18 May 2021: Database Analysis
Cochlear Implant Outcomes in Prelingually Deafened Adults with and without Sound Deprivation: Are There Differences in Quality of Life?
Andrea Canale1ADE*, Francesco Macocco1BDEF, Drita Ndrev1BDEF, Giulia Gabella1BF, Gitana Scozzari2CE, Roberto Albera1ADE, Giancarlo Pecorari1DE, Andrea Albera3ABDEDOI: 10.12659/MSM.930232
Med Sci Monit 2021; 27:e930232
Abstract
BACKGROUND: Indications for cochlear implantation (CI) are constantly being updated, and with them, the audiometric results achieved by patients. Patient satisfaction should always be considered, even in patients with lower audiological results. The aim of the present study was to compare quality of life (QoL), self-perceived hearing benefit, and audiometric results between prelingually and postlingually deafened patients, with and without sound deprivation, after CI.
MATERIAL AND METHODS: The sample included 46 patients with bilateral sensorineural hearing loss: 22 postlingually deafened and 24 prelingually deafened, further subdivided into sound-deprived (n=10) and non-sound-deprived (n=14). Auditory performance was evaluated with pure tone audiometry, speech recognition scores (SRS), and self-perceived hearing benefit, whereas QoL was evaluated with 2 self-reported questionnaires (Comprehensive Cochlear Implant Questionnaire and World Health Organization Quality of Life-BREF).
RESULTS: Audiometric results were worse in the prelingually deafened than in the postlingually deafened group, and worse in the prelingually deafened patients with sound deprivation. There was no marked difference in perceived CI benefit or QoL between the 2 groups or within the 2 prelingually deafened subgroups. No correlation was found between SRS and duration of CI use or between QoL and SRS in the prelingually and postlingually deafened groups.
CONCLUSIONS: Our findings demonstrate better auditory performance for the postlingually deafened group and no differences in perceived QoL or benefit of CI between the groups. The sound-deprived patients had equal scores on the perceived QoL questionnaire. These analyses suggest that sound-deprived, prelingually deafened patients may benefit from CI.
Keywords: Cochlear Implants, Deafness, Quality of Life, Audiometry, Pure-Tone, Cochlear Implantation, Hearing, Hearing Loss, Sensorineural, Sound, Speech Discrimination Tests, Surveys and Questionnaires, young adult
Background
Until the mid-1990s, patients with prelingual deafness were not considered suitable candidates for cochlear implantation (CI). Studies conducted at the time showed that such patients could achieve improvements in the perception of environmental sounds but not in speech recognition [1,2]. Much has changed in the last 15 years with advances in surgery, anesthesiology, and CI technology. Modern devices provide sound processing and stimulation strategies that allow better speech understanding by facilitating central deciphering of CI stimulation of the auditory nerve [3]. Furthermore, audiological CI indication criteria have been expanded to include the prelingually deafened, as demonstrated in recent studies [4].
Especially in prelingually deafened candidates, there is wide variability in audiological outcomes, owing to the multiple factors at play: earlier implantation [5,6], severity [7,8] and age at onset of the hearing loss (prelingual or perilingual) [9], and etiology of the deafness. While all these factors except etiology play a minor role in postlingual deafness [10–12], they are of extreme importance for predicting audiometric outcome and speech recognition results in prelingually deafened patients.
Recent evidence supports the hypothesis that there is no correlation between audiological CI results and reported benefit in quality of life (QoL) [13,14]. This is true in prelingually deafened patients and has been documented in a recent review [4]. In such patients, CI is not unreasonable, but it must be accompanied by good counseling to explain what can be expected and explore what motivates the patient to undergo surgery. The review concluded that more data need to be collected and that future studies involving late-implanted, prelingually deafened adults should categorize patients by criteria of residual hearing, age at onset of hearing loss, and etiology. It is equally important to differentiate patients based on their response to QoL questionnaires.
The aim of the present study was to compare social and audiometric data between a group of late prelingually deaf CI patients, with and without sound deprivation, and a group of postlingual deaf CI patients to evaluate possible differences in subjective hearing benefit and perceived QoL.
