19 May 2021: Animal Study
Pretreatment with Propofol Reduces Pulmonary Injury in a Pig Model of Intestinal Ischemia-Reperfusion via Suppressing the High-Mobility Group Box 1 Protein (HMGB1)/Toll-Like Receptor 4 (TLR4)/Protein Kinase R (PKR) Signaling Pathway
Wen-yu Bian1BC, Ya-ping Chen2DF, Bo Xu3AE*, Jun Tang4AEDOI: 10.12659/MSM.930478
Med Sci Monit 2021; 27:e930478
Abstract
BACKGROUND: Propofol improves rodent pulmonary injury after intestinal ischemia-reperfusion (IIR). However, its effect and underlying mechanisms in large animals remain unclear. Here, we examined whether pretreatment with propofol could relieve lung injury during IIR in pigs, then investigated the underlying mechanism.
MATERIAL AND METHODS: A porcine model of IIR-induced lung injury was built by clamping the super mesenteric artery for 2 h and loosening the clamp for 4 h. Randomized grouping was used, and pigs were assigned to a sham-operated group, an IIR with saline pretreatment group, and an IIR with propofol pretreatment group. Pulmonary histopathologic changes, permeability, and oxygenation were assessed to evaluate the effect of propofol. We assessed levels of methane dicarboxylic aldehyde (MDA), superoxide dismutase (SOD), myeloperoxidase (MPO), high-mobility group box 1 protein (HMGB1), Toll-like receptor 4 (TLR4), and double-stranded RNA activated protein kinase R (PKR) to investigate the underlying mechanism.
RESULTS: IIR caused severe lung damage, including morphological changes, high permeability, airway resistance, low static compliance, hypoxemia, and acidemia. Pulmonary and plasma MDA content and MPO activity increased, whereas SOD activity decreased. The HMGB1/TLR4/PKR signaling pathway was activated following IIR. Pretreatment with propofol markedly attenuated lung injury (such as reducing the lung edema and permeability), increased MDA content and MPO activity, and restored SOD activity induced by IIR, accompanied by inhibiting the effect of the HMGB1/TLR4/PKR signaling pathway.
CONCLUSIONS: IIR caused acute lung injury in pigs. Pretreatment with propofol alleviated the lung injury, which was related to its suppression of the HMGB1/TLR4/PKR signaling pathway.
Keywords: acute lung injury, eIF-2 Kinase, HMGB1 Protein, Propofol, Toll-Like Receptor 4, Protective Agents, Swine
Background
Intestinal ischemia-reperfusion (IIR) always occurs in critical patients who experience ileus, shock, acute blood loss, or disseminated intravascular coagulopathy [1]. Generally, IIR can induce intestinal mucosal barrier injury, bacterial translocation, and remote organ damage [2,3]. As lung injury is a common type of remote organ damage caused by IIR [4], it is important to protect lung tissues during IIR. Recent reports have confirmed that high-mobility group box 1 protein (HMGB1) is essential in lung, heart, and intestinal injury after IR [5–7]. HMGB1, an endogenous ligand of Toll-like receptors 4 (TLR4), can activate TLR4 to induce inflammatory responses [8]. When activated by HMGB1, TLR4 further activates double-stranded RNA activated protein kinase R (PKR) [9–11], which is important in regulating the release of inflammatory factors (TNF-α, IL-1β, and IL-6) in acute lung injury (ALI) [12]. Thus, we inferred that HMGB1 participated in the process of IIR-induced ALI by activating the TLR4/PKR signal pathway.
Propofol is a short-acting intravenous anesthetic agent widely used in intensive care units [13]. It acts as a sedative and has antiemetic effects, immunomodulatory activity, and anxiolytic effects [14,15]. Previous studies have shown that propofol reduces lung injury induced by IIR [16,17]. Although propofol’s protective effect against IIR is mainly due to its ability to antagonize lipid peroxidation, the underlying mechanisms remain unclear. It was recently shown that propofol could protect against lipopolysaccharide (LPS)-induced ALI and ventilation-induced lung injury by inhibiting the expression of HMGB1 [18,19]. However, whether the protective effect of propofol on IIR-induced ALI is dependent on the HMGB1/TLR4/PKR signal pathway needs to be further studied.
In addition, previous studies on IIR were mainly based on rodent models [20–22]. Few studies used pigs even, even though the anatomical structure, physiological indicators, and immune system are similar between pigs and humans [23,24]. Therefore, our study assessed the lung-protection effect of propofol in miniature pigs with IIR and investigated its underlying mechanism.
Material and Methods
ANIMALS AND EXPERIMENTAL GROUPS:
All study procedures were approved by the Animal Care and Ethics Committee on Experimental Animals of Pudong Hospital (Fudan University, Shanghai, China). Male miniature pigs (Shanghai Jiagan Biotechnology Co., Ltd) weighing 9.0–12.4 kg were adapted to the laboratory environment for 1 week before experiments began. Animals were randomly assigned to a sham group, an IIR+normal saline (NS) group, and an IIR+propofol (P) group (8 per group) according to a random number table.
The flow chart is shown in Figure 1. After anesthesia induction (15 min), all the animals received intubation. In the IIR+P group, pigs received a continuous injection of propofol (AstraZeneca, Italy) 10 mg/kg/h from 10 min before clamping the SMA to the end of the operation. Pigs in the IIR+NS group received an equal amount of normal saline at the same time as the IIR+P group. Pulmonary static compliance and airway resistance were measured at baseline (T0), 2 h after ischemia (T1), and 2 h (T2) and 4h (T3) after reperfusion. At the end of the reperfusion, arterial blood gases were analyzed, after which all the pigs were euthanized. Then, pulmonary wet-to-dry weight ratio and permeability index were measured. In addition, the right upper lungs were harvested for HE staining. MPO activity, SOD activity, and MDA levels in the blood and the lung tissue were measured. Western blot analysis was used to assess the expression of HMGB1, TLR4, and PKR protein.
INTESTINAL ISCHEMIA-REPERFUSION PROTOCOL:
Following overnight fasting and free access to drinking water, the pigs were intramuscularly injected with ketamine (Hengrui Medicine, Jiangsu, China) and atropine sulfate (CR Double, China). Then, the trachea was intubated after anesthesia induction by intravenous injection of ketamine, fentanyl (Yichang Humanwell Pharmaceutical CO., China), and rocuronium bromide (N. V. ORGANON, USA). Sevoflurane (Baxter Healthcare Co., USA) and rocuronium bromide were used during general anesthesia. The left femoral artery and femoral vein were cannulated to monitor arterial pressure, collect blood samples, and for intravenous infusion. Electrocardiogram, arterial pressure, and esophageal temperature were continuously monitored during the operation. A crystalloid solution was infused to compensate for body fluid loss.
