DPSCs [41], while severe inflammation was thought to induce stem cell apoptosis [42, 43]. Based on our findings, we could postulate that the inflamed samples we obtained had been well exposed to severe inflammation, which inhibited the proliferation viability of their stem cells.
It seems that inflammation can influence the migration rate of stem cells. A previous study reported a higher migration rate of stem cells obtained from inflamed deciduous pulp samples compared with those from normal pulp tissues [35]. Park et al [44] also identified a similar phenomenon in human periodontal ligament stem cells. However, in the present study, no difference in the cell migration rate was observed between DPSCs and iDPSCs. Several biological aspects might explain the differences between our data and the findings of previous reports. In this study, we used adult pulp tissue, while Kim et al [35] performed their comparison using deciduous pulp tissue. Stem cells from different sources might react differently to inflammation, which could lead to varying results. In our study, iDPSCs might adopt the activation status at the inflamed pulp, thus manifesting a similar cell migration rate as DPSCs. Another possible explanation was that the differences in cell migration rates reported by previous studies [35,44] might reflect an individual’s genetic background but not necessarily be associated with inflammation. In fact, our results are supported by the study of Ramenzoni et al [39], who found that the migration of periodontal ligament stem cells was not affected by inflammatory cytokines.
There are several limitations to our study results that need to be considered. First, although we tried to control for confounding factors that could affect the functions of stem cells by reducing the heterogeneity of sex, age, and tooth position, absolute homogeneity could not be achieved. In fact, we had to collect both the maxillary and mandibular third molars due to difficulties in obtaining clinical specimens from eligible patients. Second, we used a relatively larger sample size than previous studies, but the 5 specimens in each group could not guarantee the complete elimination of individual differences since the sample size was still small. Therefore, the results of our study should not be overestimated but instead must be interpreted prudentially. Third, the inflammatory level of pulp tissues might have been a factor that influenced the biological properties of DPSCs; however, we did not grade the level of inflamed pulp tissues and therefore could not determine how they may have influenced our results. Further studies should attempt to clarify the precise role of inflammation on the functions of DPSCs.
Conclusions
Our data revealed differences in the biological properties of DPSCs and iDPSCs. The highly angiogenic and neurogenic potential of iDPSCs that was revealed by this study suggest their possible utility in the regeneration of the dentin-pulp complex.
Figures
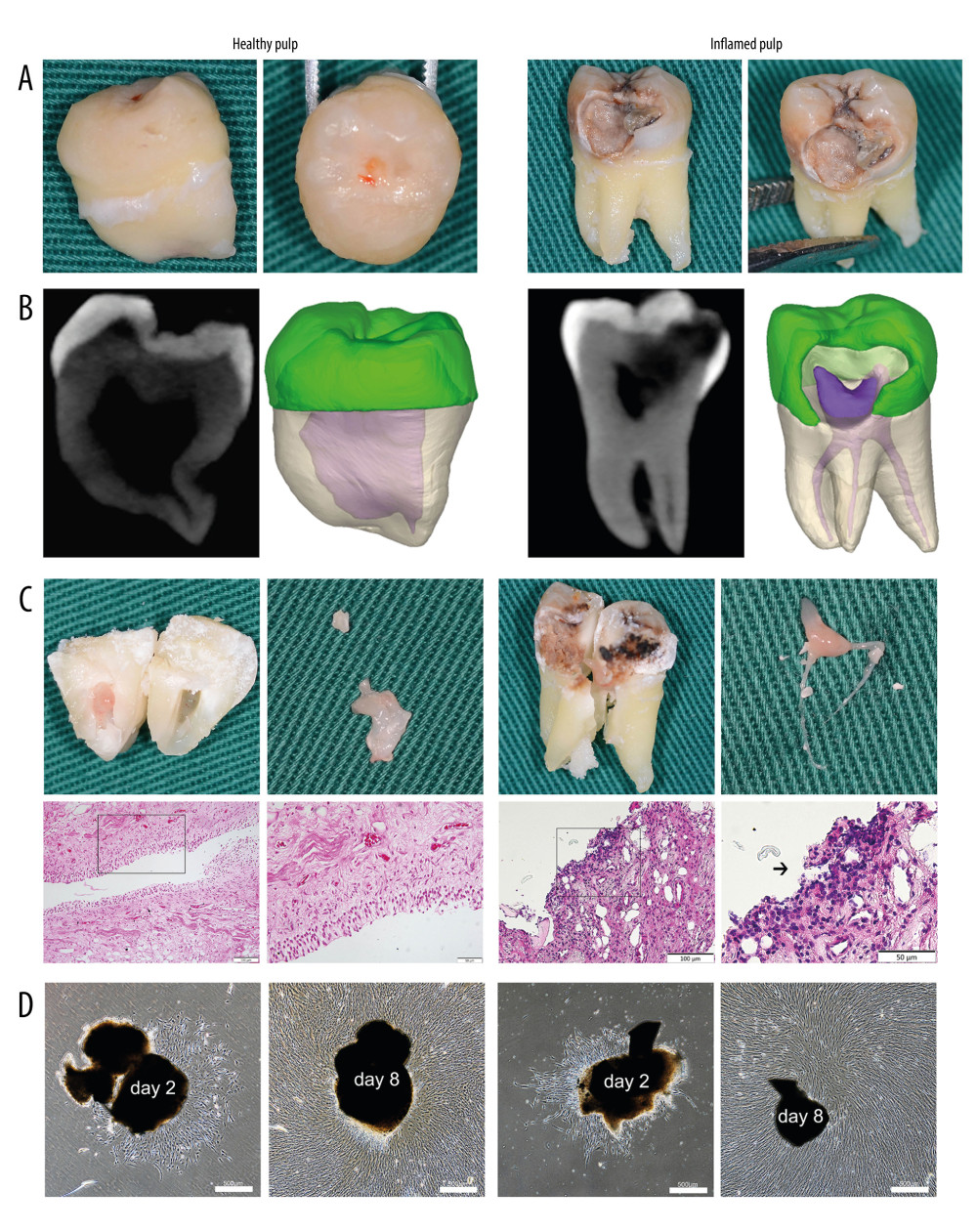 Figure 1. Tissue harvesting, cell culture and morphological characteristics. (A) The extracted teeth with healthy or inflamed pulp. (B) The three-dimensional pulp cavity morphology photographed by cone-beam computer tomography (CBCT). Please note that the carious lesion is continuous with the pulp cavity. (C) The split teeth, the exposure of pulp tissue, and the representative images of healthy and inflamed pulp tissues (hematoxylin and eosin stain, magnification, ×20; scale bar, 100 μm; magnification, ×40; scale bar, 50 μm). The arrow indicates a large number of round and deeply stained lymphocytes infiltrated in the pulp tissue. (D) DPSCs and iDPSCs showed similar morphologies at days 2 and 8 (magnification, ×4; scale bar, 500 μm). DPSCs, dental pulp stem cells; iDPSCs, inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 1. Tissue harvesting, cell culture and morphological characteristics. (A) The extracted teeth with healthy or inflamed pulp. (B) The three-dimensional pulp cavity morphology photographed by cone-beam computer tomography (CBCT). Please note that the carious lesion is continuous with the pulp cavity. (C) The split teeth, the exposure of pulp tissue, and the representative images of healthy and