Material and Methods
STATISTICAL ANALYSIS:
Categorical variables were reported as frequencies and percentages, whereas continuous variables were reported as means and standard deviations when distribution was normal (normality distribution was tested with the Shapiro-Wilk normality test), or as medians and interquartile ranges (IQRs) when distribution was not normal. Differences between the 2 groups were analyzed with a chi-square test for categorical data (with Fisher correction when needed), while continuous variables were analyzed using an independent
COMPLIANCE WITH ETHICAL STANDARDS:
The present study was an observational case series and all procedures performed were in accordance with the ethical standards of the institution and conducted according to the principles expressed in the Declaration of Helsinki.
All data were analyzed anonymously and in accordance with the ethical standards of the institution.
Results
The sample was divided into 2 groups: the PRE-LG group, composed of 24 patients (5 men and 19 women; mean age 41.5 years; range 20–55) and the POST-LG group, composed of 22 patients (12 men and 10 women; mean age 57.5 years; range, 26–75).
As regards socio-demographic data, we observed that in the PRE-LG group, 66.7% of patients were employed, 16.7% were unemployed, 16.7% were students, and 58.3% were unmarried, while in the POST-LG group, 59.1% were employed, 36.4% were retired, 4.6% unemployed, 18.2% single, and 63.6% were married. These differences were statistically significant (
The most common cause of hearing loss in the POST-LG group was Ménière’s disease (35.7%), followed by unknown etiology (21.4%), ototoxicity (14.3%), and post-infectious, post-traumatic, neuromuscular, and work-related causes (7.1% each). In the PRE-LG group, the deafness was secondary to infection (41.2%), genetic disease (35.3%), of unknown etiology (17.7%), and head trauma (5.9%). The difference in distribution of etiology was statistically significant (
Among the PRE-LG patients, 23 (95.8%) had received oralist rehabilitation with differences in duration of hearing aid fitting; 1 patient (4.2%) had complete auditory deprivation since birth. In this group, 10 patients (41.7%) received the implant in the sound-deprived ear, ie, without acoustic stimulation for over 15 years (D), and 14 patients (58.3%) continued to use their hearing aids until CI (S). In addition, 12 patients (50%) had unilateral CI, 5 patients (20.8%) had undergone bilateral CI, and 7 patients (29.2%) had bimodal stimulation.
In the POST-LG group, 3 patients (13.6%) had unilateral CI, 5 (22.7%) had undergone bilateral CI, and 14 patients (63.6%) had bimodal stimulation.
Table 1 lists SRS and audiometric results in the 2 groups. One patient (4.1%) in the PRE-LG group was unable to perform the speech recognition test. In the other patients, SRS ranged between 10% and 100%. Table 2 lists the distribution of patients in relation to SRS. In contrast, Table 3 lists SRS distribution in the PRE-LG D and S subgroups. The median SRS was 40% (range, 20%–80%) for subgroup D and 80% (range, 40–90%) for subgroup S (
At least 12 months elapsed between the surgery and questionnaire administration: the mean period was 43 months (range, 12–96). The percentage of respondents was 71% (17 patients) in the PRE-LG group and 64% (14 patients) in the POST-LG group.
There was a statistically significant difference between the 2 groups for only 3 of 28 items on the CCIQ questionnaire, in which responses are given on a 5-point Likert-like scale (never, rarely, sometimes, often, always). Concerning the item “I still have trouble conversing in rooms where there is an echo, like large auditoriums or gyms,” the median response was 2 (interquartile range [IQR], 1–3) for the PRE-LG group and 3 (range, 2–4) for the POST-LG group (
The relationships between the SRS and duration of CI use, as well as between the SRS and the perceived QoL in the PRE-LG and the POST-LG groups, were evaluated with a Spearman rank correlation (Figures 1–4). A strong negative correlation was found between SRS and mean QoL score for each patient in the POST-LG group (Spearman rho −0.60,
Discussion
Audiological improvement in late-implanted PRE-LG deafened patients is commonly considered worse than that achieved in POST-LG adults. Moreover, the wide range of audiometric performance that persists among PRE-LG deafened patients [4] is due to multiple prognostic factors, the foremost being oralist rehabilitation and sound deprivation in the ear that has undergone CI [17]. In the present study, the sample consisted of 24 PRE-LG deafened patients with and without a long duration of sound deprivation and 22 POST-LG deafened patients.