After stabilization of the respiratory and circulatory systems under anesthesia, pigs underwent surgery on warming pads. A laparotomy was performed to construct the IIR model. We separated abdominal tissue to occlude the superior mesenteric artery (SMA) using a microvascular clamp. During the next 2 h, humid sterile cotton balls were placed on the laparotomy incision to reduce fluid loss. Then, we removed the vascular clamp, and confirmed intestinal reperfusion by the pulsation of the superior mesenteric artery. A change in bowel loops color from dark purple to dark red 5 min after unclamping the SMA indicated that the model was successfully established. Pigs in which the model was not successfully established were not included in the experiment. The SMA was exposed but not occluded in the sham group. After 4 h of intestinal reperfusion, all the pigs were euthanized for further study.
ARTERIAL BLOOD GASES:
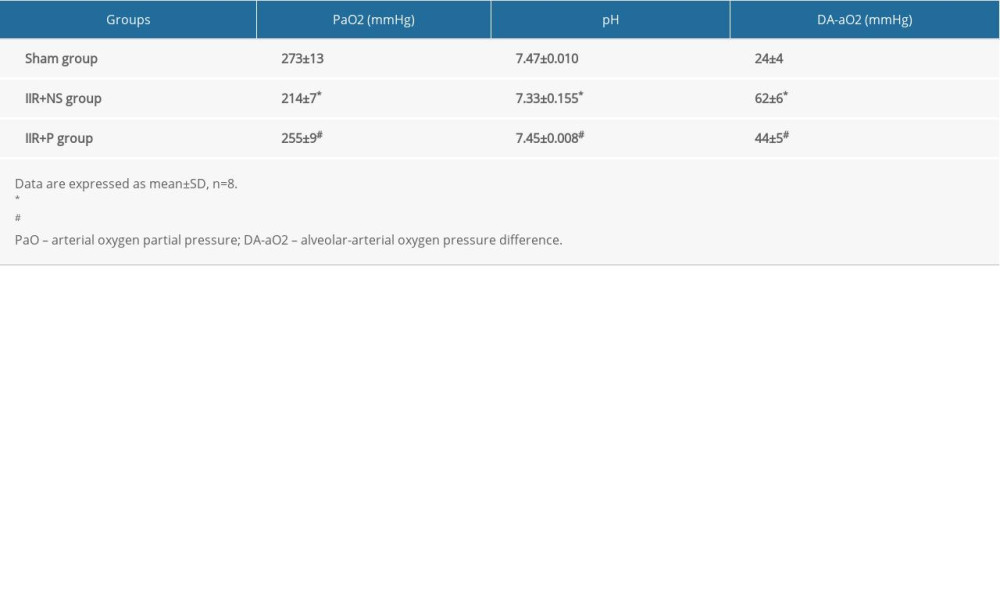
Before being euthanized, 0.5 ml of arterial blood was drawn for blood gas analysis (ABL800; Radiometer, Copenhagen, Denmark), which included PaO2, PaCO2, and pH. The alveolar-arterial oxygen pressure difference (DA-aO2) was calculated according to the following formula: DA-aO2=(PiO2- PaCO2×1/R)-PaO2. In this formula, PiO2=FiO2×(barometric pressure-47), and R is the respiratory quotient (ie, the volume of the evolved carbon dioxide volume to the volume of the absorbed oxygen) [24].
PULMONARY STATIC COMPLIANCE AND AIRWAY RESISTANCE:
After the animals were anesthetized, the pulmonary static compliance and airway resistance were measured using the electrocardiomonitor’s respiratory function module (PRO-400V2, 730PRO, General Electric Co.) at baseline (T0), 2 h after ischemia (T1), and at 2 h (T2) and 4 h (T3) after reperfusion. At the time of measurement, the flow velocity–pressure sensor used for children was connected in series between the ventilator and the airway intubation tubes. Muscle relaxants were administered before the test to prevent the effects of the experimental animals’ respiratory movement on pulmonary compliance and airway resistance.
PULMONARY WET-TO-DRY WEIGHT RATIO AND PERMEABILITY INDEX:
At the end of the experiment, pigs were sacrificed using intracardiac thiopental sodium (50 mg/kg) [25]. The chest was opened, and the proximal artery was clamped to prevent blood from entering the lung. Then, 10 ml/kg normal saline at 4°C was infused into the right lung’s bronchoalveolar space and harvested using a sterile centrifuge tube, and each animal received bronchoalveolar lavage 3 times. The bronchoalveolar lavage fluid (BALF) was centrifuged at 3000 rpm. The supernatant was used to detect protein levels using a BALF protein assay kit (Nanjing Jiancheng Biology Engineering Institute, Nanjing, China). Serum protein content was determined using a protein assay kit obtained from Nanjing Jiancheng Biology Engineering Institute. The pulmonary permeability index (PPI) was calculated using the following formula: PPI=BALF protein/serum protein.
At the same time, the left upper lobes of the lungs were removed and weighed using a precision balance (wet weight). After being dried for 24 h at 95°C, they were reweighed (dry weight). Before and after drying, the ratio of the lung weight was calculated as an index of pulmonary edema [19].
HISTOPATHOLOGIC EXAMINATION:
The right upper lungs were harvested 4 h after reperfusion, fixed in 10% formaldehyde, and embedded in paraffin to make sections, which were stained with hematoxylin and eosin (HE staining) for light microscope analysis. Two blinded pathologists scored the histopathologic injury. The criteria for lung histopathologic injury score were according to a previous report [26]. The score of normal lung tissue was zero, slight inflammatory injury was 1, mild to moderate inflammatory injury without damage to the lung architecture was 2, moderate inflammatory injury with alveolar septae thickening was 3, moderate to severe inflammatory injury (formation of nodules or areas of pneumonitis) was 4, and severe inflammatory injury (total obliteration of the normal lung tissue in the field) was 5.
DETECTION OF MYELOPEROXIDASE ACTIVITY IN LUNG TISSUES AND BLOOD:
Lung tissues were homogenized, and the supernatants were used to assess pulmonary myeloperoxidase (MPO) activity with an MPO detection kit (Nanjing Jiancheng Biological Engineering Institute) according to the manufacturer’s instructions. The MPO activity in blood was also measured using the MPO detection kit.
DETECTION OF MDA CONTENT AND SOD ACTIVITY IN LUNGS AND BLOOD:
Superoxide dismutase (SOD) activity and methane dicarboxylic aldehyde (MDA) content in lungs and blood were measured using a kit from Nanjing Jiancheng Biological Engineering Institute. The experimental procedure was performed according to manufacturer’s instructions.
DETECTION OF HMGB1/TLR4/PKR SIGNAL PATHWAY IN LUNGS:
HMGB1/TLR4/PKR pathway-related proteins were measured by western blot. The BCA kit (71285, NOVAGEN, Darmstadt, Germany) was used to evaluate the protein concentration. We mixed the protein sample (30 μg) with 10% SDS gel buffer at a 1: 1 ratio and then put it in 95°C water for 5 min to degrade the protein.