inflamed pulp tissues (hematoxylin and eosin stain, magnification, ×20; scale bar, 100 μm; magnification, ×40; scale bar, 50 μm). The arrow indicates a large number of round and deeply stained lymphocytes infiltrated in the pulp tissue. (D) DPSCs and iDPSCs showed similar morphologies at days 2 and 8 (magnification, ×4; scale bar, 500 μm). DPSCs, dental pulp stem cells; iDPSCs, inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 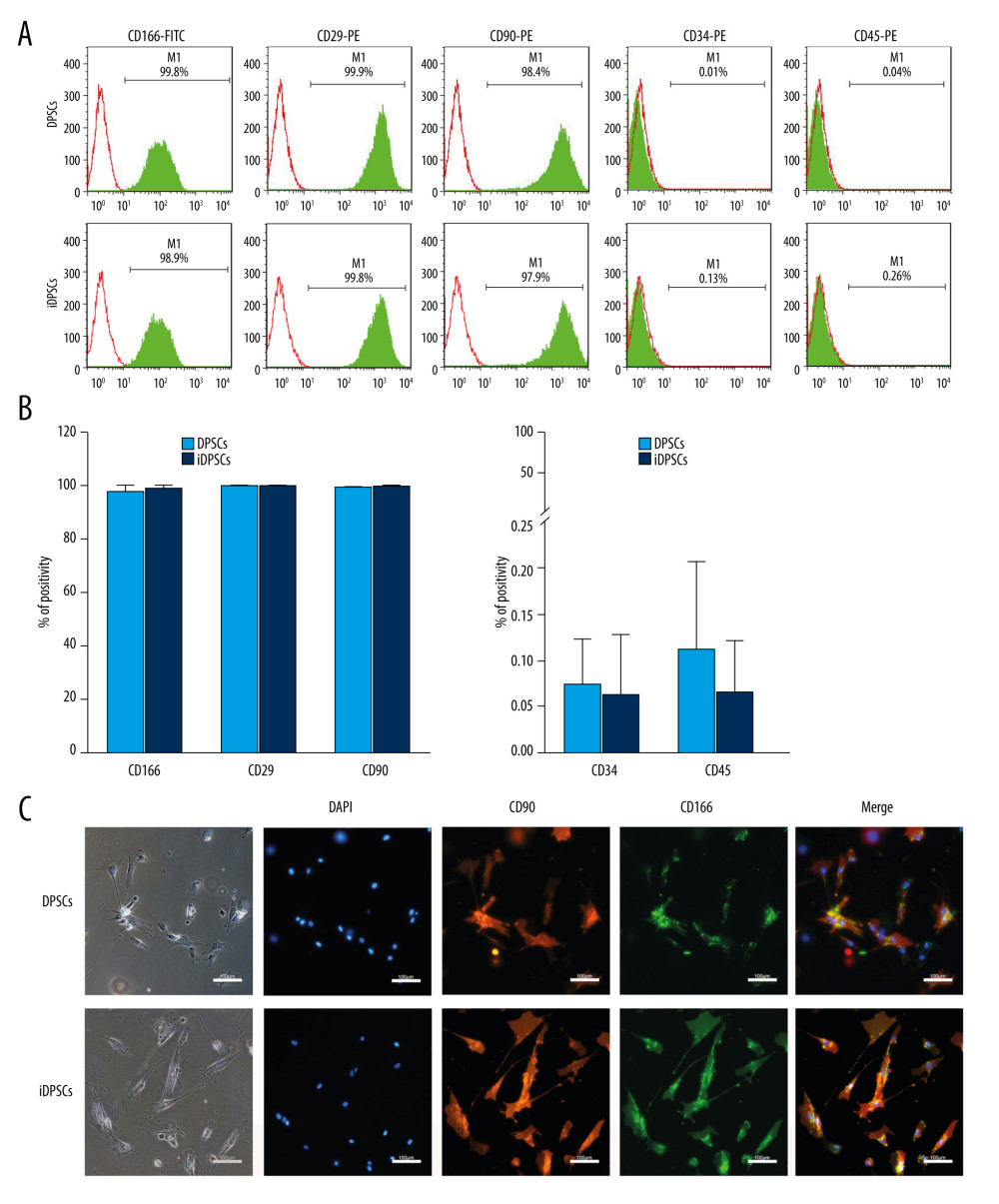 Figure 2. The immunophenotype characteristics of the DPSCs and iDPSCs. (A) Immunophenotype analysis by flow cytometry. The third-passage cells were incubated with monoclonal antibodies against the cell-surface marker antigens CD29, CD90, CD166, CD34, and CD45, followed by fluorescein-conjugated secondary antibodies. Both DPSCs and iDPSCs showed similar cell-surface marker expressions. (B) The percentage of positivity of the markers were calculated from 5 samples. (C) Immunofluorescence staining. The nuclei were stained with DAPI (blue). Both DPSCs and iDPSCs co-expressed CD166 and CD90 (magnification, ×20; Scale bar, 100 μm). DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; MSCs – mesenchymal stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 2. The immunophenotype characteristics of the DPSCs and iDPSCs. (A) Immunophenotype analysis by flow cytometry. The third-passage cells were incubated with monoclonal antibodies against the cell-surface marker antigens CD29, CD90, CD166, CD34, and CD45, followed by fluorescein-conjugated secondary antibodies. Both DPSCs and iDPSCs showed similar cell-surface marker expressions. (B) The percentage of positivity of the markers were calculated from 5 samples. (C) Immunofluorescence staining. The nuclei were stained with DAPI (blue). Both DPSCs and iDPSCs co-expressed CD166 and CD90 (magnification, ×20; Scale bar, 100 μm). DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; MSCs – mesenchymal stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 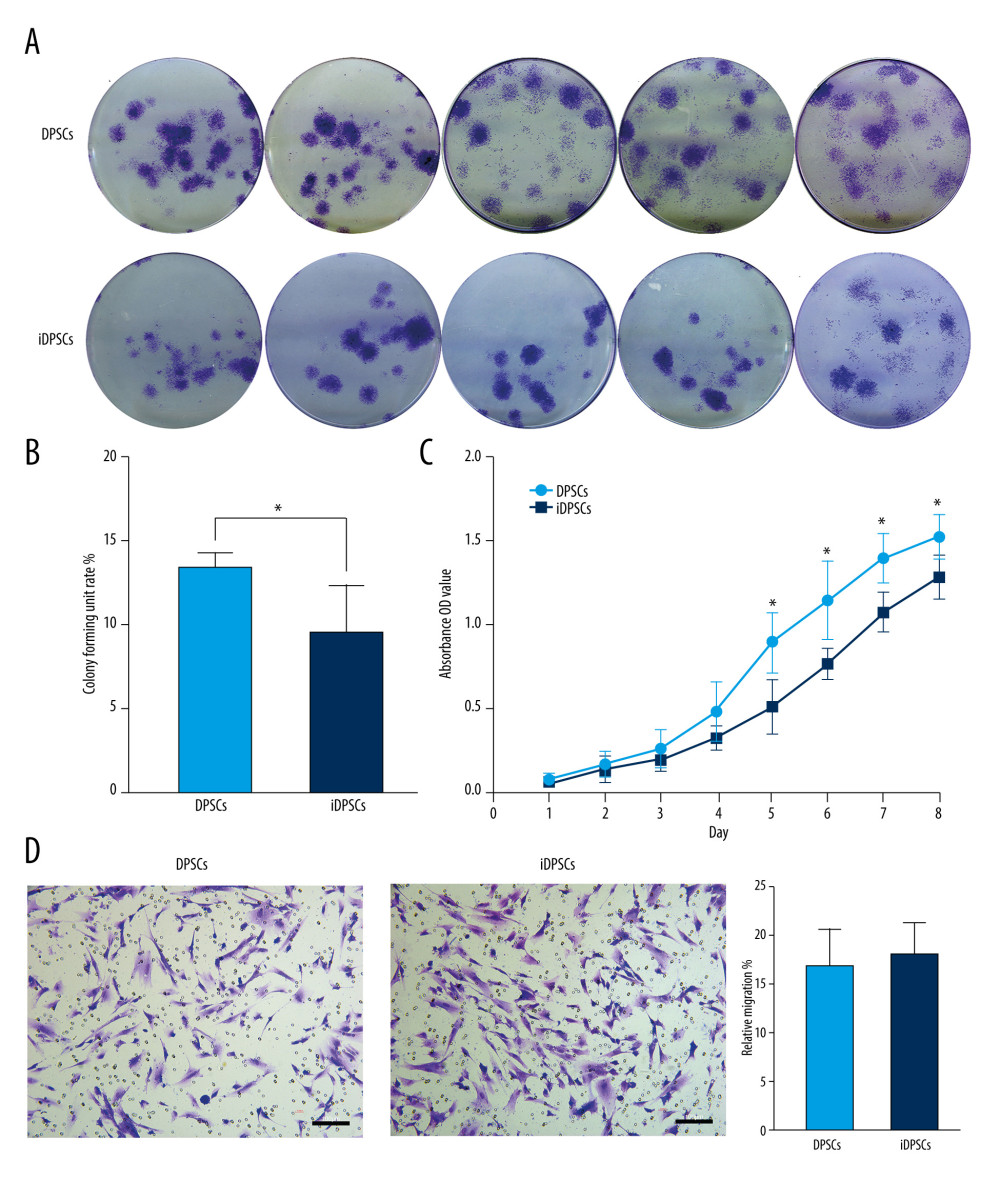 Figure 3. Clonogenic ability, cell proliferation and cell migration of the DPSCs and iDPSCs. (A) The clonogenic potential of DPSCs was significantly higher than that of iDPSCs judged from violet crystal staining. (B) The average colony formation efficiency was calculated from 5 samples. (C) DPSCs showed higher proliferation ability than iDPSCs from days 5 to 8 with CCK-8 test. * P<0.05. (D) The successful migration of DPSCs and iDPSCs showed similar migration potentials with crystal violet staining (magnification, ×20; scale bar, 100 μm); the bar graph represents the average migration rate analyzed from 5 samples. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 3. Clonogenic ability, cell proliferation and cell migration of the DPSCs and iDPSCs. (A) The clonogenic potential of DPSCs was significantly higher than that of iDPSCs judged from violet crystal staining. (B) The average colony formation efficiency was calculated from 5 samples. (C) DPSCs showed higher proliferation ability than iDPSCs from days 5 to 8 with CCK-8 test. * P<0.05. (D) The successful migration of DPSCs and iDPSCs showed similar migration potentials with crystal violet staining (magnification, ×20; scale bar, 100 μm); the bar graph represents the average migration rate analyzed from 5 samples. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 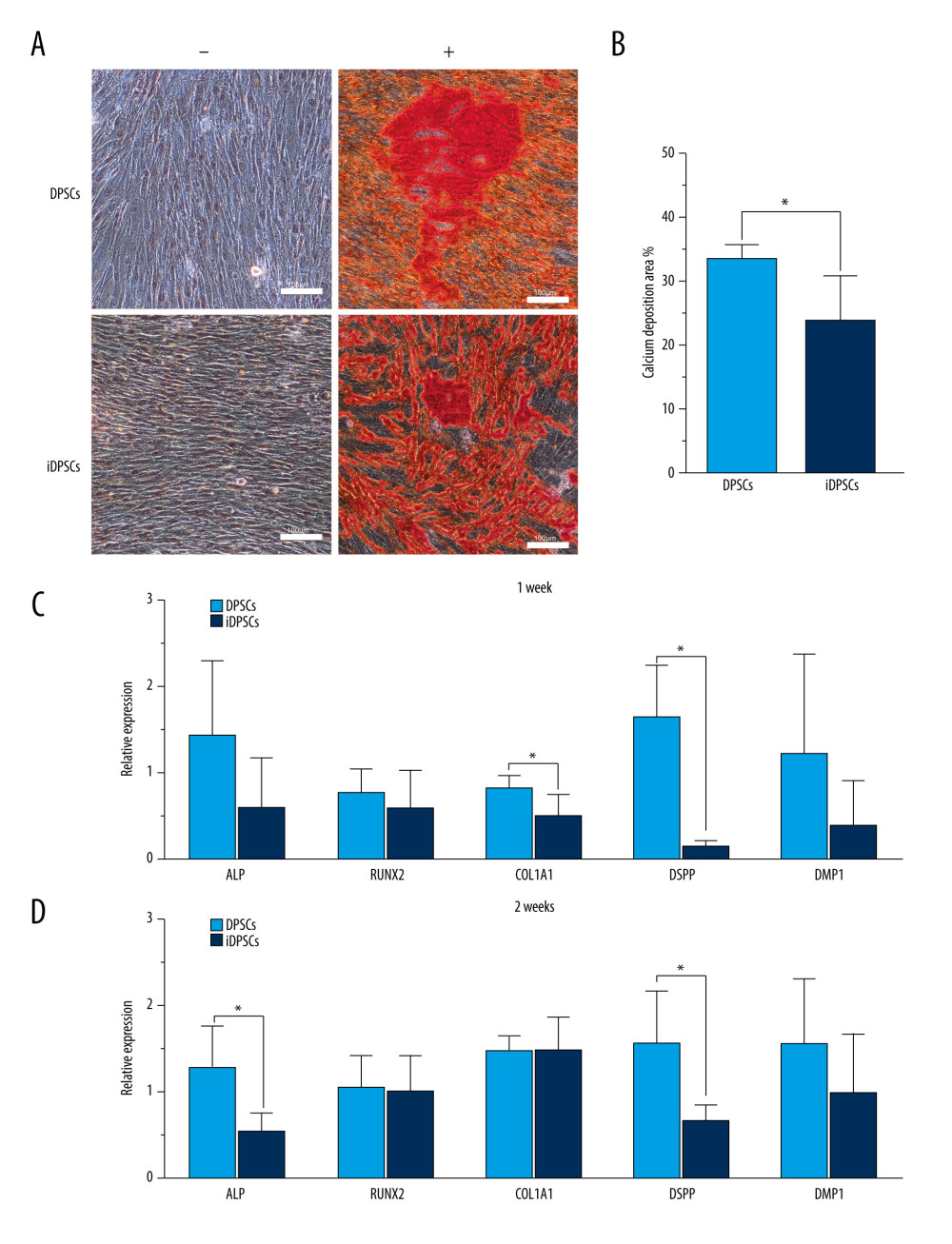 Figure 4. Osteogenic/dentinogenic differentiation induction analysis. (A) Mineralized nodules were detected after 14 days of induction following alizarin red (AR) staining. “+”, inductive medium; “−”, control medium. Scale bar, 100 μm. (B) The bar chart represents the percentage of area positively stained for AR (mean±standard deviation) analyzed from 5 samples, * P<0.05. (C, D) Several osteogenic/dentinogenic lineage-specific genes were examined by quantitative real-time polymerase chain reaction after 1-week and 2-week induction, * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 4. Osteogenic/dentinogenic differentiation induction analysis. (A) Mineralized nodules were detected after 14 days of induction following alizarin red (AR) staining. “+”, inductive medium; “−”, control medium. Scale bar, 100 μm. (B) The bar chart represents