Our results showed that audiometric outcomes were poorer in the PRE-LG patients, with a wide difference in SRS within the group. Furthermore, subgroup analysis showed a difference in SRS (albeit not statistically significant) between the 2 groups (40% vs 80% for the D and S groups, respectively), compared to a median of 80% for the whole PRE-LG group.
This finding indicates that, in contrast to acquired hearing loss, outcomes are poorer in patients with congenital deafness in cases in which the ear that has undergone CI had sound deprivation [10,18]. An anomalous finding emerging from our study regards hearing performance: SRS did not correlate with the duration of use of the CI in the POST-LG and PRE-LG groups. The absence of a correlation in the POST-LG group is in line with previous data. Modern devices enable patients to achieve satisfying hearing results within very short rehabilitation times [18]. Studies have shown, however, that the prelingually deafened need more time and rehabilitation to reach similar outcomes [12,18]. No reliable correlation could be found because of the heterogeneous prognostic factors in the PRE-LG group. Auditory performance is correlated with rehabilitation benefit [13].
The questionnaire response rate was 71% in the PRE-LG group and 64% in the POST-LG group because for some patients, the questionnaires were too difficult and long to fill out. In our study, the responses to the CCIQ questionnaire were statistically significantly different between the 2 groups for only 3 of 28 items. Moreover, for 2 of those 3 items, the PRE-LG group responses indicated much more satisfaction than in the POST-LG group, even among those with better auditory performance. The median of the PRE-LG group response reveals that, despite the significantly lower SRS than that in the POST-LG group, these patients reported greater benefit in understanding speech, in environments with echo and when enjoying music. A plausible explanation is that postlingually deafened adults learned how to listen with normal binaural hearing in a room with echo and how to appreciate the melody in music.
We found no difference in the median QoL scores between the PRE-LG group and the POST-LG group. This lack of a significant difference clearly reveals how CI is important for prelingually deafened patients because it can enable them to reach a relatively higher QoL score despite the low SRS and increased social isolation that seemed to be indicated by our data: 58% of the PRE-LG group patients were unmarried and 16.7% were unemployed at the time of the questionnaire compilation. These findings are in line with previous studies and are a major reason why CI is increasingly proposed to prelingually deafened patients [9,19].
Analysis of the correlation between SRS and QoL in each group showed a weak positive correlation for the PRE-LG group. However, such a correlation was not statistically significant, indicating that QoL does not differ in this group with an SRS of 20% compared to patients with an SRS of 100%. In contrast, we found a strongly and statistically significant negative correlation between SRS and mean QoL in the POST-LG group. The explanation could be that hearing loss in the better-performing patients in the POST-LG group occurred later in life, when they transitioned from normal hearing to CI more rapidly and without the use of a hearing aid (data not shown). Therefore, they did not experience long-term difficulties in hearing, but rather, had a preserved, recent memory of normal hearing that lessened appreciation of hearing with a CI. The absence of a correlation between SRS and QoL was noted for the PRE-LG group and was particularly evident on subgroup analysis.
A comparison between the 6 prelingually deafened patients with sound deprivation (and lower SRS) and the 11 PRE-LG patients without sound deprivation showed no statistically relevant difference in mean QoL scores (2.46 in group D vs 2.75 in group S;
A limitation of the present study was the small number of patients recruited into the PRE-LG D subgroup; in fact, these patients are often excluded from presurgical counseling because they are often poor candidates for CI. In fact, however, there is no evidence in the literature supporting the benefits of CI in prelingually deprived patients. Another limit was the absence of a QoL questionnaire pre-CI, meaning that we could not compare changes in perceived QoL.
Conclusions
Our findings showed better auditory performance in the patients in the POST-LG group and no differences between the groups in perceived QoL or benefit of CI. The sound-deprived patients had lower SRS than the sound-stimulated patients but equal scores on the perceived QoL questionnaire. A subjective evaluation of QoL revealed that sound-deprived, prelingually deafened patients may be eligible for CI.
