05 April 2021: Clinical Research
Effects of Different Exposure Days to Gonadotropin-Releasing Hormone Agonist (GnRH-a) on Live Birth Rates in the Depot GnRH-a Protocol: A Retrospective Analysis of 7007 Cycles
Jianyuan Song1ABCDEFG, Wei Wu1BC, Liu Jiang2CD, Cuicui Duan3BC, Jian Xu1AG*DOI: 10.12659/MSM.929854
Med Sci Monit 2021; 27:e929854
Abstract
BACKGROUND: In controlled ovarian hyperstimulation protocols worldwide, depot gonadotropin-releasing hormone agonist (GnRH-a) pretreatment is generally used for pituitary desensitization. The delay between the GnRH-a administration and starting gonadotropin treatment varies greatly, from 25 to 60 days. However, the association between exposure days to GnRH-a before the onset of gonadotropin administration and the clinical outcomes remains unknown.
MATERIAL AND METHODS: This retrospective study included 7007 patients who underwent fresh embryo transfers between February 2016 and July 2019. The duration of pituitary downregulation was categorized into 3 groups: group 1, ≤30 days; group 2, 31-35 days; and group 3, ≥36 days. The rates of live birth were compared as the main outcome measure. Logistic regression analysis was also performed after controlling for a range of confounders.
RESULTS: The number of patients in groups 1, 2, and 3 was 2001, 2824, and 2182, respectively. Group 3 (≥36 days) had a noticeably higher live birth rate (48.1%) than the other 2 groups (42.6% and 43.9%, P=0.001). The rate of live birth was remarkably enhanced in group 3 (adjusted odds ratio: 1.264, 95% confidence interval: 1.098, 1.455, P=0.001) after controlling for confounders, while the difference was not found in group 2 (P=0.512) compared with group 1.
CONCLUSIONS: In the depot GnRH-a protocol, live birth rates are higher among patients needing a longer time to achieve the goal of pituitary downregulation.
Keywords: Live Birth, Ovulation Induction, Pregnancy Rate, Birth Rate, Chorionic Gonadotropin, Embryo Transfer, Gonadotropin-Releasing Hormone, Pituitary Gland, Pregnancy
Background
Among the various controlled ovarian hyperstimulation protocols worldwide, the depot gonadotropin-releasing hormone agonist (GnRH-a) protocol is recognized as safe and effective for pituitary desensitization [1]. In the depot GnRH-a protocol consisting of the injection of 1 dose of long-acting GnRH-a analogues on day 2 of the menses, patients are scheduled to report back to the reproductive center for confirmation of sufficient desensitization within 25–60 days. The benefits of using long-acting GnRH-a have been well documented and include the prevention of a premature luteinizing hormone surge and a larger number of oocytes [2]. Particularly in uncontrolled conditions such as an epidemic period, the use of long-acting GnRH-a is theoretically less likely to require scheduling embryo transfers on weekends or having too many patients on a particular day. The depot GnRH-a regimen is welcomed both by patients and physicians because it simplifies pharmacological treatment and allows flexibility in timing the start of gonadotropin (Gn).
Many investigators have considered appropriate dosage of the long-acting depot GnRH-a [3–5], the difference between the depot GnRH-a protocol (single injection) and the ultralong GnRH-a protocol (3 injections) [6], and the effects of other exogenous agents that might potentially impair reproduction [7,8]. However, the duration of exposure to GnRH-a before ovarian stimulation varies greatly even in patients receiving the same standard depot GnRH-a protocol. In a previous study we showed that a prolonged pituitary downregulation protocol was effective in improving the outcomes of in vitro fertilization (IVF) treatment, resulting in the exposure days to GnRH-a ranging from 25 to 60 days before the stimulation phase [9]. However, the effect of different durations of desensitization on clinical outcomes is not fully understood. This retrospective study compared the laboratory and pregnancy outcomes according to the delay between GnRH-a administration and the start of Gn.
