02 June 2021: Clinical Research
Nomogram Model for Prediction of Portal Vein Thrombosis in Patients with Liver Cirrhosis After Splenectomy: A Retrospective Analysis of 2 Independent Cohorts
Hai-Liang Yuan1ABCEG, Min Wang2BCDG, Wei-Wei Chu1BDF, Fang-Xian Li1DE, Jing-Jing Lu1DG, Yan Li1ABDE*DOI: 10.12659/MSM.929844
Med Sci Monit 2021; 27:e929844
Abstract
BACKGROUND: The aim of this study was to establish and validate an easy-to-use nomogram to predict portal vein thrombosis (PVT) in patients with cirrhosis after splenectomy and to test its predictive ability.
MATERIAL AND METHODS: This retrospective study included 315 patients with cirrhosis who underwent splenectomy at 2 high-volume medical centers. The least absolute shrinkage and selection operator (LASSO) regression method was used to select the predictors in the training cohort, and multivariable logistic regression analysis was performed to establish the predictive nomogram model. We determined the prediction value of the nomogram by the area under the receiver operating characteristic curve (AUROC), the calibration curve, and decision curve analysis. Finally, the applicability of the nomogram was internally and independently validated.
RESULTS: The predictors of PVT included portal vein diameter, splenic vein diameter, body mass index, and platelet count. Based on the clinical and radiomic models, the nomogram had good predictive efficiency for predicting PVT in patients with cirrhosis after splenectomy, with an AUROC of 0.887 (0.856 in internal validation and 0.796 in independent validation). The decision curve analysis revealed that the nomogram had good clinical application value.
CONCLUSIONS: We successfully developed an easy-to-use nomogram to predict the probability of PVT in patients with cirrhosis after splenectomy. The nomogram can help clinicians make timely, individualized clinical decisions for PVT in patients with cirrhosis after splenectomy.
Keywords: Liver Cirrhosis, nomograms, Splenectomy, Venous Thrombosis, Body Mass Index, Clinical Decision Rules, Organ Size, Platelet Count, Portal Vein, Risk Assessment
Background
Liver cirrhosis is a pathological state of chronic liver damage caused by various factors, such as non-alcoholic steatohepatitis, viral hepatitis, autoimmune hepatitis, and cholestatic liver disease [1]. Portal vein thrombosis (PVT) is the formation of thrombosis in the portal vein or its branches, which can lead to portal hypertension and a series of pathophysiological changes [2]. The incidence of PVT in the general population is low at about 1% [3], while that in patients with liver cirrhosis is considerably higher [4], ranging from 1% to 25% [5–8]. PVT is mainly caused by the decrease in blood flow velocity of the portal vein [9].
Splenectomy is a main surgical treatment of hypersplenism and gastrointestinal bleeding caused by cirrhosis [10]. However, the incidence of PVT after splenectomy ranges from 9.8% to 47.9% [11]. Most patients with PVT lack specific clinical manifestations and sometimes show only mild abdominal pain, which they often ignore [12]. At present, the diagnosis of PVT mainly relies on imaging examination, especially Doppler ultrasound, which is often the first choice of examination because of its fast, noninvasive, and repeatable features [13]. However, imaging can detect only existing PVT and cannot be used to screen for PVT in high-risk patients. PVT can cause liver function damage, mesenteric vein embolism, intestinal perforation, and even death. Therefore, early identification of PVT in the high-risk population and timely treatment are particularly important.
At present, the research on PVT after splenectomy is focused on high-risk factors [14,15]. The proposed risk factors include the diameter of the splenic vein (DSV), diameter of the portal vein (DPV), splenic vein flow velocity, splenic volume, plasma D-dimer level, postoperative platelet count, and mean platelet volume [16,17]. However, there is no consensus on PVT risk factors, and no standardized clinical guidelines exist. Similarly, there is no related research on a PVT prediction model or nomogram in patients with cirrhosis after splenectomy. We believe a concise and efficient visualization model is urgently needed. Therefore, the main goal of this study was to establish and verify a nomogram prediction model of PVT risk in patients with cirrhosis after splenectomy, so that clinicians can screen and identify high-risk patients as early as possible.
Material and Methods
PATIENTS:
The primary cohort of our study included 219 patients who had cirrhosis after undergoing splenectomy at the First Affiliated Hospital of Nanchang University from January 2014 to August 2019. The independent validation cohort consisted of 96 patients with cirrhosis after undergoing splenectomy at the Beilun Branch Hospital of the First Affiliated Hospital of Zhejiang University from January 2015 to August 2019. The patient screening was performed using the same criteria as the primary cohort.
The indications of splenectomy in patients with cirrhosis met 1 of the following conditions: white blood cell <3.0×109/L and/or platelet count <30×10109/L; grade II splenomegaly with upper gastrointestinal bleeding or obvious hypersplenism; or grade III or above splenomegaly.
The inclusion criteria were as follows: (1) liver cirrhosis confirmed by liver imaging and/or histology; (2) splenectomy combined with devascularization for portal hypertension; and (3) PVT as confirmed by ultrasound, computed tomography, magnetic resonance imaging, or other imaging examinations.
The exclusion criteria were as follows: (1) incomplete data; (2) PVT occurred before splenectomy; (3) patients who had been taking antiplatelet or anticoagulant drugs for a long time; and (4) the presence of diseases including hematological diseases, malignant tumors, autoimmune system diseases. A flow diagram detailing the screening process is shown in Figure 1.