Figures
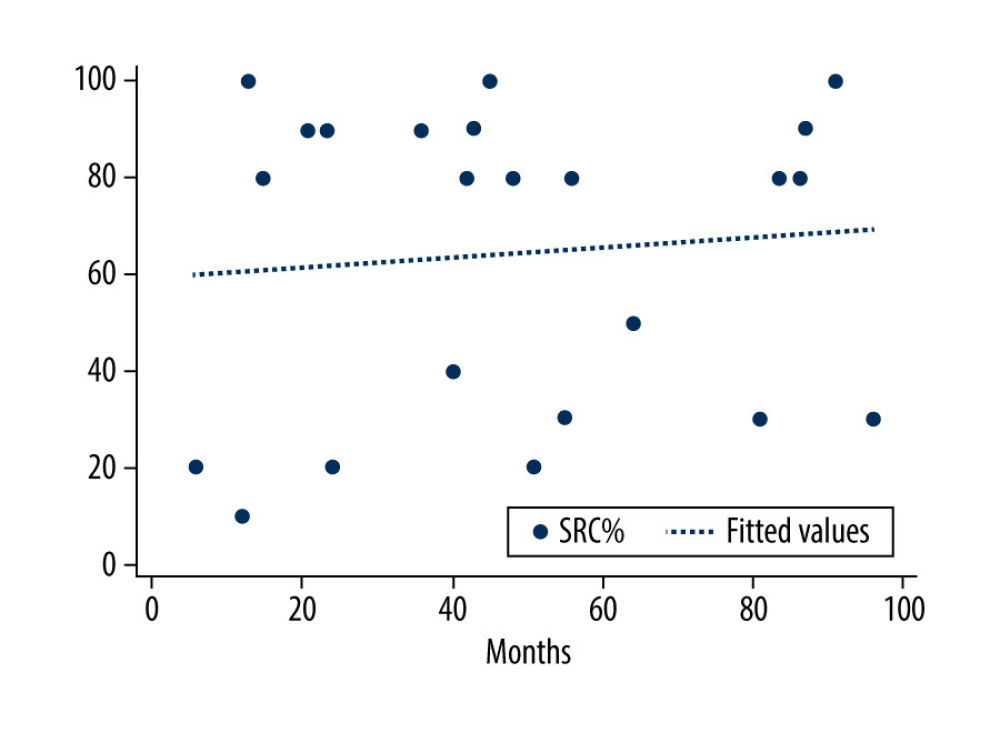 Figure 1. Correlation between months of cochlear implant use and speech recognition score in the prelingually deafened group (rs=0.07; P=0.736). SRS – speech recognition score, PRE-LG – prelingually deafened patients.
Figure 1. Correlation between months of cochlear implant use and speech recognition score in the prelingually deafened group (rs=0.07; P=0.736). SRS – speech recognition score, PRE-LG – prelingually deafened patients. 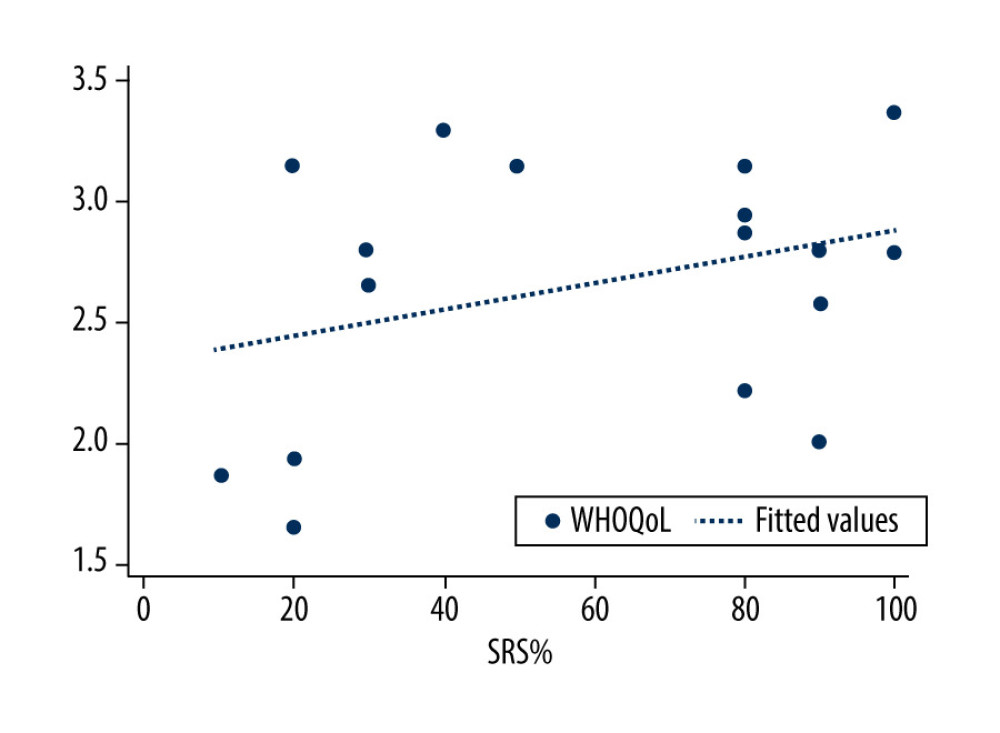 Figure 2. Correlation between months of cochlear implant use and speech recognition score in the postlingually deafened group (rs=0.11; P=0.618). SRS – speech recognition score, POST-LG – postlingually deafened patients.