Material and Methods
STUDY POPULATION:
This retrospective cohort study of IVF and intracytoplasmic sperm injection (ICSI) treatment using GnRH-a was conducted at Women’s Hospital, School of Medicine, Zhejiang University, China from February 2016 to July 2019. The inclusion criteria were the following: (1) patients undergoing a first stimulation cycle; (2) cycles with the depot GnRH-a protocol; and (3) patients undergoing fresh embryo transfers. The exclusion criteria were (1) egg donors; (2) preimplantation genetic testing or diagnosis; (3) patients without retrieved oocytes; and (4) patients with all embryos frozen. The study was carried out according to the Institutional Review Committee at Zhejiang University, China and in adherence to STROBE guidelines [10]. Written informed consent was waived due to the retrospective nature of the study.
STIMULATION PROTOCOLS:
The depot GnRH-a protocol was the same as previously described. Briefly, 3.75 mg of GnRH-a was administered on day 2 during menses [9]. Women would have an appointment at the IVF department 25–60 days later for confirmation of downregulation. Then ovarian stimulation would be started by daily injection of recombinant human follicle-stimulating hormone (75–300 IU). IVF or ICSI was used to achieve fertilization on day 3, after which 1 or 2 embryos were transferred.
STANDARD OF DOWNREGULATION AND DEFINITION OF GROUPS:
The confirmation of successful downregulation was evaluated by follicle diameter (<5 mm), serum estradiol concentration (<50 pg/mL), luteinizing hormone level (<5 IU/L), and endometrial thickness (<5 mm) [9]. After 25–60 days of pituitary downregulation, Gn was injected. The duration of pituitary downregulation was classified into 3 groups: group 1, ≤30 days; group 2, 31–35 days; and group 3, ≥36 days.
CLINICAL OUTCOMES ASSESSMENT:
Live birth rates were calculated and compared among the 3 groups as the primary outcome measure. The secondary outcomes included the rates of clinical pregnancy, implantation, and miscarriage.
STATISTICAL ANALYSIS:
In the present study, categorical variables were summarized as number (n) with percentage and quantitative data as mean±standard deviation (SD). To assess demographics, Pearson’s chi-squared and analysis of variance were carried out for analysis of categorical and continuous data, respectively. Frequency distribution was categorized according to percentiles of the duration of exposure to GnRH-a. Confounding variables were adjusted in the logistic regression model to explore whether exposure time to GnRH-a before the start of Gn was associated with the outcome of live birth. Significance was defined as
Results
BASIC CHARACTERISTICS:
As presented in Table 1, the average age of women in the 3 groups was 30.2±3.7 years (mean±SD), 30.0±3.7 years, and 30.1±3.1 years, respectively, P=0.076. Women with a longer downregulation period had higher levels of follicle-stimulating hormone, P=0.000. No significant differences were found for years of infertility, anti-Müllerian hormone levels, body mass index, antral follicle count (AFC), or the infertility types among the 3 groups.
OVARIAN STIMULATION CHARACTERISTICS:
Table 2 describes the procedures for ovarian stimulation. Women with a longer exposure time to GnRH-a had significantly higher Gn dosage, estradiol level, progesterone level, number of retrieved oocytes, variable blastocysts, and 2 visualized pronuclei. The 3 groups had comparable results for serum luteinizing hormone level, endometrial thickness, and type of treatment.
PREGNANCY OUTCOMES:
The rate of live birth was 48.1% (1049/2182) in group 3, which was remarkably higher than in groups 1 and 2 (42.6% and 43.9%, respectively, P=0.001) (Table 3), while groups 1 and 2 did not differ markedly in live birth rates. The 3 groups had miscarriage rates of 32.1%, 26.8%, and 20.6%, respectively, but no significant differences were detected in the clinical pregnancy rates and implantation rates between groups.
MULTIVARIABLE LOGISTIC REGRESSION ANALYSIS:
Logistic regression analysis was also conducted. Table 4 shows that in patients undergoing the depot GnRH-a protocol, the independent factors correlated with pregnancy rate were the duration of pituitary downregulation, maternal age, AFC, the number of embryos transferred, and endometrial thickness (all P<0.05). The live birth rate was markedly enhanced in group 3 (adjusted odds ratio: 1.264, 95% confidence interval: 1.098, 1.455, P=0.001), while the difference was not found in group 2 (P=0.512).