STATISTICAL ANALYSIS:
SPSS version 25.0 (IBM, Armonk, NY, USA) and R software (V.3.6.3, R Foundation for Statistical Computing, Vienna, Austria) were used for statistical analysis. The continuous data with a normal distribution were expressed by mean and standard deviation, and the comparison between the 2 groups was conducted by
DEVELOPMENT AND VALIDATION OF THE NOMOGRAM MODEL:
The primary cohort of patients with cirrhosis after splenectomy were randomly divided into 2 groups in a proportion of 2: 1, with the first group as the training group and the second as the internal validation group. Based on the data set, the LASSO [18,19] method was used to select the optimal predictive features of risk factors of PVT in patients with liver cirrhosis after splenectomy. Then, multivariate logistic regression analysis was performed on the variables chosen in the LASSO method. According to the multivariate logistic regression results, the nomogram was constructed to visually score the individual risk probabilities of PVT in patients with cirrhosis who underwent splenectomy [20,21]. The receiver operating characteristic (ROC) curve was used to test the discriminant efficiency of the model. To determine the clinical usefulness of the model, decision curve analysis was used to quantify the net benefit under different threshold probabilities in the cohort [22].
Results
PATIENT CHARACTERISTICS:
In total, 315 patients with cirrhosis after splenectomy were enrolled in our final study cohort. The rate of PVT was 32.4% in the primary group and 16.7% in the independent validation group. All data, including the demographic, clinical, and laboratory characteristics of patients with cirrhosis enrolled in the 2 clinical centers, are provided in Table 1.
PREDICTIVE VARIABLES AND NOMOGRAM CONSTRUCTION:
In all, 20 features were incorporated into the LASSO regression model, and the 4 best predictors for PVT in liver cirrhosis were selected: DPV, DSV, body mass index (BMI), and platelet count (Figure 2).
Patients with cirrhosis after splenectomy in the primary group were randomly divided into a training data set (145 cases, about 67%) and internal validation data set (74 cases, about 33%). The 4 predictive variables (DPV, DSV, BMI, and platelet count) were combined to establish a prediction model of PVT after hepatectomy by multivariable logistic regression with backward step-wise selection based on the training data set. The selected clinical characteristics in the training data set are shown in Table 2. The nomogram for prediction of PVT in patients with cirrhosis after splenectomy is shown in Figure 3.
APPARENT ACCURACY OF THE NOMOGRAM:
The discriminative ability and predictive performance of the nomogram were represented by the ROC curve (Figure 4). The nomogram held good predictive efficiency in predicting the PVT in patients with cirrhosis after splenectomy with an area under the receiver operating characteristic curve of 0.887 (0.856 in internal validation and 0.796 in independent validation).
The calibration curves of the nomogram showed good agreement between prediction and observation (Figure 5). We obtained a good calibration curve in the nomogram, and the Hosmer-Lemeshow test was not significant in either data set (P>0.05), which indicated a high reliability of the nomogram’s prediction ability.
CLINICAL USE OF THE NOMOGRAM:
The decision curve analysis for the nomogram is presented in Figure 6. Results on the net benefit of the prediction model showed a superior risk threshold probability compared with the baseline, ranging from 5.9% to 73.6%. For instance, if the personal threshold probability of a patient with liver cirrhosis after splenectomy was 10%, the net benefit was 0.148, which was superior to the treatment “all” (all patients receive preventive anticoagulation intervention) with 0.124 and treatment “none” (no patients receive preventive anticoagulation intervention). If the risk threshold probabilities were 5% (<5.9%) and 75% (>73.6%), the net benefits were 0.172 and −0.013, respectively.
Discussion
Splenectomy is one of the surgical methods used to address complications of liver cirrhosis, such as hypersplenism and esophageal and gastric variceal bleeding. Splenectomy combined with devascularization is currently the main surgical procedure for the clinical treatment of portal hypertension and for the prevention of bleeding from esophageal and gastric varices [10]. However, the risk of complications after splenectomy should also be considered. PVT, a complication with a high incidence after splenectomy, is difficult to predict and can affect patient prognosis [23]. In this study, the overall incidence of PVT was 27.6%, which was close to that found in the reference literature [11,24]. Our study demonstrated that DSV, DPV, BMI, and platelet count may be related to the formation of PVT after splenectomy, which is consistent with previous research results [23,25,26]. However, sex, age, Child-Pugh classification, and operation type were not statistically associated with PVT after splenectomy in the present study.
The platelet count reflects the coagulation mechanism. Our results showed that postoperative platelet changes were related to the occurrence of PVT, through LASSO regression analysis (odds ratio was 1.011). Postoperative platelet changes may be one of the conditions necessary for PVT formation. The hematopoietic function of patients with liver cirrhosis usually does not decrease significantly. However, the destruction of platelets is increased and the destruction of the spleen after splenectomy is decreased because of the influence of hypersplenism. In the present study, the number of platelets in most patients increased sharply in the short term, which increased platelet aggregation. The blood was in a hypercoagulable state, which increased the risk of thrombosis, accordingly. A surge in the number of platelets is related to the formation of PVT [6]. Therefore, we need to be vigilant with patients with a sharp increase in platelet count after surgery, assess the risk of PVT according to the predictive model, and take timely preventive measures. However, we also found that some patients had a sharp increase in platelet count after surgery, without the presence of PVT. Some studies have suggested that relevant indexes of platelet function, such as C protein and P selectin, when expressed in patients with PVT after splenectomy, are related to the occurrence of PVT [27]. C protein and P selectin have been used as indicators of the degree of platelet activation [28]. Although, in the present study, the expression was already high in patients with cirrhosis, the platelet aggregation ability was also affected to a certain extent if the number of platelets did not increase significantly after surgery. Therefore, a change in postoperative platelet value may be one of the conditions for the formation of PVT, and clinical studies are needed for further verification.