The samples were transferred to PVDF membranes (Bio-Rad, Hercules, CA, USA), and 5% nonfat dry milk was used to block the membrane at 4°C. Then, the PVDF membranes were incubated with primary antibodies against HMGB1 (1: 500, Biorbyt, Cambridge, UK), TLR4 (1: 500, Biorbyt, Cambridge, UK), PKR (1: 2000, Abcam, Cambridge, UK), and GAPDH (1: 1000, Santa Cruz, CA, USA) at 4°C for 12 h. After washing, the membranes were incubated with corresponding secondary antibodies (1: 2000, Santa Cruz, CA, USA) for 2 h at room temperature. The protein was colored for 3–5 min with ECL luminescent substrates (Beyotime, Jiangsu, China) after washing. GAPDH, as an invariant control, was also measure by western blot.
STATISTICAL ANALYSIS:
SPSS 26.0 software (IBM Corp., Armonk, NY, USA) was used for analyzing study data. Results are presented as the mean±standard deviation (SD). Data were evaluated with the
Results
PULMONARY GAS EXCHANGE:
Arterial PaO2 and pH were significantly lower and alveolar-arterial oxygen pressure difference (DA-aO2) was higher in the IIR+NS group than in the sham group (Table 1, P<0.01). Pretreatment with propofol significantly increased oxygenation. Arterial PaO2 and pH were significantly higher, whereas DA-aO2 was lower in the propofol +IIR group compared to the IIR+NS group (Table 1, P<0.01).
PULMONARY STATIC COMPLIANCE AND AIRWAY RESISTANCE:
Pulmonary static compliance decreased in a time-dependent manner in the IIR+NS group, whereas it was maintained at a steady level in the sham group. In the IIR+NS group, static lung compliance decreased at 2 h after reperfusion (T2) (Figure 2, P<0.05) and continued to decrease until 4 h after reperfusion (T3) (Figure 2, P<0.01) compared with the sham group at the same time points. Propofol partially reversed the decrease of static lung compliance induced by IIR at T3 (Figure 2, P<0.05).
Pulmonary airway resistance increased in a time-dependent manner in the IIR+NS group, whereas it was maintained at a steady level in the sham group. In the IIR+NS group, airway resistance began to increase at T2 (Figure 2, P<0.05) and continued to increase at T3 (P<0.01), compared with the sham group at the same time points. Propofol inhibited the increase of airway resistance induced by IIR at T3 (Figure 1, P<0.05).
PULMONARY PERMEABILITY:
The indicators of pulmonary edema included wet-to-dry lung ratio (W/DR) and the permeability index. The W/DR was significantly higher in the IIR+NS group compared to the sham group (Figure 3, P<0.01), and propofol reduced the change in the W/DR induced by IIR (Figure 3, P<0.05). Similarly, the permeability index was higher in the IIR+NS group than in the sham group (Figure 3, P<0.01), and propofol significantly decreased the permeability index during IIR (Figure 3, P<0.01).
HISTOLOGICAL CHANGE IN LUNG TISSUE:
As shown in Figure 4, histological analysis showed that lung tissues were clear, with a complete alveolar wall, no interstitial exudation, and normal small blood vessels without bleeding or edema (Figure 4A1, 4A2). Intestinal ischemia for 2 h and reperfusion for 4 h caused severe damage to lung tissues, as indicated by alveolar collapse, a widened alveolar septum, small blood vessel bleeding, and neutrophil infiltration (Figure 4B1, 4B2). Pretreatment with propofol reduced IIR-induced lung damage, which included decreasing pulmonary interstitial hemorrhage, edema, and neutrophil infiltration (Figure 4C1, 4C2). Also, the histopathologic injury score revealed similar results (Figure 4D). The score of the IIR+NS group was higher than that of the sham group, while propofol pretreatment significantly reduced the score compared to the normal saline pretreatment group (Figure 4D P<0.05 IIR+P group vs IIR+P group).
CHANGE OF MPO IN LUNGS AND BLOOD:
MPO activity in lung tissues and plasma increased in the IIR+NS group compared to the sham group (Figure 5, P<0.01). Pulmonary and plasma MPO activity decreased in the IIR+P group at the end of reperfusion compared to the IIR+NS group (Figure 5, P<0.05).
CHANGE IN REACTIVE OXYGEN SPECIES IN LUNGS AND BLOOD:
IIR increased the MDA levels and decreased the SOD activity both in the lungs and in blood (Figure 6, P<0.01 IIR+NS group vs sham group). Pretreatment with propofol partially reversed the changes of MDA levels and SOD activity (Figure 6, P<0.05 IIR+P group vs IIR+NS group).
PROPOFOL INHIBITED PROTEIN EXPRESSION OF HMGB1, TLR4, AND PKR IN LUNG TISSUE:
To evaluate if the HMGB1/TLR4/PKR signaling pathway had a role in the effect of propofol on lung damage during IIR, our study used western blot analysis to detect the protein expression of HMGB1, TLR4, and PKR. IIR elevated the protein levels of HMGB1, TLR4, and PKR in the lung tissue (Figure 7, P<0.01 IIR+NS group vs sham group). Propofol (10 mg/kg/h) significantly prevented the increase in protein expression of HMGB1, TLR4, and PKR induced by IIR (Figure 7, P<0.05 IIR+P group vs IIR+NS group). The HMGB1/TLR4/PKR signaling pathway could have a role in the protective effect of propofol on pulmonary injury after IIR.
Discussion
This study showed that propofol protects against pulmonary injury in a pig model of IIR, which was consistent with previous reports in rodents [27,28]. Propofol markedly reduced neutrophil activation and decreased oxidative stress induced by IIR, accompanied by inhibiting the HMGB1/TLR4/PKR signaling pathway.
Rodents are commonly used as a model to study acute lung injury induced by IIR [20,21]. Recently, large-animal models have been increasingly used to study IIR. Compared with rodents, it is easier to develop safe preclinical protocols directly transferable to humans using large animals [29]. Among the large animals, pigs are the ideal model for human IIR research because of their similarity in size, physiology, immunology [25], and organ development [30]. Furthermore, there is a strong morphological and functional resemblance of the organ systems between humans and pigs, including the intestines [31]. In the present study, we selected pigs for establishing an animal model of IIR to accurately assess the protective effects propofol against lung tissue damage induced by IIR. Our laboratory’s work further demonstrated that propofol could alleviate lung injury caused by IIR in a pig model. Consistent with propofol’s beneficial effects in rodents, propofol improved pulmonary oxygenation, decreased airway resistance and pulmonary microvascular permeability, and restored the pathological changes associated with lung injury in pigs exposed to IIR [20,21].