Discussion
The depot GnRH-a protocol offers the advantages of better compliance, convenience, and lower cost for patients, as well as less stress regarding injections and risk of infection [4,11]. In particular, the protocol allows flexibility in the timing of Gn administration to start ovarian stimulation, ranging from 25 to 60 days after administration of a single depot dose of GnRH-a. This flexibility enables cycle commencement to better accommodate clinics and patients. However, the evidence on the effect of exposure time to GnRH-a on the live birth is insufficient. The present retrospective study indicates that IVF/ICSI cycles using the depot GnRH-a protocol result in higher live birth rates among patients who need a longer period of time to achieve the goal of pituitary downregulation.
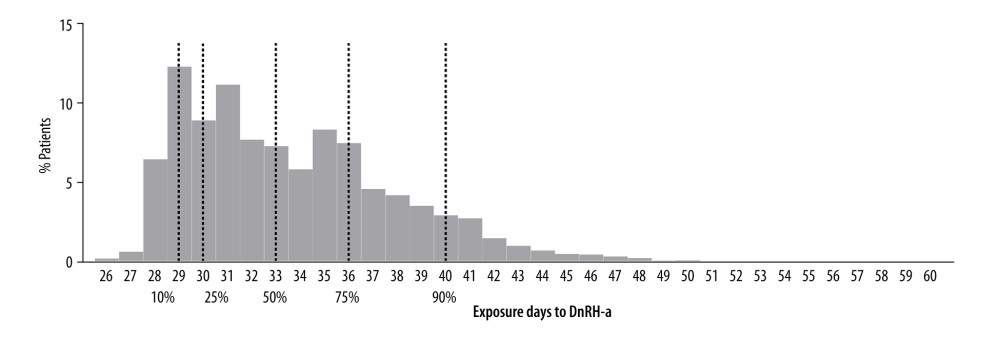
The depot GnRH-a pretreatment has been widely used in assisted reproduction for decades [12]. Two studies of 378 cycles and 273 cycles clarified that prolonged use of GnRH-a might increase the live birth rates compared with the regular GnRH-a long protocol, but exposure days to GnRH-a were not reported [13,14]. Compared with the conventional GnRH-a protocol, in which 0.1 mg of GnRH-a is administered daily, 3.75 mg of a long-acting GnRH-a resulted in longer exposure time to GnRH-a before the onset of Gn treatment [5,9]. The results of our study demonstrated that hormone suppression time with GnRH-a ranged from 26 to 60 days, and for the majority of patients (90%), the was between 28 and 42 days (Figure 1). Approximately 5% of the population had more than 42 days of exposure to GnRH-a. These results suggested that the duration of exposure to GnRH-a varied greatly among different populations.
The association between exposure days to GnRH-a and clinical outcomes remains unknown. A study of 86 patients found no difference in clinical outcomes between a long (15–24 days) and a very long (25–40 days) protocol of GnRH-a administration for ICSI [15]. More recently, an investigation of 506 cycles reported similar live birth rates between women receiving 14 days of downregulation and 7–28 additional days [16]. These studies showed that the rate of live births might not benefit from prolonging GnRH-a, which accorded with the findings of our study. In our results, group 1 (≤30 days) and group 2 (31–35 days) did not differ significantly in the rate of live birth. Interestingly, group 3 showed statistical significance for ≥36 days, which was not reported in previous studies.
To analyze the effect of different exposure days to GnRH-a on live birth rates, we categorized the duration of pituitary downregulation into 3 groups: group 1, ≤30 days; group 2, 31–35 days; and group 3, ≥36 days. Our data showed that the live birth rate was 48.1% in group 3, which was significantly higher than in the other 2 groups (Table 3). Baseline characteristics of patients were similar between groups (Table 1), which minimized selection bias in this study. It is noteworthy that exposure days to GnRH-a was not significantly influenced by parameters of ovarian aging such as maternal age and AFC. These results suggest that when predicting the ultrasound day for patients undergoing the depot GnRH-a protocol, age and AFC might not be key considerations.