DPV and DSV reflect the changes in hemodynamics. The present study found that both were positively correlated with PVT formation, and their odds ratio values were 1.260 and 1.417, respectively. This suggested that the larger their values, the greater the risk of PVT formation after splenectomy. When DPV and DSV increase, the following are observed: (1) the reduction of blood flow and flow velocity in the veins after splenectomy is more obvious; (2) it is easier for the blood to form a vortex; (3) the contact time of platelets and fibrinogen in the blood with the inner wall of the blood vessel increases; and (4) the clotting factor and concentration increases to a certain extent, eventually leading to thrombosis [29]. Studies have shown that in cirrhotic patients with portal hypertension, the wider the portal and splenic veins are, the more likely it is to cause the venous vessel wall to be damaged by the extended high pressure in the lumen [30]. At the same time, the blood flow rate is slow, promoting coagulation, and when substance removal is slow, it promotes the formation of PVT [31]. This is consistent with the results of the present study. Therefore, it is necessary to be alert to patients having significantly increased portal vein and splenic vein diameters during the perioperative period to assess their postoperative PVT risk and take preventive measures in a timely manner.
A large cohort study in the Middle East showed that obesity is associated with venous thrombosis [32]. In addition, obesity is associated with cancer, diabetes, cardiovascular disease, hypercholesterolemia, and other chronic diseases [33,34]. In the general population, it is also associated with venous thromboembolism [35]. A large public health cohort study of 57 054 participants in Denmark found that all obesity measurements were predictors of venous thromboembolism [36]. However, a follow-up study of 6708 subjects in Norway showed that waist circumference is the best anthropometric indicator for obesity and can predict the risk of venous thromboembolism [37]. Several studies have shown that obesity is an independent risk factor associated with PVT in patients with cirrhosis after liver transplantation [38,39]. Similarly, a study in France showed that central obesity may be the primary risk factor of noncirrhotic PVT [40]. The potential mechanism of PVT in obesity is the presence of inflamed dysfunctional adipose tissue, which can promote hemostasis, inflammation, and the mechanical participation of central