IIR induces several functional changes in the body. Firstly, damage to the intestinal mucosal barrier results in mucosal malabsorption and undernutrition [32,33], which, in turn, may lead to the invasion of enteric bacteria into lymph nodes, spleen, and the whole body through the injured mucosal barrier, thus resulting in systemic inflammatory response syndrome (SIRS) and triggering the release of inflammatory mediators [34,35]. The inflammatory mediators can cause remote organ damage, especially acute lung injury [36,37]. The mechanism underlying lung injury during IIR is complex and not yet fully clarified. IIR can cause cell membrane phospholipid degradation, leading to chemokine release, such as leukotriene, platelet-activating factor, and complement [38]. Chemokines act as attractants, leading to the accumulation of neutrophils in vascular and lung tissues and the release of oxygen-free radicals and proteolytic enzymes, which results in endothelial damage, microvascular leakage, and edema in the lung [39]. The results of the present study showed that IIR significantly increased the polymorphonuclear (PMN) cells infiltration and oxygen-free radical damage in lung tissue. In addition, myeloperoxidase (MPO) activity, malondialdehyde (MDA) content increased, and superoxide dismutase (SOD) activity decreased in the IIR group. MPO is a marker enzyme of polymorphonuclear (PMN) cells, and increased activity of MPO in tissues indicates an increase of PMN infiltration [40]. Furthermore, MDA is one of the major products of membrane lipid peroxidation, and its content is proportional to the degree of damage induced by oxygen-free radicals [41]. In addition, SOD is an oxygen-free radical-scavenging enzyme that protects cells from damage [42]. Large amounts of oxygen-free radicals activated by PMN and lipid peroxidation caused by reactive oxygen species are the pathological indicators of lung injury after IR [43]. SOD is a kind of reactive oxygen species (ROS), which are always generated in the development of oxidative stress induced by ischemia-reperfusion injury [44].
Propofol has a chemical structure similar to that of vitamin E and has comparable antioxidant properties [45]. When IIR occurs, lipid oxidation reaction in the cell membrane alters the permeability of alveolar capillaries [46]. Propofol has been reported to interfere with peroxynitrite to eliminate reactive oxygen species (ROS) and protect lung tissue [47]. Previous studies have shown that propofol reduces lung injury induced by IIR [16,17]. Similar to these previous results, we found that pretreatment with propofol markedly attenuated lung injury (such as reducing the lung edema and permeability), increased MDA content and MPO activity, and restored SOD activity reduced by IIR. In addition, we evaluated the effect of propofol on the HMGB1/TLR4/PKR signal pathway. HMGB1 is a nonhistone chromatin protein present in all cell types [48]. It mainly regulates gene transcription as a critical damage-associated molecular pattern molecule (DAMP) after being released. Excessive amounts of extracellular HMGB1 can cause tissue injury and organ dysfunction in the pathogenesis of many different diseases of both sterile and infectious origin [49–51], such as lung injury induced by ischemia-reperfusion injury [52,53]. After activation, HMGB1 can bind to its receptor TLR4 to activate it [8]. Similar to previous studies, the present study also confirmed that IIR induced the activation of HMGB1, followed by activation of TLR4. PKR is a widely expressed serine and threonine protein kinase, which was first confirmed as an anti-viral protein induced by interferon (IFN) [54]. Li et al reported that inhibition of PKR could regulate lung protein permeability, the release of proinflammatory cytokines (TNF-α, IL-1β, and IL-6), and lung apoptosis in LPS-induced lung injury [12]. Studies have shown that PKR is normally activated TLR4 [55]. In our study, we found the expression of PKR was elevated in lung tissue of the IIR model pigs. Furthermore, propofol could inhibit the expression of HMGB1/TLR4/PKR, which participates in the protective effect of propofol against pulmonary injury during IIR.
Conclusions
Our data showed that propofol used in anesthesia has a protective effect during IIR, as indicated by changes in the histologic and biochemical markers in lung injury. The effect of propofol can be mediated by the inhibition of oxidant generation and infiltration of neutrophils with the involvement of the HMGB1/TLR4/PKR signaling pathway.
Figures
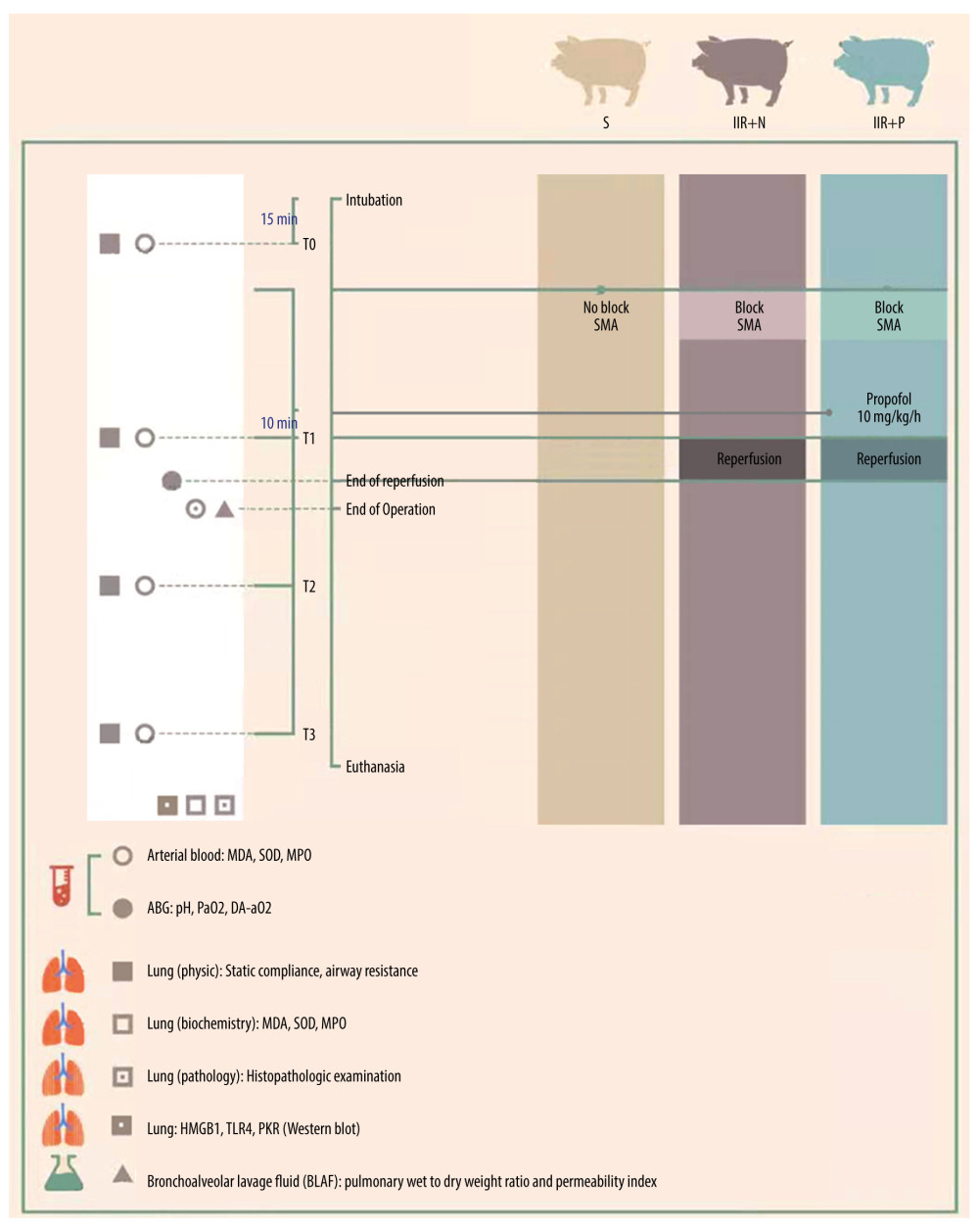 Figure 1. Study flow chart.