Extensive research has explored the potential independent factors for successful assisted reproduction [17–21]. Similarly, the logistic regression analysis in present study demonstrated that the independent factors were age, AFC, the number of embryos transferred, and endometrial thickness. In line with this, we observed that the duration of pituitary downregulation was another contributing factor correlated with the live birth rate (adjusted odds ratio: 1.264, 95% confidence interval: 1.098, 1.455,
However, more than 60 days of GnRH-a exposure before IVF might have limited benefits on the rate of pregnancy [22,23]. GnRH-a was shown to cause extrapituitary adverse effects such as inhibition of ovarian steroidogenesis [24], disrupted differentiation of granulosa cells [25], and changes in GnRH receptor representation [26]. Such effects would offer an explanation for the difference in outcomes in our study compared with previous studies. Our research has some limitations. First, data on the live birth rate among patients with longer exposure to GnRH-a (>60 days) was not obtained, which might limit the wide clinical application of our findings. Second, this study was retrospective and observational in design. The conclusion that a longer duration of GnRH-a pretreatment leads to higher live birth rates cannot be drawn.
Conclusions
In IVF/ICSI cycles of the depot GnRH-a protocol, live birth rates were higher among patients who needed a longer time to achieve the goal of pituitary downregulation.
References
1. Marcus SF, Ledger WL, Efficacy and safety of long-acting GnRH agonists in in vitro fertilization and embryo transfer: Hum Fertil (Camb), 2001; 4(2); 85-93
2. Vlaisavljević V, Kovacic B, Gavrić-Lovrec V, Reljic M, Simplification of the clinical phase of IVF and ICSI treatment in programmed cycles: Int J Gynaecol Obstet, 2000; 69(2); 135-42
3. Li Y, Yang D, Zhang Q, Clinical outcome of one-third-dose depot triptorelin is the same as half-dose depot triptorelin in the long protocol of controlled ovarian stimulation: J Hum Reprod Sci, 2012; 5(1); 14-19
4. Safdarian L, Mohammadi FS, Alleyassin A, Clinical outcome with half-dose depot triptorelin is the same as reduced-dose daily buserelin in a long protocol of controlled ovarian stimulation for ICSI/embryo transfer: A randomized double-blind clinical trial (NCT00461916): Hum Reprod, 2007; 22(9); 2449-54
5. Dal Prato L, Borini A, Cattoli M, GnRH analogs: depot versus short formulations: Eur J Obstet Gynecol Reprod Biol, 2004; 115(Suppl 1); S40-43
6. Manno M, Tomei F, Cervi M, Comparison of protocols efficacy in poor responders: Differences in oocytes/embryos competence with different protocols, a retrospective study: Fertil Steril, 2009; 91(4 Suppl); 1431-33
7. De Franciscis P, Guadagno M, Miraglia N, Follicular PB levels in women attending in vitro fertilization: Role of endometriosis on the outcome: Ital J Gynaecol Obstet, 2018; 30; 21-27
8. Schiattarella A, Colacurci N, Morlando M, Plasma and urinary levels of lead and cadmium in patients with endometriosis: Ital J Gynaecol Obstet, 2018; 30; 47-52
9. Song J, Duan C, Cai W, Comparison of GnRH-a prolonged protocol and short GnRH-a long protocol in patients with thin endometrium for assisted reproduction: A retrospective cohort study: Drug Des Devel Ther, 2020; 14; 3673-82
10. von Elm E, Altman DG, Egger M, The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies: Lancet, 2007; 370(9596); 1453-57