obesity. A recent study showed that visceral fat is a hazard factor for PVT in patients with cirrhosis after liver transplantation [41]. The results of our study showed that the BMI was a hazard factor for PVT in patients with cirrhosis after resection. This may be related to the abnormal coagulation factors in obese patients, which resulted in blood viscosity. Similarly, patients with obesity are likely to be less active, which can easily cause blood stasis and increase the risk of thrombosis. Therefore, health education aimed at guiding patients in proper activities before splenectomy to prevent the occurrence of thrombosis should be enforced.
Our research has some limitations. First, this study used a retrospective analysis method. Therefore, the reliability and effectiveness of the data are limited, and we cannot completely eliminate the possibility of selection bias. Further, other confounding factors related to PVT in patients with liver cirrhosis undergoing splenectomy, such as diet, alcohol consumption, and exercise, were not included as variables in this study. Finally, all patients in this study were Chinese, and the sample size of this study was very small, which can be improved upon by larger studies including different populations.
Conclusions
In this study, we developed and validated an easy-to-use nomogram for predicting PVT in patients with cirrhosis after undergoing splenectomy. With the easy-to-use nomogram, clinicians could perform pretreatment of PVT management and facilitate timely, individualized clinical decision-making for different PVT risks in patients with cirrhosis after splenectomy.
Figures
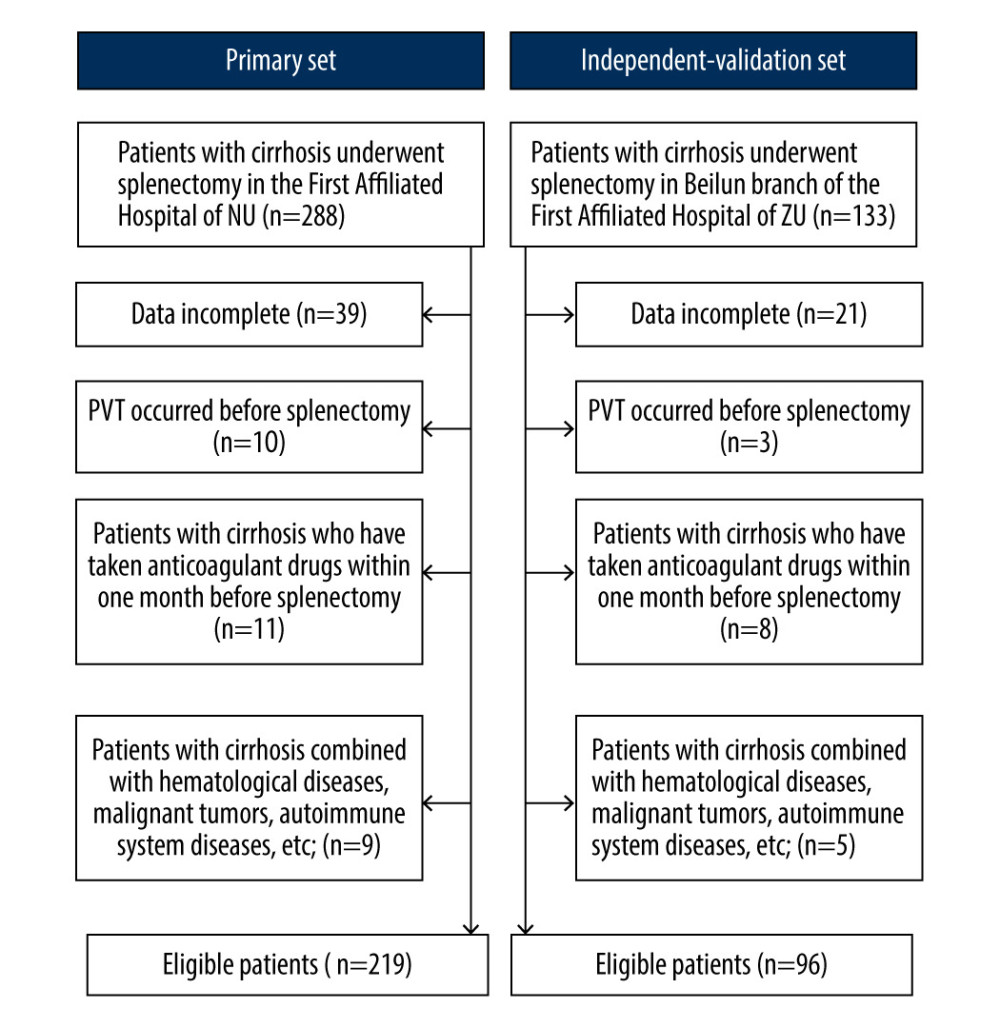 Figure 1. Flow chart of our study. NU – Nanchang University; ZU – Zhejiang University.
Figure 1. Flow chart of our study. NU – Nanchang University; ZU – Zhejiang University. 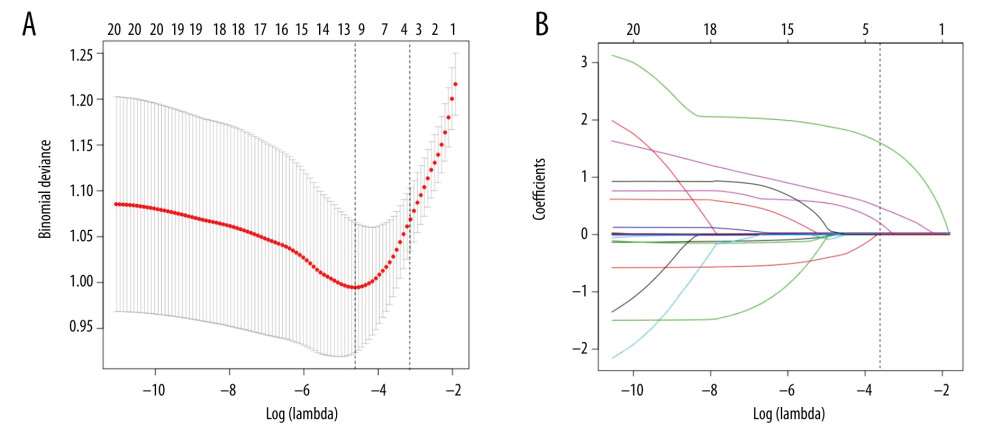 Figure 2. LASSO feature selection model. (A) LASSO coefficients of 20 candidate variables. (B) Identification of the optimal penalization coefficient (λ) in the LASSO model was achieved by 10-fold cross-validation and the minimum criterion.
Figure 2. LASSO feature selection model. (A) LASSO coefficients of 20 candidate variables. (B) Identification of the optimal penalization coefficient (λ) in the LASSO model was achieved by 10-fold cross-validation and the minimum criterion. 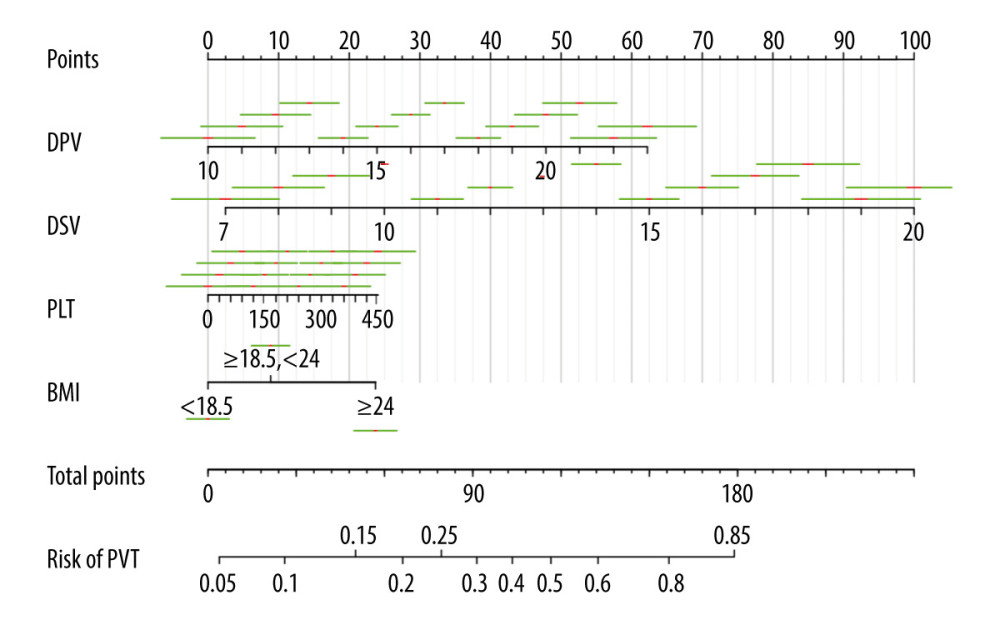 Figure 3. The nomogram for prediction of portal vein thrombosis in patients with cirrhosis after splenectomy.