the percentage of area positively stained for AR (mean±standard deviation) analyzed from 5 samples, * P<0.05. (C, D) Several osteogenic/dentinogenic lineage-specific genes were examined by quantitative real-time polymerase chain reaction after 1-week and 2-week induction, * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 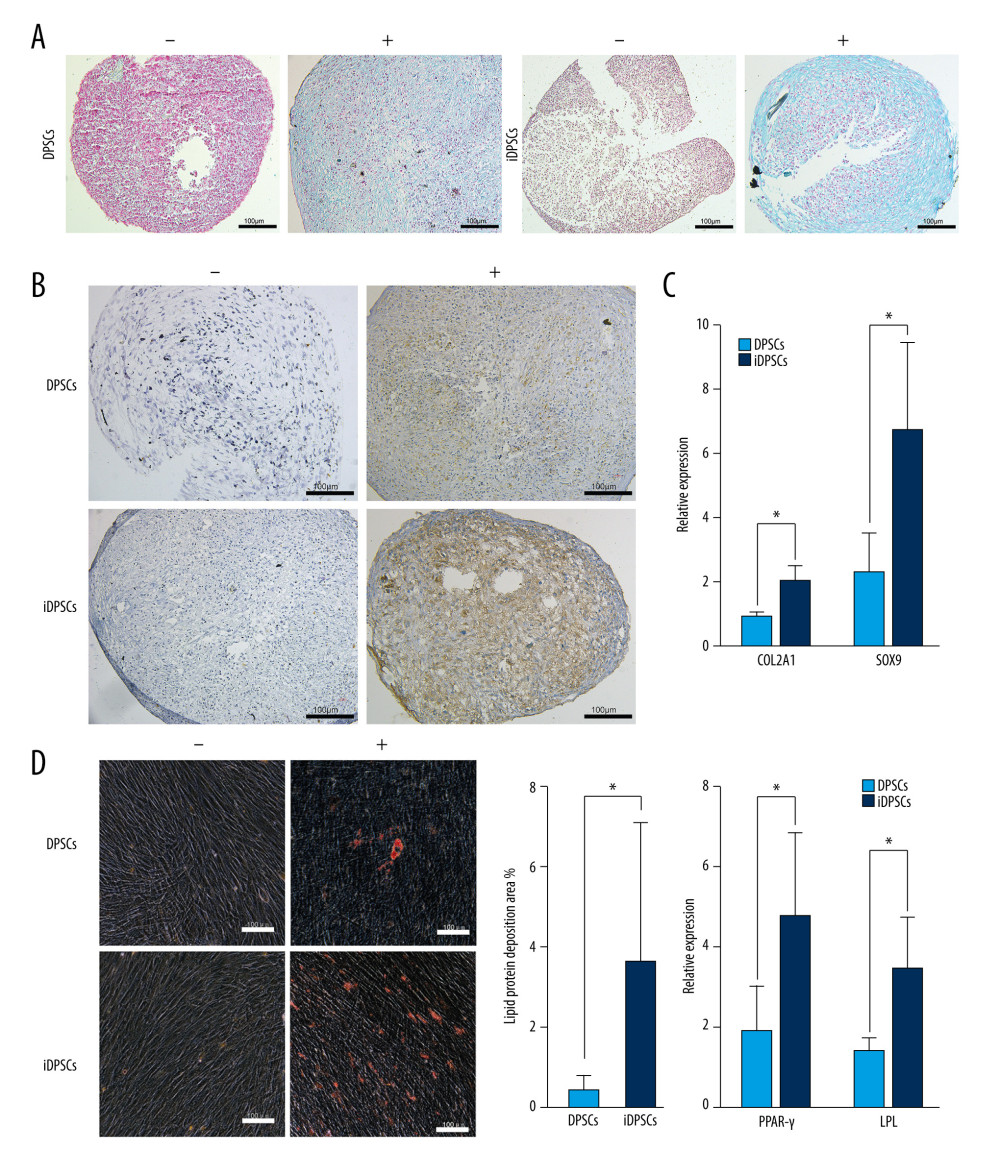 Figure 5. Chondrogenic and adipogenic differentiation induction analysis. In A and B, DPSCs and iDPSCs were cultured in non-differentiation control medium (−) and chondrogenic differentiation medium (+) for 28 days. (A) The matrix mucopolysaccharides were stained positively by Alcian blue after induction (magnification, ×20; scale bar, 100 μm). (B) Type II collagen was detected by immunohistochemical staining on micromasses (magnification, ×20; scale bar, 100 μm). (C) Quantitative real-time polymerase chain reaction measurements for chondrogenic markers SOX9 and Col2a1, * P<0.05. (D) The adipogenic differentiation analysis. The DPSCs and iDPSCs were cultured in non-differentiated control medium (−) and adipogenic differentiation medium (+) for 28 days. The presence of adipocytes was assessed by detecting lipid drops. Lipid vacuoles were stained positively for Oil Red O. Scale bar, 100 μm. The bar graph represents the percentage of area positively stained for Oil Red O (mean±standard deviation) analyzed from 5 samples. * P<0.05. Quantitative real-time polymerase chain reaction measurements for adipogenic markers PPARγ and LPL. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 5. Chondrogenic and adipogenic differentiation induction analysis. In A and B, DPSCs and iDPSCs were cultured in non-differentiation control medium (−) and chondrogenic differentiation medium (+) for 28 days. (A) The matrix mucopolysaccharides were stained positively by Alcian blue after induction (magnification, ×20; scale bar, 100 μm). (B) Type II collagen was detected by immunohistochemical staining on micromasses (magnification, ×20; scale bar, 100 μm). (C) Quantitative real-time polymerase chain reaction measurements for chondrogenic markers SOX9 and Col2a1, * P<0.05. (D) The adipogenic differentiation analysis. The DPSCs and iDPSCs were cultured in non-differentiated control medium (−) and adipogenic differentiation medium (+) for 28 days. The presence of adipocytes was assessed by detecting lipid drops. Lipid vacuoles were stained positively for Oil Red O. Scale bar, 100 μm. The bar graph represents the percentage of area positively stained for Oil Red O (mean±standard deviation) analyzed from 5 samples. * P<0.05. Quantitative real-time polymerase chain reaction measurements for adipogenic markers PPARγ and LPL. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 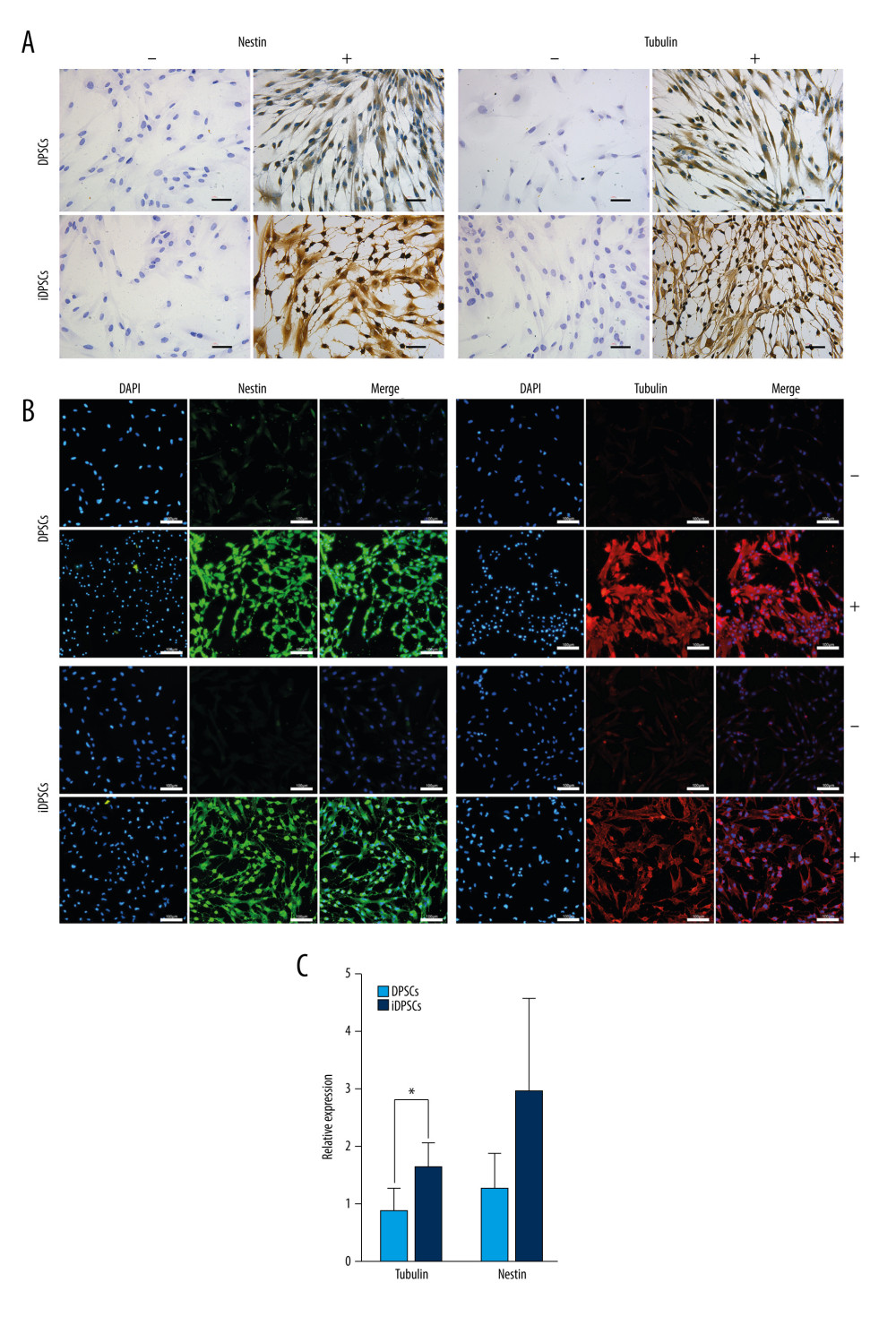 Figure 6. Expression of nestin and tubulin in DPSCs and iDPSCs. The cells were cultured in non-differentiated control medium (−) and neurogenic differentiation medium (+) for 48 hours. (A) Non-stimulated cells demonstrated the absence or much weaker staining for nestin and tubulin than stimulated cells (immunohistochemistry staining, magnification, ×40; scale bar, 50 μm). (B) Immunofluorescence staining of DPSCs and iDPSCs. All nuclei were stained with DAPI (blue). The cells were stained with antibody to nestin (green), and tubulin (red). Magnification, ×20; scale bar, 100 μm. (C) Quantitative real-time polymerase chain reaction measurements for neurogenic markers nestin and tubulin. The mRNA levels of 5 independent samples (n=5) were measured. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 6. Expression of nestin and tubulin in DPSCs and iDPSCs. The cells were cultured in non-differentiated control medium (−) and neurogenic differentiation medium (+) for 48 hours. (A) Non-stimulated cells demonstrated the absence or much weaker staining for nestin and tubulin than stimulated cells (immunohistochemistry staining, magnification, ×40; scale bar, 50 μm). (B) Immunofluorescence staining of DPSCs and iDPSCs. All nuclei were stained with DAPI (blue). The cells were stained with antibody to nestin (green), and tubulin (red). Magnification, ×20; scale bar, 100 μm. (C) Quantitative real-time polymerase chain reaction measurements for neurogenic markers nestin and tubulin. The mRNA levels of 5 independent samples (n=5) were measured. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). 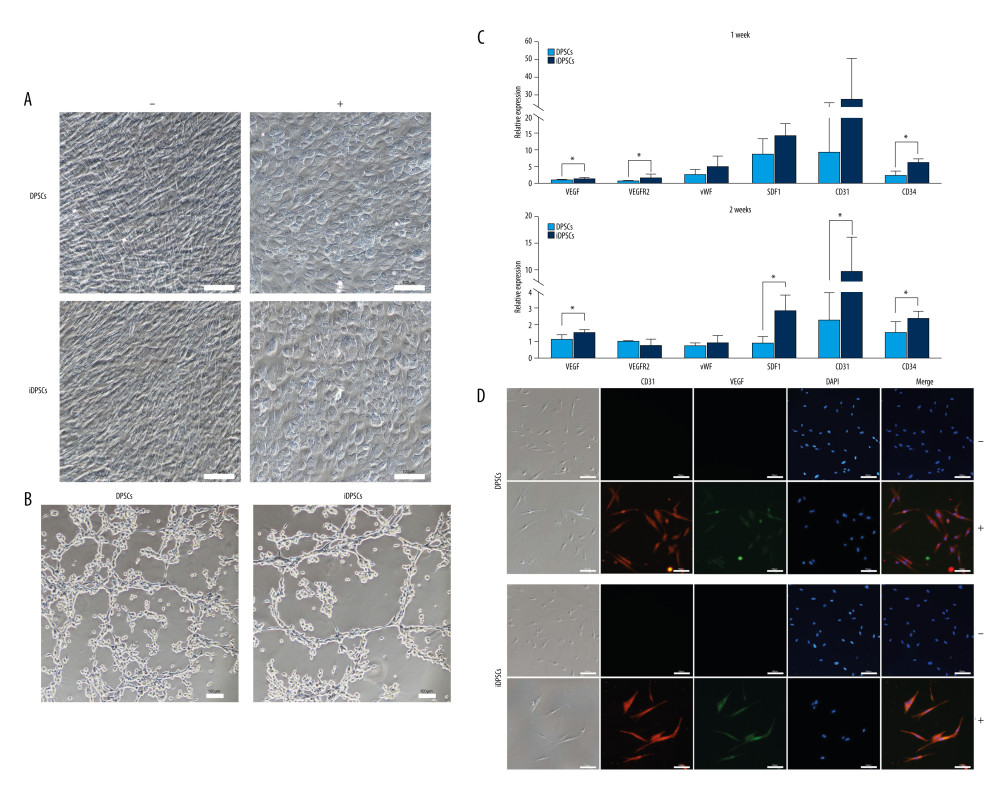 Figure 7. Angiogenic differentiation induction analysis. The cells were cultured in non-differentiated control medium (−) and angiogenic differentiation medium (+) for 2 weeks. (A) The cells showed a flat, paving-stone like shape after 14-day angiogenic induction (magnification, ×20; scale, 100 μm). (B) DPSCs and iDPSCs could form tubular network-like structure by Matrigel angiogenesis assay (magnification, ×20; scale, 100 μm). (C) several angiogenic lineage-specific genes were detected by real-time PCR. mRNA levels were tested in 5 independent samples. * P<0.05. (D) Expression of CD31 and VEGF in DPSCs and iDPSCs by immunofluorescence staining. All nuclei were stained with DAPI (blue). The cells were stained with antibody to VEGF (green), and CD31 (red). It was confirmed that DPSCs and iDPSCs co-expressed VEGF and CD31. magnification, ×20; scale bar, 100 μm. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 7. Angiogenic differentiation induction analysis. The cells were cultured in non-differentiated control medium (−) and angiogenic differentiation medium (+) for 2 weeks. (A) The cells showed a flat, paving-stone like shape after 14-day angiogenic induction (magnification, ×20; scale, 100 μm). (B) DPSCs and iDPSCs could form tubular network-like structure by Matrigel angiogenesis assay (magnification, ×20; scale, 100 μm). (C) several angiogenic lineage-specific genes were detected by real-time PCR. mRNA levels were tested in 5 independent samples. * P<0.05. (D) Expression of CD31 and VEGF in DPSCs and iDPSCs by immunofluorescence staining. All nuclei were stained with DAPI (blue). The cells were stained with antibody to VEGF (green), and CD31 (red). It was confirmed that DPSCs and iDPSCs co-expressed VEGF and CD31. magnification, ×20; scale bar, 100 μm. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). References
1. Cushley S, Duncan HF, Lappin MJ, Pulpotomy for mature carious teeth with symptoms of irreversible pulpitis: A systematic review: J Dent, 2019; 88; 103158
2. Friedman S, Mor C, The success of endodontic therapy – healing and functionality: J Calif Dent Assoc, 2004; 32(6); 493-503
3. Sjogren U, Hagglund B, Sundqvist G, Wing K, Factors affecting the long-term results of endodontic treatment: J Endod, 1990; 16(10); 498-504
4. Yazdani S, Jadidfard MP, Tahani B, Health technology assessment of CEM pulpotomy in permanent molars with irreversible pulpitis: Iran Endod J, 2014; 9(1); 23-29
5. Linsuwanont P, Wimonsutthikul K, Pothimoke U, Santiwong B, treatment outcomes of mineral trioxide aggregate pulpotomy in vital permanent teeth with carious pulp exposure: The retrospective study: J Endod, 2017; 43(2); 225-30
6. Gurusamy N, Alsayari A, Rajasingh S, Rajasingh J, Adult stem cells for regenerative therapy: Prog Mol Biol Transl Sci, 2018; 160; 1-22
7. Iohara K, Murakami M, Takeuchi N, A novel combinatorial therapy with pulp stem cells and granulocyte colony-stimulating factor for total pulp regeneration: Stem Cells Transl Med, 2013; 2(7); 521-33
8. Gronthos S, Mankani M, Brahim J, Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo: Proc Natl Acad Sci USA, 2000; 97(25); 13625-30
9. Tirino V, Paino F, De Rosa A, Papaccio G, Identification, isolation, characterization, and banking of human dental pulp stem cells: Methods Mol Biol, 2012; 879; 443-63
10. Ashri NY, Ajlan SA, Aldahmash AM, Dental pulp stem cells. Biology and use for periodontal tissue engineering: Saudi Med J, 2015; 36(12); 1391-99
11. Wang Z, Pan J, Wright JT, Putative stem cells in human dental pulp with irreversible pulpitis: An exploratory study: J Endod, 2010; 36(5); 820-25
12. Alongi DJ, Yamaza T, Song Y, Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential: Regen Med, 2010; 5(4); 617-31
13. Pereira LO, Rubini MR, Silva JR, Comparison of stem cell properties of cells isolated from normal and inflamed dental pulps: Int Endod J, 2012; 45(12); 1080-90
14. Attar A, Eslaminejad MB, Tavangar MS, Dental pulp polyps contain stem cells comparable to the normal dental pulps: J Clin Exp Dent, 2014; 6(1); e53-59