11. Albuquerque LE, Tso LO, Saconato H, Depot versus daily administration of gonadotrophin-releasing hormone agonist protocols for pituitary down regulation in assisted reproduction cycles: Cochrane Database Syst Rev, 2013; 2013(1); CD002808
12. Depalo R, Jayakrishan K, Garruti G, GnRHa versus GnRH antagonist in in vitro fertilization and embryo transfer (IVF/ET): Reprod Biol Endocrinol, 2012; 10; 26
13. Ren J, Sha A, Han D, Does prolonged pituitary down-regulation with gonadotropin-releasing hormone agonist improve the live-birth rate in in vitro fertilization treatment?: Fertil Steril, 2014; 102(1); 75-81
14. Xu B, Geerts D, Hu S, The depot GnRH agonist protocol improves the live birth rate per fresh embryo transfer cycle, but not the cumulative live birth rate in normal responders: A randomized controlled trial and molecular mechanism study: Hum Reprod, 2020; 35(6); 1306-18
15. Loutradis D, Drakakis P, Kallianidis K, The effect of the duration of GnRH-agonist down regulation before ovarian stimulation on the biological and clinical outcome after intracytoplasmic sperm injection: Eur J Obstet Gynecol Reprod Biol, 1998; 80(2); 251-55
16. Dessolle L, Ferrier D, Colombel A, Prolonging GnRH-agonist to achieve ovarian suppression does not compromise the results of a long protocol: Eur J Obstet Gynecol Reprod Biol, 2011; 159(1); 111-14
17. Metello JL, Tomás C, Ferreira P, Can we predict the IVF/ICSI live birth rate?: JBRA Assist Reprod, 2019; 23(4); 402-7
18. McLernon DJ, Steyerberg EW, Te Velde ER, Predicting the chances of a live birth after one or more complete cycles of in vitro fertilisation: Population-based study of linked cycle data from 113 873 women: BMJ, 2016; 355; i5735
19. van Loendersloot LL, van Wely M, Limpens J, Predictive factors in in vitro fertilization (IVF): A systematic review and meta-analysis: Hum Reprod Update, 2010; 16(6); 577-89
20. Kasius A, Smit JG, Torrance HL, Endometrial thickness and pregnancy rates after IVF: A systematic review and meta-analysis: Hum Reprod Update, 2014; 20(4); 530-41
21. González-Foruria I, Peñarrubia J, Borràs A, Age, independent from ovarian reserve status, is the main prognostic factor in natural cycle in vitro fertilization: Fertil Steril, 2016; 106(2); 342-47.e2
22. Kaponis A, Chatzopoulos G, Paschopoulos M, Ultralong administration of gonadotropin-releasing hormone agonists before in vitro fertilization improves fertilization rate but not clinical pregnancy rate in women with mild endometriosis: A prospective, randomized, controlled trial: Fertil Steril, 2020; 113(4); 828-35
23. Georgiou EX, Melo P, Baker PE, Long-term GnRH agonist therapy before in vitro fertilisation (IVF) for improving fertility outcomes in women with endometriosis: Cochrane Database Syst Rev, 2019; 2019(11); CD013240
24. Dor J, Bider D, Shulman A, Effects of gonadotrophin-releasing hormone agonists on human ovarian steroid secretion in vivo and in vitro-results of a prospective, randomized in-vitro fertilization study: Hum Reprod, 2000; 15; 1225-30
25. Gerrero H, Stein P, Asch R, Effect of a gonadotropin-releasing hormone agonist on luteinizing hormone receptors and steroidogenesis in ovarian cells: Fertil Steril, 1993; 59; 803-8
26. Peng C, Fan NC, Ligier M, Expression and regulation of gonadotropin-releasing hormone (GnRH) and GNRH receptor messenger ribonucleic acids in human granulosa-luteal cells: Endocrinology, 1994; 35; 1740-46
Tables
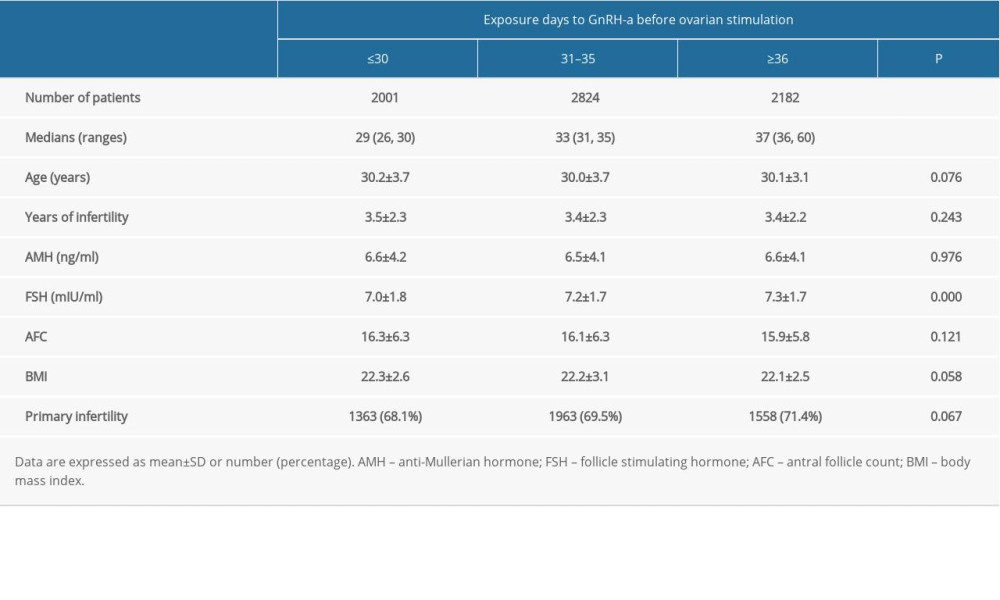 Table 1. Demographic characteristics of the patients.