Figure 2. Correlation between months of cochlear implant use and speech recognition score in the postlingually deafened group (rs=0.11; P=0.618). SRS – speech recognition score, POST-LG – postlingually deafened patients. 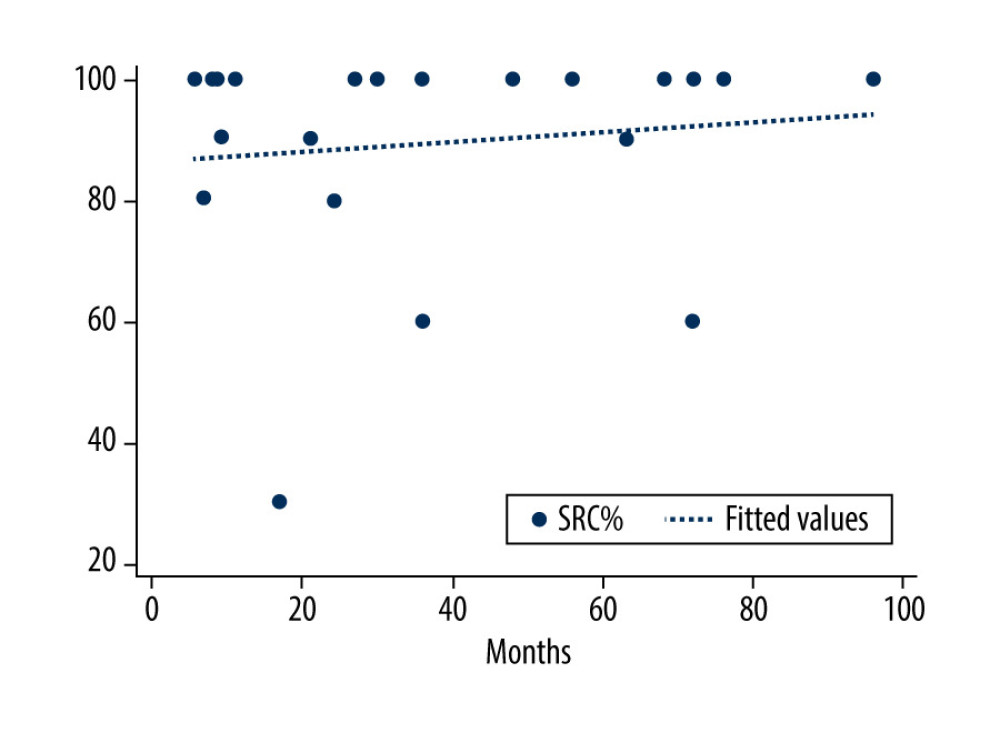 Figure 3. Correlation between speech recognition score and mean quality of life score for each patient in the prelingually deafened group (rs=0.32; P=0.216). SRS – speech recognition score, PRE-LG – prelingually deafened patients.