Figure 3. The nomogram for prediction of portal vein thrombosis in patients with cirrhosis after splenectomy. 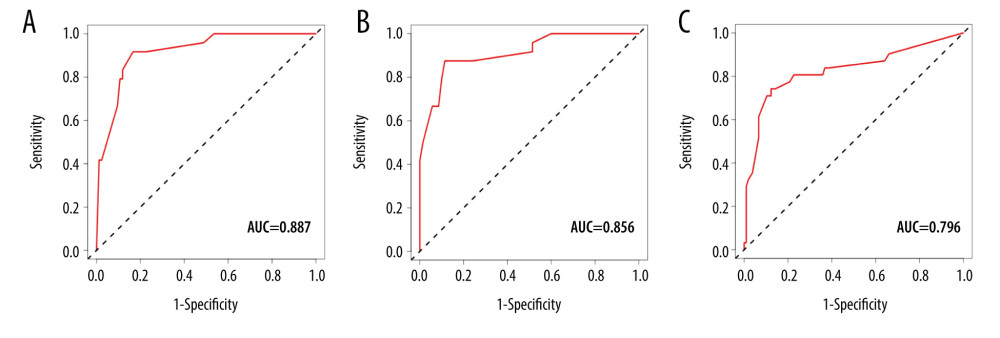 Figure 4. The receiver operating characteristic (ROC) curves for (A) training, (B) internal validation, and (C) independent validation set cohorts.
Figure 4. The receiver operating characteristic (ROC) curves for (A) training, (B) internal validation, and (C) independent validation set cohorts. 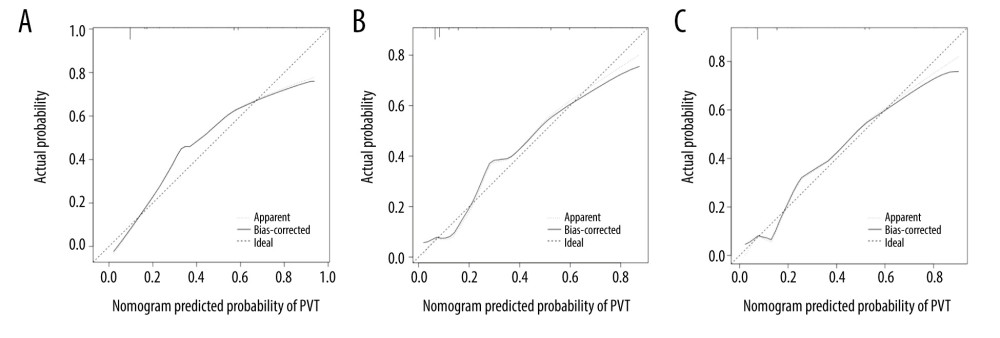 Figure 5. Calibration curve plot in each set. (A) the training set; (B) the internal validation set; (C) the independent validation set.
Figure 5. Calibration curve plot in each set. (A) the training set; (B) the internal validation set; (C) the independent validation set. 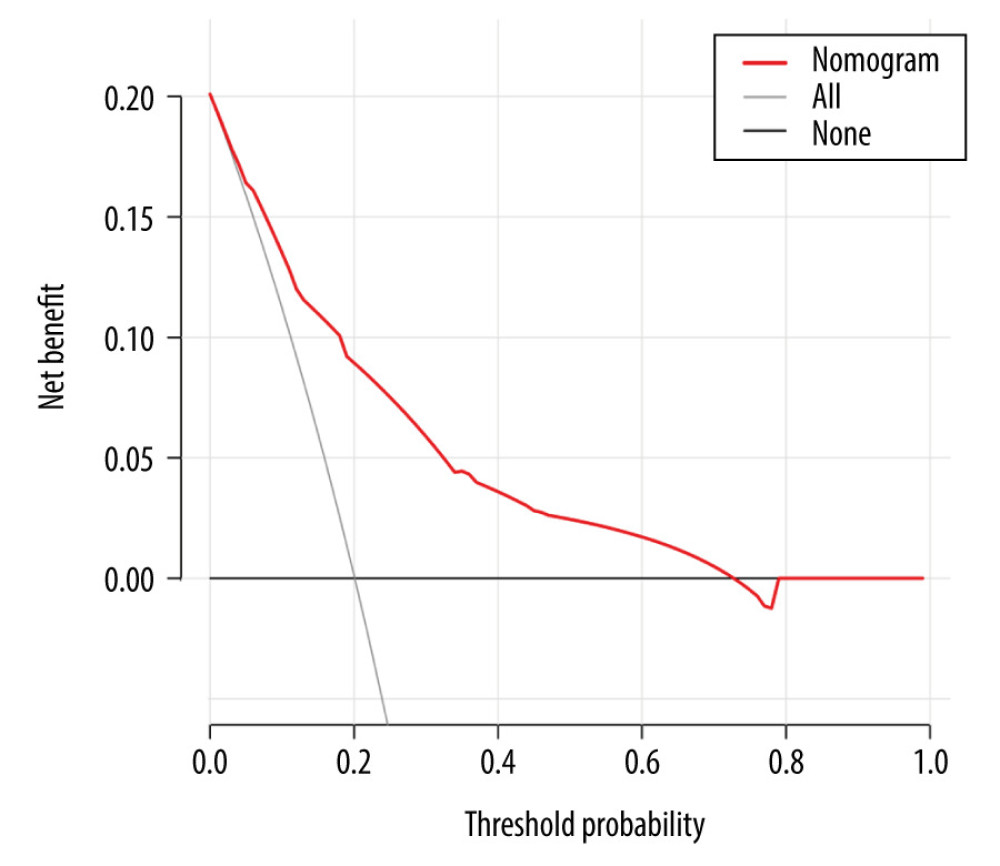 Figure 6. Decision curve analysis for the nomogram.