Table 1. Demographic characteristics of the patients.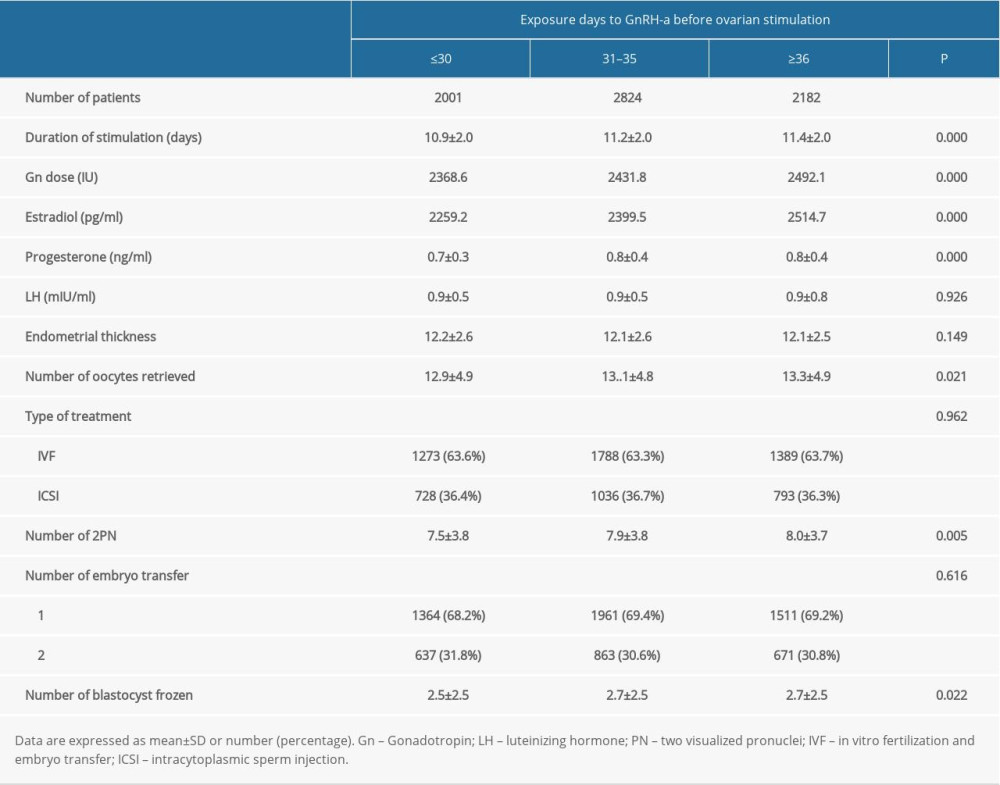 Table 2. Ovarian stimulation data.