Figure 3. Correlation between speech recognition score and mean quality of life score for each patient in the prelingually deafened group (rs=0.32; P=0.216). SRS – speech recognition score, PRE-LG – prelingually deafened patients. 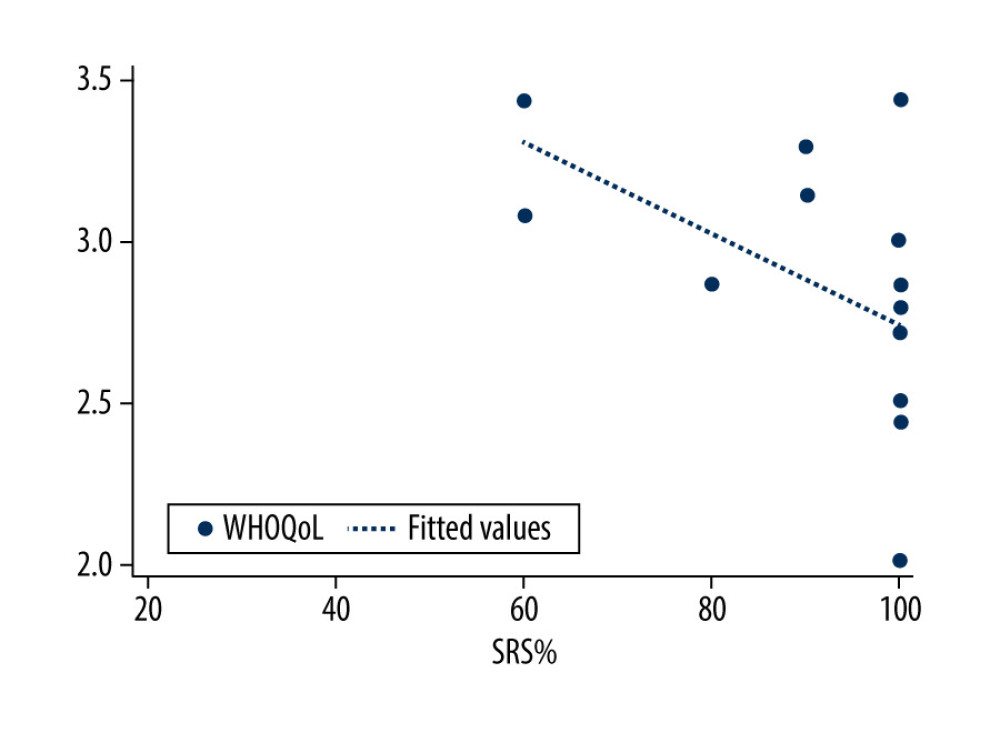 Figure 4. Correlation between speech recognition score and mean quality of life score for each patient in the postlingually deafened group (rs=−0.60; P=0.024). SRS – speech recognition score, POST-LG – postlingually deafened patient.
Figure 4. Correlation between speech recognition score and mean quality of life score for each patient in the postlingually deafened group (rs=−0.60; P=0.024). SRS – speech recognition score, POST-LG – postlingually deafened patient. References
1. Waltzman SB, Cohen NL, Shapiro WH, Use of a multichannel cochlear implant in the congenitally and prelingually deaf population: Laryngoscope, 1992; 102(4); 395-99
2. Skinner MW, Binzer SM, Fears BT, Study of the performance of four prelinguistically or perilinguistically deaf patients with a multi-electrode, intracochlear implant: Laryngoscope, 1992; 102(7); 797-806
3. Blamey P, Artieres F, Baskent D, Factors affecting auditory performance of postlinguistically deaf adults using cochlear implants: An update with 2251 patients: Audiol Neurotol, 2013; 18; 36-47
4. Pattisapu P, Lindquist NR, Appelbaum EN, A systematic review of cochlear implant outcomes in prelingually-deafened, late-implanted patients: Otol Neurotol, 2020; 41; 444-51
5. Teoh SW, Pisoni DB, Miyamoto RT, Cochlear implantation in adults with prelingual deafness. Part I. Clinical results: Laryngoscope, 2004; 114(9); 1536-40
6. Tong MC, Leung EK, Au A, Age and outcome of cochlear implantation for patients with bilateral congenital deafness in a Cantonese-speaking population: Ear Hear, 2007; 28(2 Suppl); 56S-58S
7. Yang WS, Moon IS, Kim HN, Delayed cochlear implantation in adults with prelingual severe-to-profound hearing loss: Otol Neurotol, 2011; 32(2); 223-28
8. van Dijkhuizen JN, Boermans PP, Briaire JJ, Intelligibility of the patient’s speech predicts the likelihood of cochlear implant success in prelingually deaf adults: Ear Hear, 2016; 37(5); e302-10
9. Kumar RS, Mawman D, Sankaran D, Cochlear implantation in early deafened, late-implanted adults: Do they benefit?: Cochlear Implants Int, 2016; 17(Suppl 1); 22-25
10. Canale A, Dalmasso G, Dagna F, Monaural or binaural sound deprivation in postlingual hearing loss: Cochlear implant in the worse ear: Laryngoscope, 2016; 126(8); 1905-10
11. Canale A, Santagata F, Massaia M, Cochlear implant in elderly deaf patients with adverse predictors of audiological outcome: Otorinolaringologia, 2019; 69(1); 21-25
12. Sorrentino F, Gheller F, Lunardi G, Cochlear implantation in adults with auditory deprivation: What do we know about it?: Am J Otolaryngol, 2020; 41(2); 102366
13. McRackan TR, Bauschard M, Hatch JL, Meta-analysis of quality-of-life improvement after cochlear implantation and associations with speech recognition abilities: Laryngoscope, 2018; 128(4); 982-90