Figure 1. Study flow chart. 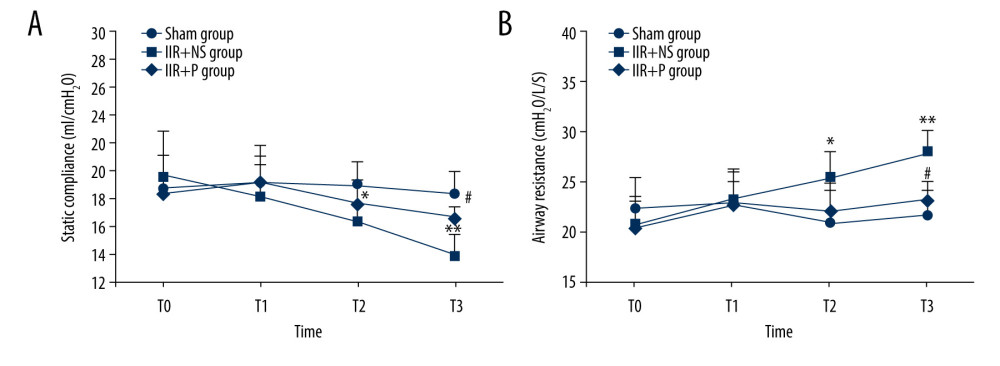 Figure 2. (A, B) Pulmonary static compliance and airway resistance at T0 (15 min after intubation), T1 (2 h after ischemia), T2 (2 h after reperfusion), and T3 (4 h after reperfusion) in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 2. (A, B) Pulmonary static compliance and airway resistance at T0 (15 min after intubation), T1 (2 h after ischemia), T2 (2 h after reperfusion), and T3 (4 h after reperfusion) in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. 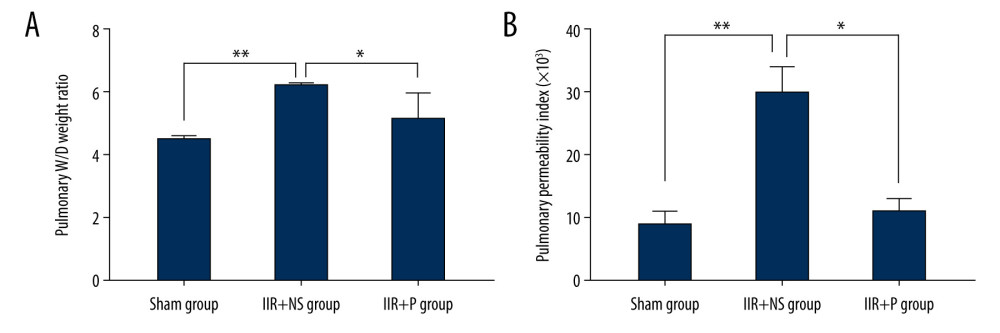 Figure 3. (A, B) Pulmonary wet/dry (W/D) weight ratio and pulmonary permeability index in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 3. (A, B) Pulmonary wet/dry (W/D) weight ratio and pulmonary permeability index in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. 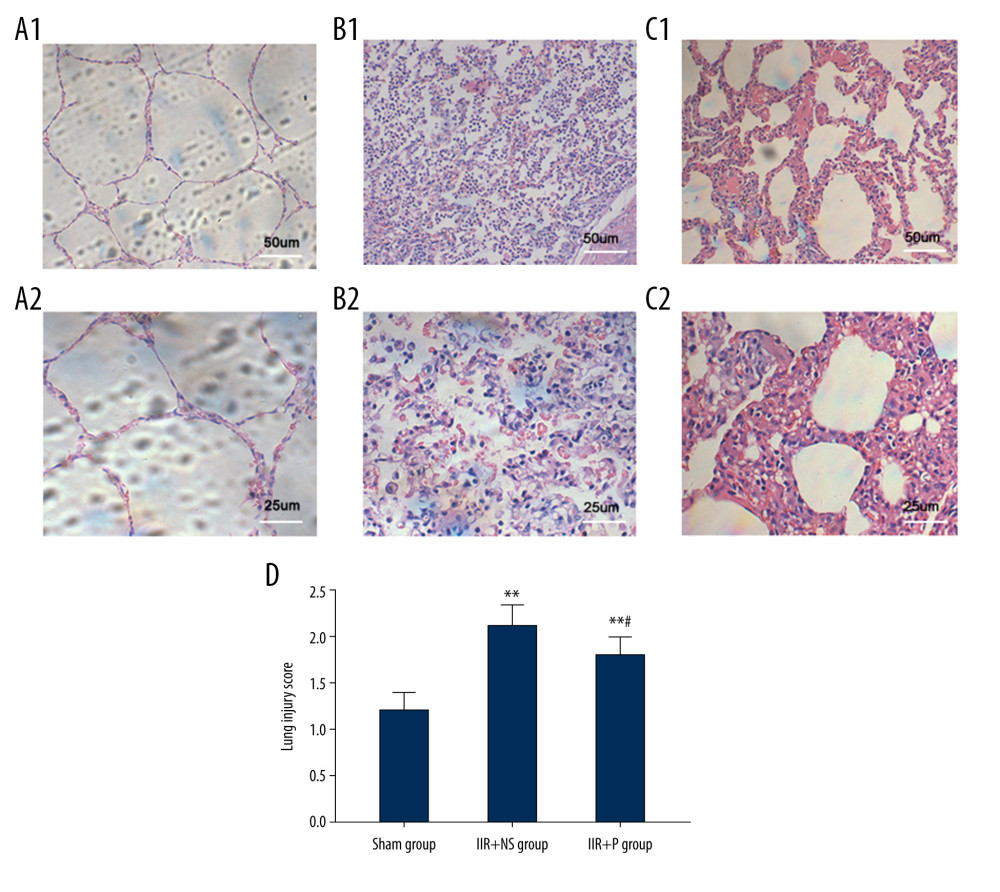 Figure 4. Histopathologic sections of the lung tissue. The sections of a sham IIR pig (A1, A2), a saline-pretreated IIR pig (B1, B2), and a propofol-pretreated IIR pig (C1, C2). The lung tissue of the pig with IIR shows severe damage, with alveolar collapse, a widened alveolar septum, small blood vessel bleeding, and neutrophil infiltration. Propofol significantly attenuated the changes induced by IIR. A1-C1 are displayed with 100 times magnification, while A2-C2 are displayed with 200 times magnification. The lung histopathologic score is displayed in panel D. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. a, ×100; b, ×200. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 4. Histopathologic sections of the lung tissue. The sections of a sham IIR pig (A1, A2), a saline-pretreated IIR pig (B1, B2), and a propofol-pretreated IIR pig (C1, C2). The lung tissue of the pig with IIR shows severe damage, with alveolar collapse, a widened alveolar septum, small blood vessel bleeding, and neutrophil infiltration. Propofol significantly attenuated the changes induced by IIR. A1-C1 are displayed with 100 times magnification, while A2-C2 are displayed with 200 times magnification. The lung histopathologic score is displayed in panel D. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. a, ×100; b, ×200. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. 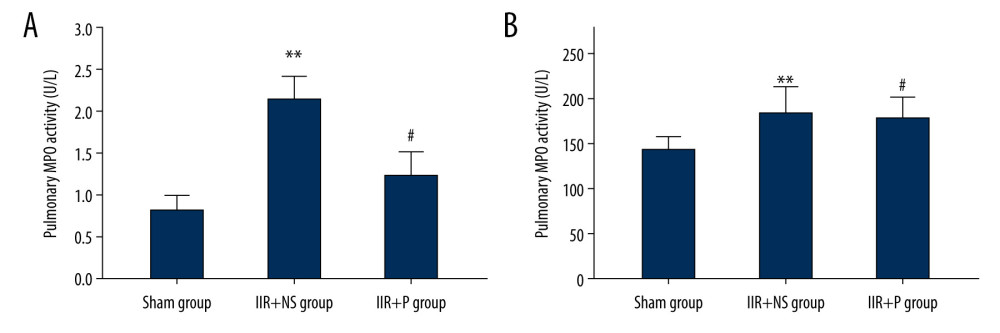 Figure 5. (A, B) Pulmonary and plasma myeloperoxidase (MPO) activity. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 5. (A, B) Pulmonary and plasma myeloperoxidase (MPO) activity. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. 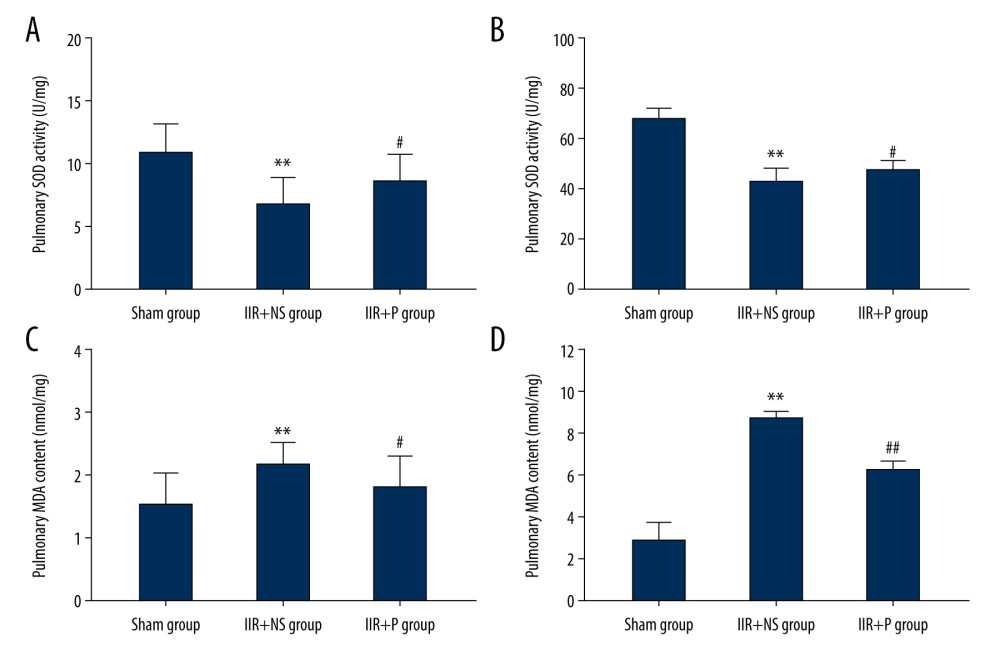 Figure 6. (A–D) Pulmonary and plasma superoxide dismutase (SOD) activity and malondialdehyde (MDA) levels. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 6. (A–D) Pulmonary and plasma superoxide dismutase (SOD) activity and malondialdehyde (MDA) levels. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. 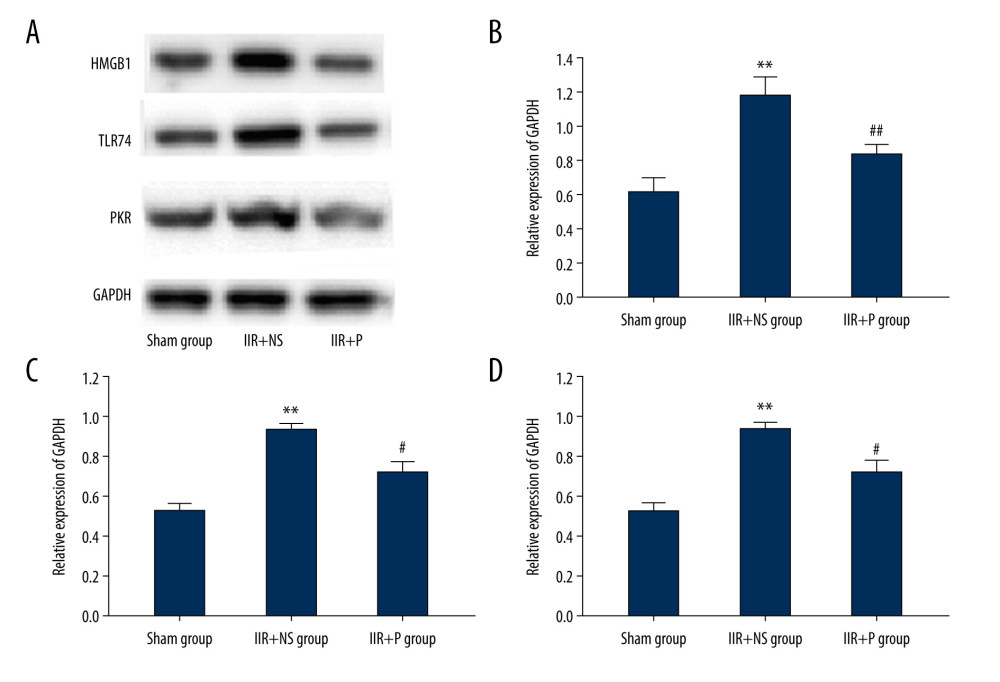 Figure 7. (A–D) Protein expression level of HMGB1/TLR4/PKR by western blot, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). ** P<0.01 compared with the sham group. # P<0.05 compared with IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 7. (A–D) Protein expression level of HMGB1/TLR4/PKR by western blot, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). ** P<0.01 compared with the sham group. # P<0.05 compared with IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. References
1. Vollmar B, Menger MD, Intestinal ischemia/reperfusion: Microcirculatory pathology and functional consequences: Langenbecks Arch Surg, 2011; 396; 13-29
2. Hassoun HT, Kone BC, Mercer DW, Post-injury multiple organ failure: The role of the gut: Shock, 2001; 15; 1-10
3. Cavriani G, Oliveira-Filho RM, Trezena AG, Lung microvascular permeability and neutrophil recruitment are differently regulated by nitric oxide in a rat model of intestinal ischemia-reperfusion: Eur J Pharmacol, 2004; 494; 241-49
4. Ware LB, Matthay MA, The acute respiratory distress syndrome: N Engl J Med, 2000; 342; 1334