Table 2. Ovarian stimulation data.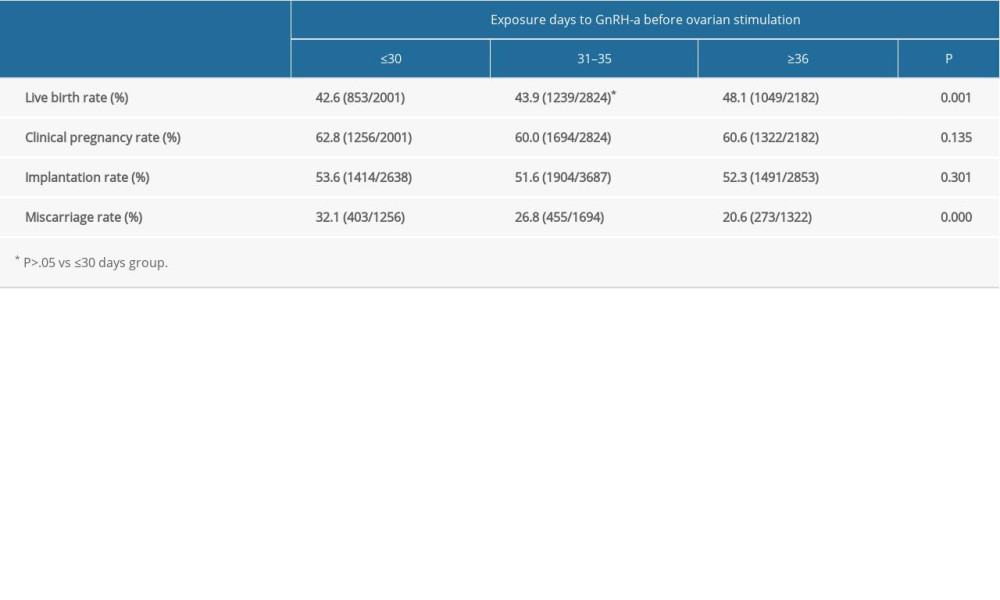 Table 3. Pregnancy outcomes.
Table 3. Pregnancy outcomes.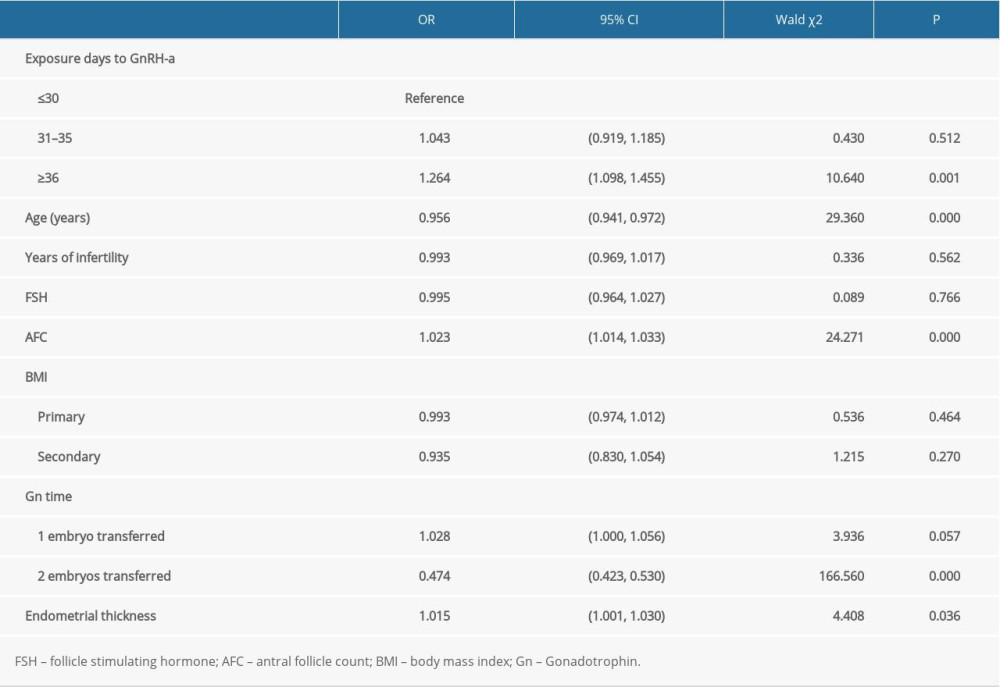 Table 4. Logistic regression analysis of the live birth.
Table 4. Logistic regression analysis of the live birth. Table 1. Demographic characteristics of the patients.
Table 1. Demographic characteristics of the patients. Table 2. Ovarian stimulation data.
Table 2. Ovarian stimulation data. Table 3. Pregnancy outcomes.
Table 3. Pregnancy outcomes. Table 4. Logistic regression analysis of the live birth.
Table 4. Logistic regression analysis of the live birth. In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952