15. Malekfar A, Valli KS, Kanafi MM, Bhonde RR, Isolation and characterization of human dental pulp stem cells from cryopreserved pulp tissues obtained from teeth with irreversible pulpitis: J Endod, 2016; 42(1); 76-81
16. Park YT, Lee SM, Kou X, Karabucak B, The role of interleukin 6 in osteogenic and neurogenic differentiation potentials of dental pulp stem cells: J Endod, 2019; 45(11); 1342-48
17. Chalisserry EP, Nam SY, Park SH, Anil S, Therapeutic potential of dental stem cells: J Tissue Eng, 2017; 8; 2041731417702531
18. Zhang PP, Liang SX, Wang HL, Differences in the biological properties of mesenchymal stromal cells from traumatic temporomandibular joint fibrous and bony ankylosis: A comparative study: Anim Cells Syst (Seoul), 2021; 25(5); 296-311
19. Phelan K, May KM, Basic techniques in mammalian cell tissue culture: Curr Protoc Toxicol, 2016; 70; A3B1-22
20. Li Z, Chen S, Ma K, Comparison of different methods for the isolation and purification of rat nucleus pulposus-derived mesenchymal stem cells: Connect Tissue Res, 2020; 61(5); 426-34
21. Huang AH, Chen YK, Chan AW, Isolation and characterization of human dental pulp stem/stromal cells from nonextracted crown-fractured teeth requiring root canal therapy: J Endod, 2009; 35(5); 673-81
22. Li H, Liu S, Hu Y, Endosulfan promotes cell migration via PTP4A3-mediated signaling pathways in HUVECs: Ecotoxicol Environ Saf, 2020; 192; 110267
23. Rodig SJ, Growing adherent cells for staining: Cold Spring Harb Protoc, 2020; 2020(8); 099614
24. Liang SX, Wang HL, Zhang PP, Differential regulation of blood vessel formation between traumatic temporomandibular joint fibrous ankylosis and bony ankylosis in a sheep model: J Craniomaxillofac Surg, 2019; 47(11); 1739-51
25. Nakashima M, Iohara K, Sugiyama M, Human dental pulp stem cells with highly angiogenic and neurogenic potential for possible use in pulp regeneration: Cytokine Growth Factor Rev, 2009; 20(5–6); 435-40
26. Aksel H, Huang GT, Human and swine dental pulp stem cells form a vascularlike network after angiogenic differentiation in comparison with endothelial cells: A quantitative analysis: J Endod, 2017; 43(4); 588-95
27. Aranha AM, Zhang Z, Neiva KG, Hypoxia enhances the angiogenic potential of human dental pulp cells: J Endod, 2010; 36(10); 1633-37
28. Zimna A, Kurpisz M, Hypoxia-inducible factor-1 in physiological and pathophysiological angiogenesis: Applications and therapies: Biomed Res Int, 2015; 2015; 549412
29. Rombouts C, Giraud T, Jeanneau C, About I, Pulp vascularization during tooth development, regeneration, and therapy: J Dent Res, 2017; 96(2); 137-44
30. Dissanayaka WL, Zhang C, The role of vasculature engineering in dental pulp regeneration: J Endod, 2017; 43(9S); S102-6
31. Goncalves SB, Dong Z, Bramante CM, Tooth slice-based models for the study of human dental pulp angiogenesis: J Endod, 2007; 33(7); 811-14
32. Li J, Diao S, Yang H, IGFBP5 promotes angiogenic and neurogenic differentiation potential of dental pulp stem cells: Dev Growth Differ, 2019; 61(9); 457-65
33. Bojnordi MN, Haratizadeh S, Darabi S, Hamidabadi HG, Neural derivation of human dental pulp stem cells via neurosphere technique: Bratisl Lek Listy, 2018; 119(9); 550-53
34. Cacci E, Ajmone-Cat MA, Anelli T, Biagioni S, Minghetti L, In vitro neuronal and glial differentiation from embryonic or adult neural precursor cells are differently affected by chronic or acute activation of microglia: Glia, 2008; 56(4); 412-25
35. Kim J, Park JC, Kim SH, Treatment of FGF-2 on stem cells from inflamed dental pulp tissue from human deciduous teeth: Oral Dis, 2014; 20(2); 191-204
36. Kokkas AB, Goulas A, Varsamidis K, Irreversible but not reversible pulpitis is associated with up-regulation of tumour necrosis factor-alpha gene expression in human pulp: Int Endod J, 2007; 40(3); 198-203
37. Boyle M, Chun C, Strojny C, Chronic inflammation and angiogenic signaling axis impairs differentiation of dental-pulp stem cells: PLoS One, 2014; 9(11); e113419
38. Yuan H, Zhao H, Wang J, MicroRNA let-7c-5p promotes osteogenic differentiation of dental pulp stem cells by inhibiting lipopolysaccharide-induced inflammation via HMGA2/PI3K/Akt signal blockade: Clin Exp Pharmacol Physiol, 2019; 46(4); 389-97