Figure 6. Decision curve analysis for the nomogram. References
1. Parola M, Pinzani M, Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues: Mol Aspects Med, 2019; 65; 37-55
2. Qi X, Han G, Fan D, Management of portal vein thrombosis in liver cirrhosis: Nat Rev Gastroenterol Hepatol, 2014; 11; 435-46
3. Eliasson A, Bergqvist D, Bjorck M, Incidence and risk of venous thromboembolism in patients with verified arterial thrombosis: A population study based on 23,796 consecutive autopsies: J Thromb Haemost, 2006; 4; 1897-902
4. Intagliata NM, Caldwell SH, Tripodi A, Diagnosis, development, and treatment of portal vein thrombosis in patients with and without cirrhosis: Gastroenterology, 2019; 156; 1582-1599.e1
5. Berry K, Taylor J, Liou IW, Portal vein thrombosis is not associated with increased mortality among patients with cirrhosis: Clin Gastroenterol Hepatol, 2015; 13; 585-93
6. Francoz C, Belghiti J, Vilgrain V, Usefulness of screening and anticoagulation: Gut, 2005; 54; 691-97
7. Violi F, Corazza GR, Caldwell SH, Portal vein thrombosis relevance on liver cirrhosis: Italian Venous Thrombotic Events Registry: Intern Emerg Med, 2016; 11; 1155
8. Nery F, Chevret S, Condat B, Causes and consequences of portal vein thrombosis in 1,243 patients with cirrhosis: results of a longitudinal study: Hepatology, 2015; 61; 660-67
9. Tsochatzis EA, Senzolo M, Germani G, Portal vein thrombosis in cirrhosis: Aliment Pharmacol Ther, 2010; 31; 366-74
10. Ruiz-Tovar J, Priego P, Portal vein thrombosis after splenic and pancreatic surgery: Adv Exp Med Biol, 2017; 906; 241-51
11. Kinjo N, Kawanaka H, Akahoshi T, Risk factors for portal venous thrombosis after splenectomy in patients with cirrhosis and portal hypertension: Br J Surg, 2010; 97; 910-16
12. He S, He F, Predictive model of portal venous system thrombosis in cirrhotic portal hypertensive patients after splenectomy: Int J Clin Exp Med, 2015; 8; 4236-42
13. Handa P, Crowther M, Douketis JD, Portal vein thrombosis: A clinician-oriented and practical review: Clin Appl Thromb Hemost, 2014; 20; 498-506
14. Kuroki T, Kitasato A, Tokunaga T, Predictors of portal and splenic vein thrombosis after laparoscopic splenectomy: A retrospective analysis of a single-center experience: Surg Today, 2018; 48; 804-9
15. Rottenstreich A, Kleinstern G, Spectre G, Risk factors, prevention, management and outcomes: World J Surg, 2018; 42; 675-81
16. Cagin YF, Atayan Y, Erdogan MA, Incidence and clinical presentation of portal vein thrombosis in cirrhotic patients: Hepatobiliary Pancreat Dis Int, 2016; 15; 499-503
17. von Kockritz L, De Gottardi A, Trebicka J, Portal vein thrombosis in patients with cirrhosis: Gastroenterol Rep (Oxf), 2017; 5; 148-56
18. Sauerbrei W, Royston P, Binder H, Selection of important variables and determination of functional form for continuous predictors in multivariable model building: Stat Med, 2007; 26; 5512-28
19. Friedman J, Hastie T, Tibshirani R, Regularization paths for generalized linear models via coordinate descent: J Stat Softw, 2010; 33; 1-22
20. Iasonos A, Schrag D, Raj GV, How to build and interpret a nomogram for cancer prognosis: J Clin Oncol, 2008; 26; 1364-70
21. Balachandran VP, Gonen M, Smith JJ, More than meets the eye: Lancet Oncol, 2015; 16; e173-80
22. Vickers AJ, Cronin AM, Elkin EB, Extensions to decision curve analysis, a novel method for evaluating diagnostic tests, prediction models and molecular markers: BMC Med Inform Decis Mak, 2008; 8; 53
23. Makdissi FF, Herman P, Machado MA, Portal vein thrombosis after esophagogastric devascularization and splenectomy in schistosomal portal hypertension patients: What’s the real importance?: Arq Gastroenterol, 2009; 46; 50-56
24. Dong F, Luo SH, Zheng LJ, Incidence of portal vein thrombosis after splenectomy and its influence on transjugular intrahepatic portosystemic shunt stent patency: World J Clin Cases, 2019; 7; 2450-62
25. Fujita F, Lyass S, Otsuka K, Portal vein thrombosis following splenectomy: identification of risk factors: Am Surg, 2003; 69; 951-56