5. Yang C, Yang W, He Z, Kaempferol improves lung ischemia-reperfusion injury via antiinflammation and antioxidative stress regulated by SIRT1/HMGB1/NF-κB axis: Front Pharmacol, 2020; 10; 1635
6. Dong LY, Chen F, Xu M, Quercetin attenuates myocardial ischemia-reperfusion injury via downregulation of the HMGB1-TLR4-NF-kappaB signaling pathway: Am J Transl Res, 2018; 10(5); 1273-83
7. Wang J, He GZ, Wang YK, TLR4-HMGB1-, MyD88- and TRIF-dependent signaling in mouse intestinal ischemia/reperfusion injury: World J Gastroenterol, 2015; 21(27); 8314-25
8. Lotze MT, Tracey KJ, High-mobility group box 1 protein (HMGB1): Nuclear weapon in the immune arsenal: Nat Rev Immunol, 2005; 5(4); 331-42
9. Li R, Shang Y, Yu Y, High-mobility group box 1 protein participates in acute lung injury by activating protein kinase R and inducing M1 polarization: Life Sci, 2020; 246; 117415
10. Asakura Y, Fujiwara Y, Kato N, Serine/threonine kinase PKR: A sentinel kinase that discriminates a signaling pathway mediated by TLR4 from those mediated by TLR3 and TLR9: Am J Hematol, 2007; 82(7); 640-42
11. Webster SJ, Ellis L, O’Brien LM: Microbes Infect, 2016; 18; 472-83
12. Li Y, Xiao J, Tan Y, Inhibition of PKR ameliorates lipopolysaccharide-induced acute lung injury by suppressing NF-κB pathway in mice: Immunopharmacol Immunotoxicol, 2017; 39(4); 165-72
13. Lewis SR, Schofield-Robinson OJ, Alderson P, Propofol for the promotion of sleep in adults in the intensive care unit: Cochrane Database Syst Rev, 2018; 1; CD012454
14. Bhakta P, Ghosh BR, Singh U, Incidence of postoperative nausea and vomiting following gynecological laparoscopy: A comparison of standard anesthetic technique and propofol infusion: Acta Anaesthesiol Taiwan, 2016; 54; 108-13
15. Vasileiou I, Xanthos T, Koudouna E, Propofol: A review of its non-anaesthetic effects: Eur J Pharmacol, 2009; 605; 1-8
16. Vasileiou I, Kalimeris K, Nomikos T, Propofol prevents lung injury following intestinal ischemia-reperfusion: J Surg Res, 2012; 172; 146-52
17. Zhao W, Zhou S, Yao W, Propofol prevents lung injury after intestinal ischemia-reperfusion by inhibiting the interaction between mast cell activation and oxidative stress: Life Sci, 2014; 108; 80-87
18. Wang T, Wei XY, Liu B, Effects of propofol on lipopolysaccharide-induced expression and release of HMGB1 in macrophages: Braz J Med Biol Res, 2015; 48(4); 286-91
19. Feng Z, Wang JW, Wang Y, Propofol protects lung endothelial barrier function by suppression of high-mobility group box 1 (HMGB1) release and mitochondrial oxidative damage catalyzed by HMGB1: Med Sci Monit, 2019; 25; 3199-211
20. Chai D, Zhang L, Xi S, Nrf2 activation induced by Sirt1 ameliorates acute lung injury after intestinal ischemia/reperfusion through NOX4-mediated gene regulation: Cell Physiol Biochem, 2018; 46(2); 781-92
21. Zhu Q, He G, Wang J, Down-regulation of toll-like receptor 4 alleviates intestinal ischemia reperfusion injury and acute lung injury in mice: Oncotarget, 2017; 8(8); 13678-89
22. Iida T, Takagi T, Katada K, Rapamycin improves mortality following intestinal ischemia-reperfusion via the inhibition of remote lung inflammation in mice: Digestion, 2015; 92(4); 211-19
23. Rothkötter HJ, Anatomical particularities of the porcine immune system – a physician’s view: Dev Comp Immunol, 2009; 33(3); 267-72
24. Yandza T, Tauc M, Saint-Paul MC, The pig as a preclinical model for intestinal ischemia-reperfusion and transplantation studies: J Surg Res, 2012; 178; 807-19
25. Demir T, Turgut B, Ozercan I, Trimetazidine for prevention of induced ischemia and reperfusion of guinea pig retina: Clin Ophthalmol, 2010; 4; 21-26
26. Dong WW, Liu YJ, Lv Z, Lung endothelial barrier protection by resveratrol involves inhibition of HMGB1 release and HMGB1-induced mitochondrial oxidative damage via an Nrf2-dependent mechanism: Free Radic Biol Med, 2015; 88; 404-16
27. Vasileiou I, Kalimeris K, Nomikos T, Propofol prevents lung injury following intestinal ischemia-reperfusion: J Surg Res, 2012; 172(1); 146-52