39. Ramenzoni LL, Russo G, Moccia MD, Periodontal bacterial supernatants modify differentiation, migration and inflammatory cytokine expression in human periodontal ligament stem cells: PLoS One, 2019; 14(7); e0219181
40. Wang Y, Yan M, Wang Z, Dental pulp stem cells from traumatically exposed pulps exhibited an enhanced osteogenic potential and weakened odontogenic capacity: Arch Oral Biol, 2013; 58(11); 1709-17
41. Ma D, Gao J, Yue J, Changes in proliferation and osteogenic differentiation of stem cells from deep caries in vitro: J Endod, 2012; 38(6); 796-802
42. Goldberg M, Farges JC, Lacerda-Pinheiro S, Inflammatory and immunological aspects of dental pulp repair: Pharmacol Res, 2008; 58(2); 137-47
43. Yamaza T, Miura Y, Akiyama K, Mesenchymal stem cell-mediated ectopic hematopoiesis alleviates aging-related phenotype in immunocompromised mice: Blood, 2009; 113(11); 2595-604
44. Park JC, Kim JM, Jung IH, Isolation and characterization of human periodontal ligament (PDL) stem cells (PDLSCs) from the inflamed PDL tissue: in vitro and in vivo evaluations: J Clin Periodontol, 2011; 38(8); 721-31
Figures
 Figure 1. Tissue harvesting, cell culture and morphological characteristics. (A) The extracted teeth with healthy or inflamed pulp. (B) The three-dimensional pulp cavity morphology photographed by cone-beam computer tomography (CBCT). Please note that the carious lesion is continuous with the pulp cavity. (C) The split teeth, the exposure of pulp tissue, and the representative images of healthy and inflamed pulp tissues (hematoxylin and eosin stain, magnification, ×20; scale bar, 100 μm; magnification, ×40; scale bar, 50 μm). The arrow indicates a large number of round and deeply stained lymphocytes infiltrated in the pulp tissue. (D) DPSCs and iDPSCs showed similar morphologies at days 2 and 8 (magnification, ×4; scale bar, 500 μm). DPSCs, dental pulp stem cells; iDPSCs, inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 1. Tissue harvesting, cell culture and morphological characteristics. (A) The extracted teeth with healthy or inflamed pulp. (B) The three-dimensional pulp cavity morphology photographed by cone-beam computer tomography (CBCT). Please note that the carious lesion is continuous with the pulp cavity. (C) The split teeth, the exposure of pulp tissue, and the representative images of healthy and inflamed pulp tissues (hematoxylin and eosin stain, magnification, ×20; scale bar, 100 μm; magnification, ×40; scale bar, 50 μm). The arrow indicates a large number of round and deeply stained lymphocytes infiltrated in the pulp tissue. (D) DPSCs and iDPSCs showed similar morphologies at days 2 and 8 (magnification, ×4; scale bar, 500 μm). DPSCs, dental pulp stem cells; iDPSCs, inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 2. The immunophenotype characteristics of the DPSCs and iDPSCs. (A) Immunophenotype analysis by flow cytometry. The third-passage cells were incubated with monoclonal antibodies against the cell-surface marker antigens CD29, CD90, CD166, CD34, and CD45, followed by fluorescein-conjugated secondary antibodies. Both DPSCs and iDPSCs showed similar cell-surface marker expressions. (B) The percentage of positivity of the markers were calculated from 5 samples. (C) Immunofluorescence staining. The nuclei were stained with DAPI (blue). Both DPSCs and iDPSCs co-expressed CD166 and CD90 (magnification, ×20; Scale bar, 100 μm). DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; MSCs – mesenchymal stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 2. The immunophenotype characteristics of the DPSCs and iDPSCs. (A) Immunophenotype analysis by flow cytometry. The third-passage cells were incubated with monoclonal antibodies against the cell-surface marker antigens CD29, CD90, CD166, CD34, and CD45, followed by fluorescein-conjugated secondary antibodies. Both DPSCs and iDPSCs showed similar cell-surface marker expressions. (B) The percentage of positivity of the markers were calculated from 5 samples. (C) Immunofluorescence staining. The nuclei were stained with DAPI (blue). Both DPSCs and iDPSCs co-expressed CD166 and CD90 (magnification, ×20; Scale bar, 100 μm). DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; MSCs – mesenchymal stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 3. Clonogenic ability, cell proliferation and cell migration of the DPSCs and iDPSCs. (A) The clonogenic potential of DPSCs was significantly higher than that of iDPSCs judged from violet crystal staining. (B) The average colony formation efficiency was calculated from 5 samples. (C) DPSCs showed higher proliferation ability than iDPSCs from days 5 to 8 with CCK-8 test. * P<0.05. (D) The successful migration of DPSCs and iDPSCs showed similar migration potentials with crystal violet staining (magnification, ×20; scale bar, 100 μm); the bar graph represents the average migration rate analyzed from 5 samples. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 3. Clonogenic ability, cell proliferation and cell migration of the DPSCs and iDPSCs. (A) The clonogenic potential of DPSCs was significantly higher than that of iDPSCs judged from violet crystal staining. (B) The average colony formation efficiency was calculated from 5 samples. (C) DPSCs showed higher proliferation ability than iDPSCs from days 5 to 8 with CCK-8 test. * P<0.05. (D) The successful migration of DPSCs and iDPSCs showed similar migration potentials with crystal violet staining (magnification, ×20; scale bar, 100 μm); the bar graph represents the average migration rate analyzed from 5 samples. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 4. Osteogenic/dentinogenic differentiation induction analysis. (A) Mineralized nodules were detected after 14 days of induction following alizarin red (AR) staining. “+”, inductive medium; “−”, control medium. Scale bar, 100 μm. (B) The bar chart represents the percentage of area positively stained for AR (mean±standard deviation) analyzed from 5 samples, * P<0.05. (C, D) Several osteogenic/dentinogenic lineage-specific genes were examined by quantitative real-time polymerase chain reaction after 1-week and 2-week induction, * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 4. Osteogenic/dentinogenic differentiation induction analysis. (A) Mineralized nodules were detected after 14 days of induction following alizarin red (AR) staining. “+”, inductive medium; “−”, control medium. Scale bar, 100 μm. (B) The bar chart represents the percentage of area positively stained for AR (mean±standard deviation) analyzed from 5 samples, * P<0.05. (C, D) Several osteogenic/dentinogenic lineage-specific genes were examined by quantitative real-time polymerase chain reaction after 1-week and 2-week induction, * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 5. Chondrogenic and adipogenic differentiation induction analysis. In A and B, DPSCs and iDPSCs were cultured in non-differentiation control medium (−) and chondrogenic differentiation medium (+) for 28 days. (A) The matrix mucopolysaccharides were stained positively by Alcian blue after induction (magnification, ×20; scale bar, 100 μm). (B) Type II collagen was detected by immunohistochemical staining on micromasses (magnification, ×20; scale bar, 100 μm). (C) Quantitative real-time polymerase chain reaction measurements for chondrogenic markers SOX9 and Col2a1, * P<0.05. (D) The adipogenic differentiation analysis. The DPSCs and iDPSCs were cultured in non-differentiated control medium (−) and adipogenic differentiation medium (+) for 28 days. The presence of adipocytes was assessed by detecting lipid drops. Lipid vacuoles were stained positively for Oil Red O. Scale bar, 100 μm. The bar graph represents the percentage of area positively stained for Oil Red O (mean±standard deviation) analyzed from 5 samples. * P<0.05. Quantitative real-time polymerase chain reaction measurements for adipogenic markers PPARγ and LPL. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 5. Chondrogenic and adipogenic differentiation induction analysis. In A and B, DPSCs and iDPSCs were cultured in non-differentiation control medium (−) and chondrogenic differentiation medium (+) for 28 days. (A) The matrix mucopolysaccharides were stained positively by Alcian blue after induction (magnification, ×20; scale bar, 100 μm). (B) Type II collagen was detected by immunohistochemical staining on micromasses (magnification, ×20; scale bar, 100 μm). (C) Quantitative real-time polymerase chain reaction measurements for chondrogenic markers SOX9 and Col2a1, * P<0.05. (D) The adipogenic differentiation analysis. The DPSCs and iDPSCs were cultured in non-differentiated control medium (−) and adipogenic differentiation medium (+) for 28 days. The presence of adipocytes was assessed by detecting lipid drops. Lipid vacuoles were stained positively for Oil Red O. Scale bar, 100 μm. The bar graph represents the percentage of area positively stained for Oil Red O (mean±standard deviation) analyzed from 5 samples. * P<0.05. Quantitative real-time polymerase chain reaction measurements for adipogenic markers PPARγ and LPL. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 6. Expression of nestin and tubulin in DPSCs and iDPSCs. The cells were cultured in non-differentiated control medium (−) and neurogenic differentiation medium (+) for 48 hours. (A) Non-stimulated cells demonstrated the absence or much weaker staining for nestin and tubulin than stimulated cells (immunohistochemistry staining, magnification, ×40; scale bar, 50 μm). (B) Immunofluorescence staining of DPSCs and iDPSCs. All nuclei were stained with DAPI (blue). The cells were stained with antibody to nestin (green), and tubulin (red). Magnification, ×20; scale bar, 100 μm. (C) Quantitative real-time polymerase chain reaction measurements for neurogenic markers nestin and tubulin. The mRNA levels of 5 independent samples (n=5) were measured. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 6. Expression of nestin and tubulin in DPSCs and iDPSCs. The cells were cultured in non-differentiated control medium (−) and neurogenic differentiation medium (+) for 48 hours. (A) Non-stimulated cells demonstrated the absence or much weaker staining for nestin and tubulin than stimulated cells (immunohistochemistry staining, magnification, ×40; scale bar, 50 μm). (B) Immunofluorescence staining of DPSCs and iDPSCs. All nuclei were stained with DAPI (blue). The cells were stained with antibody to nestin (green), and tubulin (red). Magnification, ×20; scale bar, 100 μm. (C) Quantitative real-time polymerase chain reaction measurements for neurogenic markers nestin and tubulin. The mRNA levels of 5 independent samples (n=5) were measured. * P<0.05. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). Figure 7. Angiogenic differentiation induction analysis. The cells were cultured in non-differentiated control medium (−) and angiogenic differentiation medium (+) for 2 weeks. (A) The cells showed a flat, paving-stone like shape after 14-day angiogenic induction (magnification, ×20; scale, 100 μm). (B) DPSCs and iDPSCs could form tubular network-like structure by Matrigel angiogenesis assay (magnification, ×20; scale, 100 μm). (C) several angiogenic lineage-specific genes were detected by real-time PCR. mRNA levels were tested in 5 independent samples. * P<0.05. (D) Expression of CD31 and VEGF in DPSCs and iDPSCs by immunofluorescence staining. All nuclei were stained with DAPI (blue). The cells were stained with antibody to VEGF (green), and CD31 (red). It was confirmed that DPSCs and iDPSCs co-expressed VEGF and CD31. magnification, ×20; scale bar, 100 μm. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.).
Figure 7. Angiogenic differentiation induction analysis. The cells were cultured in non-differentiated control medium (−) and angiogenic differentiation medium (+) for 2 weeks. (A) The cells showed a flat, paving-stone like shape after 14-day angiogenic induction (magnification, ×20; scale, 100 μm). (B) DPSCs and iDPSCs could form tubular network-like structure by Matrigel angiogenesis assay (magnification, ×20; scale, 100 μm). (C) several angiogenic lineage-specific genes were detected by real-time PCR. mRNA levels were tested in 5 independent samples. * P<0.05. (D) Expression of CD31 and VEGF in DPSCs and iDPSCs by immunofluorescence staining. All nuclei were stained with DAPI (blue). The cells were stained with antibody to VEGF (green), and CD31 (red). It was confirmed that DPSCs and iDPSCs co-expressed VEGF and CD31. magnification, ×20; scale bar, 100 μm. DPSCs – dental pulp stem cells; iDPSCs – inflammatory dental pulp stem cells; DAPI – 4′,6-diamidino-2-phenylindole. (Figure were created using Adobe Photoshop CS3, Adobe Systems Software Ireland, Ltd.). In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952