26. Huang D, Tao M, Cao L, Risk Factors and anticoagulation effects of portal vein system thrombosis after laparoscopic splenectomy in patients with or without cirrhosis: Surg Laparosc Endosc Percutan Tech, 2019; 29; 498-502
27. Suda T, Takatori H, Hayashi T, Investigation of thrombosis volume, anticoagulants, and recurrence factors in portal vein thrombosis with cirrhosis: Life (Basel), 2020; 10; 177
28. Hung HC, Lee JC, Cheng CH, Protein S for portal vein thrombosis in cirrhotic patients waiting for liver transplantation: J Clin Med, 2020; 9(4); 1181
29. Lu S, Hu G, Chen S, A nested case-control study: Biomed Res Int, 2020; 2020; 9583706
30. Ruiz-Tovar J, Priego P, Portal vein thrombosis after splenic and pancreatic surgery: Adv Exp Med Biol, 2017; 906; 241-51
31. Fujinaga A, Ohta M, Endo Y, Clinical significance of splenic vessels and anatomical features in laparoscopic splenectomy: J Laparoendosc Adv Surg Tech A, 2020 [Online ahead of print]
32. El-Menyar A, Asim M, Al-Thani H, Obesity paradox in patients with deep venous Thrombosis: Clin Appl Thromb Hemost, 2018; 24; 986-92
33. Mokdad AH, Ford ES, Bowman BA, Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001: JAMA, 2003; 289; 76-79
34. Hendriks SH, Schrijnders D, van Hateren KJ, Association between body mass index and obesity-related cancer risk in men and women with type 2 diabetes in primary care in the Netherlands: a cohort study (ZODIAC-56): BMJ OPEN, 2018; 8; e018859
35. Glise SK, Jern S, Aberg M, Obesity in adolescent men increases the risk of venous thromboembolism in adult life: J Intern Med, 2020; 287; 734-45
36. Severinsen MT, Kristensen SR, Johnsen SP, Anthropometry, body fat, and venous thromboembolism: a Danish follow-up study: Circulation, 2009; 120; 1850-57
37. Borch KH, Braekkan SK, Mathiesen EB, Anthropometric measures of obesity and risk of venous thromboembolism: The Tromso study: Arterioscler Thromb Vasc Biol, 2010; 30; 121-27
38. Ayala R, Grande S, Bustelos R, Obesity is an independent risk factor for pre-transplant portal vein thrombosis in liver recipients: BMC Gastroenterol, 2012; 12; 114
39. Reyes L, Herrero JI, Rotellar SF, Risk factors and impact of portal vein thrombosis in liver transplantation: Rev Esp Enferm Dig, 2019; 111; 437-44
40. Bureau C, Laurent J, Robic MA, Central obesity is associated with non-cirrhotic portal vein thrombosis: J Hepatol, 2016; 64; 427-32
41. Hernandez-Conde M, Llop E, Fernandez-Carrillo C, Visceral fat is associated with cirrhotic portal vein thrombosis: Expert Rev Gastroenterol Hepatol, 2019; 13; 1017-22
Figures
 Figure 1. Flow chart of our study. NU – Nanchang University; ZU – Zhejiang University.
Figure 1. Flow chart of our study. NU – Nanchang University; ZU – Zhejiang University. Figure 2. LASSO feature selection model. (A) LASSO coefficients of 20 candidate variables. (B) Identification of the optimal penalization coefficient (λ) in the LASSO model was achieved by 10-fold cross-validation and the minimum criterion.
Figure 2. LASSO feature selection model. (A) LASSO coefficients of 20 candidate variables. (B) Identification of the optimal penalization coefficient (λ) in the LASSO model was achieved by 10-fold cross-validation and the minimum criterion. Figure 3. The nomogram for prediction of portal vein thrombosis in patients with cirrhosis after splenectomy.
Figure 3. The nomogram for prediction of portal vein thrombosis in patients with cirrhosis after splenectomy. Figure 4. The receiver operating characteristic (ROC) curves for (A) training, (B) internal validation, and (C) independent validation set cohorts.
Figure 4. The receiver operating characteristic (ROC) curves for (A) training, (B) internal validation, and (C) independent validation set cohorts. Figure 5. Calibration curve plot in each set. (A) the training set; (B) the internal validation set; (C) the independent validation set.
Figure 5. Calibration curve plot in each set. (A) the training set; (B) the internal validation set; (C) the independent validation set. Figure 6. Decision curve analysis for the nomogram.