28. Zhao W, Zhou S, Yao W, Propofol prevents lung injury after intestinal ischemia-reperfusion by inhibiting the interaction between mast cell activation and oxidative stress: Life Sci 17, 2014; 108(2); 80-87
29. Kirk AD, Crossing the bridge: Large animal models in translational transplantation research: Immunol Rev, 2003; 196; 176
30. Rothkötter HJ, Anatomical particularities of the porcine immune system: A physician’s view: Dev Comp Immunol, 2009; 33; 267
31. Ibrahim Z, Busch J, Awwad M, Selected physiologic compatibilities and incompatibilities between human and porcine organ systems: Xenotransplantation, 2006; 13; 488
32. Kong SE, Blennerhassett LR, Heel KA, Ischaemia-reperfusion injury to the intestine: Aust N Z J Surg, 1998; 68(8); 554-61
33. Carden DL, Granger DN, Pathophysiology of ischaemia reperfusion injury: J Pathol, 2000; 190; 255
34. Aksöyek S, Cinel I, Avlan D, Intestinal ischemic preconditioning protects the intestine and reduces bacterial translocation: Shock, 2002; 18; 476
35. Berg RD, Bacterial translocation from the gastrointestinal tract: Adv Exp Med Biol, 1999; 473; 11
36. Carden DL, Granger DN, Pathophysiology of ischaemia-reperfusion injury: J Pathol, 2000; 190; 255-66
37. Harward TR, Brooks DL, Flynn TC, Multiple organ dysfunction after mesenteric artery revascularization: J Vasc Surg, 1993; 18; 459-67
38. Kostopanagiotou G, Avgerinos E, Costopanagiotou C, Acute lung injury in a rat model of intestinal ischemia-reperfusion: The potential time depended role of phospholipases A(2): J Surg Res, 2008; 147; 108-16
39. Eltzschig HK, Eckle T, Ischemia and reperfusion-from mechanism to translation: Nat Med, 2011; 17(11); 1391-401
40. Kinkade JM, Pember SO, Barnes KC, Differential distribution of distinct forms of myeloperoxidase in different azurophilic granule subpopulations from human neutrophils: Biochem Biophys Res Commun, 1983; 114(1); 296-303
41. Pryor WA, Stanley JP, Letter A suggested mechanism for the production of malondialdehyde during the autoxidation of polyunsaturated fatty acids. Nonenzymatic production of prostaglandin endoperoxides during autoxidation: J Org Chem, 1975; 40(24); 3615-17
42. Hayyan M, Hashim MA, Al Nashef IM, Superoxide ion: Generation and chemical implications: Chem Rev, 2016; 116(5); 3029-85
43. Barletta KE, Ley K, Mehrad B, Regulation of neutrophil function by adenosine: Arterioscler Thromb Vasc Biol, 2012; 32(4); 856-64
44. Zhang J, Wang X, Vikash V, ROS and ROS-mediated cellular signaling: Oxid Med Cell Longev, 2016; 2016; 4350965
45. Murphy PG, Myers DS, The antioxidant potential of propofol (2,6-diisopropylphenol): Br J Anaesth, 1992; 68; 613-18
46. Vasileiou I, Kalimeris K, Nomikos T, Propofol prevents lung injury following intestinal ischemia-reperfusion: J Surg Res, 2012; 172(1); 146-52
47. Zhao W, Zhou S, Yao W, Propofol prevents lung injury after intestinal ischemia-reperfusion by inhibiting the interaction between mast cell activation and oxidative stress: Life Sci, 2014; 108(2); 80-87
48. Yang H, Antoine DJ, Andersson UM, The many faces of HMGB1: molecular structure-functional activity in inflammation, apoptosis, and chemotaxis: J Leukocyte Biol, 2013; 93; 865-73
49. Yang H, Tracey KJ, Targeting HMGB1 in inflammation: Biochim Biophys Acta, 2010; 1799; 149-56
50. Andersson U, Tracey KJ, HMGB1 is a therapeutic target for sterile inflammation and infection: Ann Rev Immunol, 2011; 29; 139-62
51. Andersson U, Yang H, Harris H, Extracellular HMGB1 as a therapeutic target in inflammatory diseases: Expert Opin Ther Targets, 2018; 22; 263-77
52. Yang Z, Deng Y, Su D, TLR4 as receptor for HMGB1-mediated acute lung injury after liver ischemia/reperfusion injury: Lab Invest, 2013; 93; 792-800
53. Yu Z, Tong Y, Zhang R, Saquinavir ameliorates liver warm ischemia-reperfusion-induced lung injury via HMGB-1- and P38/JNK-mediated TLR-4-dependent signaling pathways: Mediators Inflamm, 2017; 2017; 7083528
54. Kang R, Chen RC, Zhang QH, HMGB1 in health and disease: Mol Asp Med, 2014; 40; 1-116