14. Forli F, Lazzerini F, Fortunato S, Cochlear implant in the elderly: Results in terms of speech perception and quality of life: Audiol Neurootol, 2019; 24(2); 77-83
15. Boisvert I, Lyxell B, Mäki-Torkko E, Choice of ear for cochlear implantation in adults with monaural sound-deprivation and unilateral hearing aid: Otol Neurotol, 2012; 33; 572-79
16. De Girolamo G, Rucci P, Scocco P, Quality of life assessment: Validation of the Italian version of the WHOQOL-Brief: Epidemiol Psychiatr Sci, 2000; 9(1); 45-55
17. Boisvert I, McMahon CM, Dowell RC, Long-term asymmetric hearing affects cochlear implantation outcomes differently in adults with pre- and postlingual hearing loss: PLoS One, 2015; 10(6); e0129167
18. Krueger B, Joseph G, Rost U, Performance groups in adult cochlear implant users: Speech perception results from 1984 until today: Otol Neurotol, 2008; 29(4); 509-12
19. Forli F, Turchetti G, Giuntini G, Cochlear implant in prelingually deafened oralist adults: Speech perception outcomes, subjective benefits and quality of life improvement: Acta Otorhinolaryngol Ital, 2017; 37(5); 416-22
Figures
 Figure 1. Correlation between months of cochlear implant use and speech recognition score in the prelingually deafened group (rs=0.07; P=0.736). SRS – speech recognition score, PRE-LG – prelingually deafened patients.
Figure 1. Correlation between months of cochlear implant use and speech recognition score in the prelingually deafened group (rs=0.07; P=0.736). SRS – speech recognition score, PRE-LG – prelingually deafened patients. Figure 2. Correlation between months of cochlear implant use and speech recognition score in the postlingually deafened group (rs=0.11; P=0.618). SRS – speech recognition score, POST-LG – postlingually deafened patients.
Figure 2. Correlation between months of cochlear implant use and speech recognition score in the postlingually deafened group (rs=0.11; P=0.618). SRS – speech recognition score, POST-LG – postlingually deafened patients. Figure 3. Correlation between speech recognition score and mean quality of life score for each patient in the prelingually deafened group (rs=0.32; P=0.216). SRS – speech recognition score, PRE-LG – prelingually deafened patients.
Figure 3. Correlation between speech recognition score and mean quality of life score for each patient in the prelingually deafened group (rs=0.32; P=0.216). SRS – speech recognition score, PRE-LG – prelingually deafened patients. Figure 4. Correlation between speech recognition score and mean quality of life score for each patient in the postlingually deafened group (rs=−0.60; P=0.024). SRS – speech recognition score, POST-LG – postlingually deafened patient.
Figure 4. Correlation between speech recognition score and mean quality of life score for each patient in the postlingually deafened group (rs=−0.60; P=0.024). SRS – speech recognition score, POST-LG – postlingually deafened patient. Tables
 Table 1. Median pure tone audiometry and speech recognition scores in the prelingually deafened and postlingually deafened groups.