Figure 6. Decision curve analysis for the nomogram. Tables
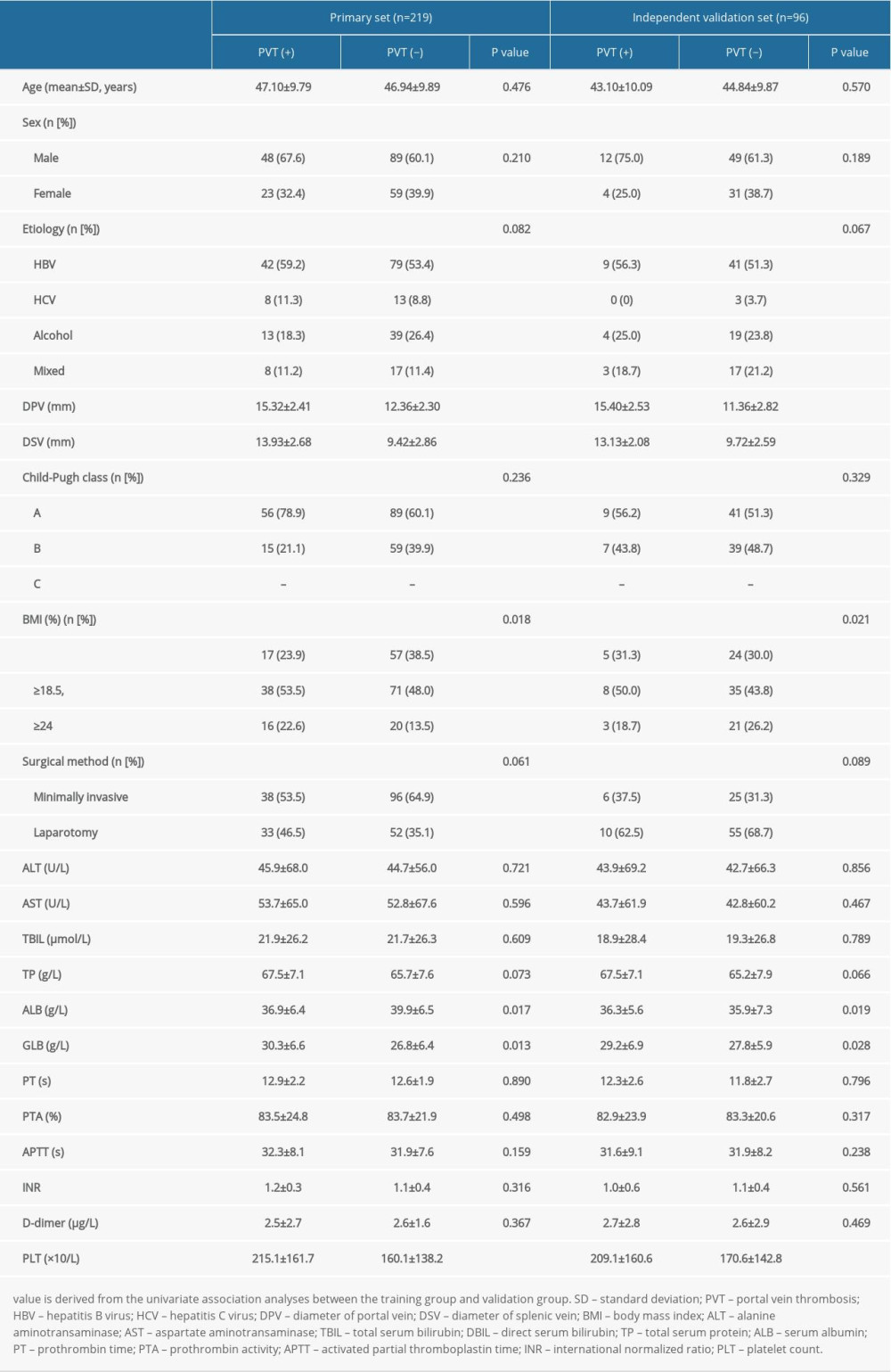 Table 1. Characteristics of patients in the primary and validation cohorts.
Table 1. Characteristics of patients in the primary and validation cohorts.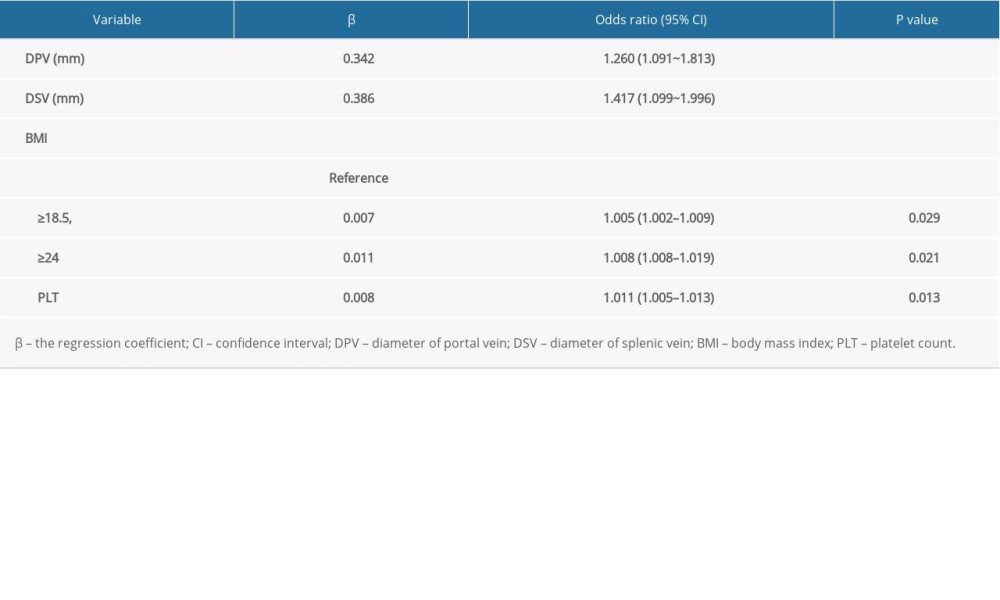 Table 2. Multivariable logistic regression analysis of the selected clinical characteristics in the training group.
Table 2. Multivariable logistic regression analysis of the selected clinical characteristics in the training group. Table 1. Characteristics of patients in the primary and validation cohorts.
Table 1. Characteristics of patients in the primary and validation cohorts. Table 2. Multivariable logistic regression analysis of the selected clinical characteristics in the training group.
Table 2. Multivariable logistic regression analysis of the selected clinical characteristics in the training group. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