55. Bat-Erdene U, Quan E, Chan K, Neutrophil TLR4 and PKR are targets of breast cancer cell glycosaminoglycans and effectors of glycosaminoglycan-induced APRIL secretion: Oncogenesis, 2018; 7; 45
Figures
 Figure 1. Study flow chart.
Figure 1. Study flow chart. Figure 2. (A, B) Pulmonary static compliance and airway resistance at T0 (15 min after intubation), T1 (2 h after ischemia), T2 (2 h after reperfusion), and T3 (4 h after reperfusion) in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 2. (A, B) Pulmonary static compliance and airway resistance at T0 (15 min after intubation), T1 (2 h after ischemia), T2 (2 h after reperfusion), and T3 (4 h after reperfusion) in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. Figure 3. (A, B) Pulmonary wet/dry (W/D) weight ratio and pulmonary permeability index in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 3. (A, B) Pulmonary wet/dry (W/D) weight ratio and pulmonary permeability index in the sham, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). * P<0.05, ** P<0.01. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. Figure 4. Histopathologic sections of the lung tissue. The sections of a sham IIR pig (A1, A2), a saline-pretreated IIR pig (B1, B2), and a propofol-pretreated IIR pig (C1, C2). The lung tissue of the pig with IIR shows severe damage, with alveolar collapse, a widened alveolar septum, small blood vessel bleeding, and neutrophil infiltration. Propofol significantly attenuated the changes induced by IIR. A1-C1 are displayed with 100 times magnification, while A2-C2 are displayed with 200 times magnification. The lung histopathologic score is displayed in panel D. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. a, ×100; b, ×200. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 4. Histopathologic sections of the lung tissue. The sections of a sham IIR pig (A1, A2), a saline-pretreated IIR pig (B1, B2), and a propofol-pretreated IIR pig (C1, C2). The lung tissue of the pig with IIR shows severe damage, with alveolar collapse, a widened alveolar septum, small blood vessel bleeding, and neutrophil infiltration. Propofol significantly attenuated the changes induced by IIR. A1-C1 are displayed with 100 times magnification, while A2-C2 are displayed with 200 times magnification. The lung histopathologic score is displayed in panel D. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. a, ×100; b, ×200. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. Figure 5. (A, B) Pulmonary and plasma myeloperoxidase (MPO) activity. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 5. (A, B) Pulmonary and plasma myeloperoxidase (MPO) activity. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. Figure 6. (A–D) Pulmonary and plasma superoxide dismutase (SOD) activity and malondialdehyde (MDA) levels. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 6. (A–D) Pulmonary and plasma superoxide dismutase (SOD) activity and malondialdehyde (MDA) levels. Propofol at 10 ml/kg or the same volume of saline was intravenously injected into pigs with a computerized infusion pump via the femoral vein 10 min before the start of reperfusion. Lung tissues and blood were collected from pigs at 4 h after reperfusion. Data are expressed as the mean±SD, n=8. ** P<0.01 compared with the sham group, # P<0.05 compared with the IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. Figure 7. (A–D) Protein expression level of HMGB1/TLR4/PKR by western blot, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). ** P<0.01 compared with the sham group. # P<0.05 compared with IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol.
Figure 7. (A–D) Protein expression level of HMGB1/TLR4/PKR by western blot, IIR+NS, and IIR+P groups. Data are expressed as the mean±SD (n=8). ** P<0.01 compared with the sham group. # P<0.05 compared with IIR+NS group. IIR – intestinal ischemia-reperfusion; NS – normal saline; P – propofol. In Press
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952