Table 1. Median pure tone audiometry and speech recognition scores in the prelingually deafened and postlingually deafened groups.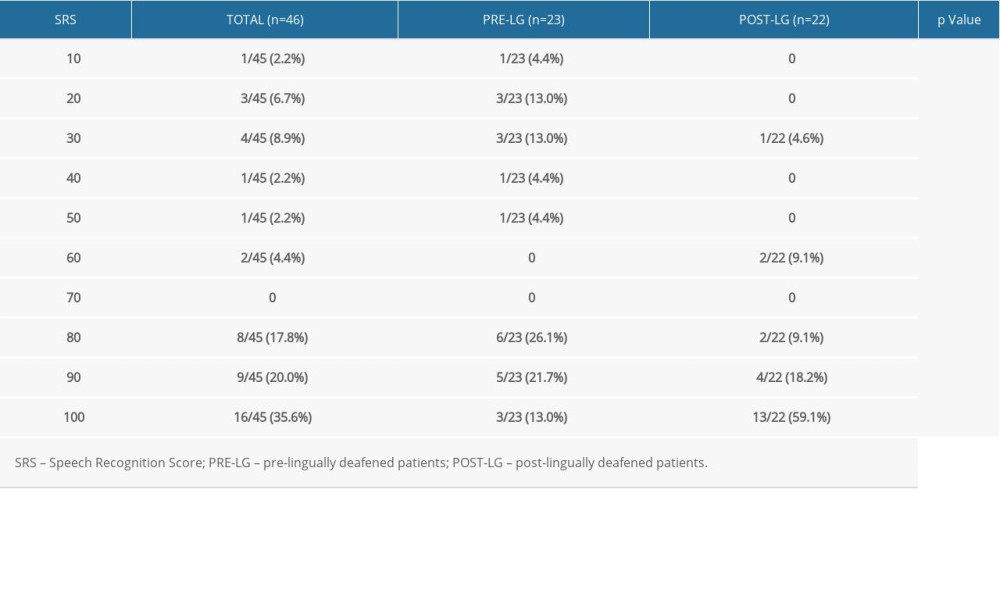 Table 2. Distribution of patients in relation to speech recognition scores.
Table 2. Distribution of patients in relation to speech recognition scores.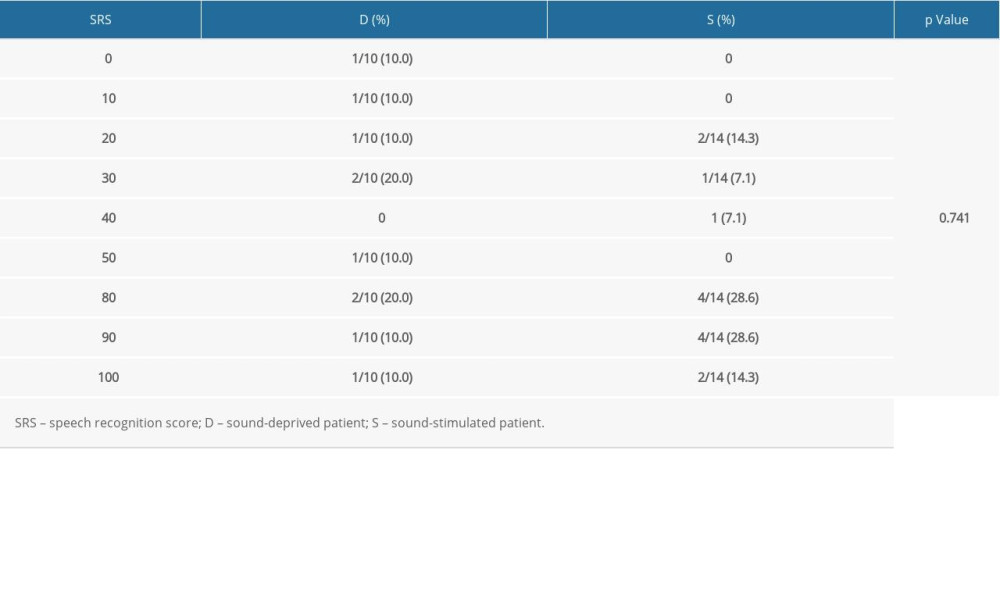 Table 3. Distribution of speech recognition scores for the D and S subgroups.
Table 3. Distribution of speech recognition scores for the D and S subgroups. Table 1. Median pure tone audiometry and speech recognition scores in the prelingually deafened and postlingually deafened groups.
Table 1. Median pure tone audiometry and speech recognition scores in the prelingually deafened and postlingually deafened groups. Table 2. Distribution of patients in relation to speech recognition scores.
Table 2. Distribution of patients in relation to speech recognition scores. Table 3. Distribution of speech recognition scores for the D and S subgroups.
Table 3. Distribution of speech recognition scores for the D and S subgroups. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








