08 April 2021: Review Articles
Current Status of MicroRNAs that Target the Wnt Signaling Pathway in Regulation of Osteogenesis and Bone Metabolism: A Review
Qiang Sun1AEF, Siyu Liu1CD, Jingyi Feng1CE, Yue Kang1BD, You Zhou1E, Shu Guo1A*DOI: 10.12659/MSM.929510
Med Sci Monit 2021; 27:e929510
Abstract
ABSTRACT: The directional differentiation of bone mesenchymal stem cells (BMSCs) is regulated by a variety of transcription factors and intracellular signaling pathways. In the past, it was thought that the directional differentiation of BMSCs was related to transforming growth factors, such as bone morphogenetic protein (BMP) and MAPK pathway. However, in recent years, some scholars have pointed out that the Wnt signaling pathway, which is a necessary complex network of protein interactions for biological growth and development, takes a significant role in this process and plays a major part in regulating the development of osteoblasts by exerting signal transduction into cells. Also, they have proved the Wnt protein therapeutic truly have positive effects on the viability and osteogenic capacity of bone graft. Recent studies have shown that microRNAs (miRNAs) play an important regulatory role in this process. MiRNAs such as miRNA-218, miRNA-335, miRNA-29, microRNA-30 and other miRNAs exert negative or positive effects on some crucial molecules in the Wnt/β-catenin pathway, which in turn affect bone metabolism and osteopathy. Thus, miRNAs have been suggested as therapeutic targets for some metabolic bone diseases. This article aims to provide an update on the current status of microRNAs that target the Wnt signaling pathway in the regulation of osteogenesis and bone metabolism and includes a discussion of future areas of research, which can be a theoretical basis for bone metabolism-related diseases.
Keywords: Bone Diseases, Metabolic, Osteogenesis, Bone Diseases, Bone and Bones, Mesenchymal Stem Cells
Background
MicroRNAs (miRNAs), conserved single-stranded noncoding RNA in eukaryotes, usually consist of 18 to 25 nucleotides. When they bind to the 3′-untranslated regions (3′-UTR) of the target mRNA, they cleave the target chain and inhibit target mRNA translation, finally affecting protein expression. Also, miRNAs can act as post-transcriptional regulators to precisely control cell differentiation by altering the expression of target mRNAs and precisely regulating various differentiation-related factors and receptors.
In general, the Wnt signaling pathway is a well-known signaling cascade that either depends on the β-catenin (canonical pathway) or functions independent of it (noncanonical pathways) [1,2], which has been proven to be essential for the osteogenesis and reduced downstream osteogenic differentiation marker genes, such as Runt-related transcription factor 2 (Runx2), impede the osteogenic process [3–5]. Duan et al [6] concluded that β-catenin, as the central target and an essential component of Wnt/β-catenin signaling pathway, is required for BMSCs to differentiate into osteoblasts, which will differentiate to mature osteocytes, programmatically. This means that promotion or inhibition of β-catenin generation or accumulation would affect bone formation. Kazuhiro et al [7] reported that the canonical Wnt signaling pathway promotes osteogenesis. They discussed the role of Wnt signaling in the bone metabolism and disorders from several aspects, such as the inhibitors of receptors such as DKK-1, sclerostin ZNRF3, and RNF43, and they explored the role of sclerostin in the integral bone metabolism. Moreover, they proposed that those receptors or inhibitors can be used as targets to treat bone metabolic disorders such as osteoporosis, osteoarthritis, rheumatoid arthritis, neoplasms, and multiple myeloma. Artificial antagonists may cure these disorders, but there are also other problems, because the Wnt signaling pathway also plays a vital role in cancer stem cell survival.
Recent studies have reported that various important molecules in the Wnt signaling pathway can be targeted and regulated by some miRNAs; interestingly, one miRNA seem to have several target gene [8,9]. Amjadi-Moheb et al [8] concluded that some miRNAs target to the ligands, receptors, antagonists, and intercellular molecules. For example, Wnt1, Wnt3, Wnt5A, DKK-1, SFRP1, and APC can directly activate the Wnt signaling pathway. Activation or inhibition of the Wnt signaling pathway and expression of specific miRNAs are closely related to the development of osteogenesis and metabolic osteopathy as the disease-causing gene or disease-treating gene [10–13]. Hence, this review focuses on miRNAs binding to the Wnt signaling pathway and discusses how miRNAs affect osteogenesis and bone metabolism. We discuss the current understanding of microRNAs that target the Wnt signaling pathway in the regulation of osteogenesis and bone metabolism, and consider future areas of research. We hope this article will provide a theoretical basis for understanding bone metabolism-related diseases.
Wnt Signaling Pathway
At present, there are 19 members in the Wnt family, which are highly conserved secreted glycoproteins [14]. Based on downstream differences, Wnt proteins are further divided to canonical Wnt proteins and noncanonical Wnt proteins; the former, such as Wnt1, Wnt2, Wnt3, Wnt3a, can interact with LRP/FZD to activate Wnt/β-catenin signaling pathway; in contrast, the noncanonical Wnt proteins, including Wnt 4, Wnt 5, Wnt 6, Wnt 7a, and Wnt 11, function by binding to different receptors. Moreover, due to the downstream differences, noncanonical Wnt signaling is further divided to the Wnt Planar Cell Polarity pathway and Wnt/Ca2+ pathway; the PCP pathway relies on FZDs and Dvl and activate RhoA and JNK to develop its effects, while the Wnt/Ca2+pathway activate heterotrimeric G protein and change the intracellular Ca2+concentration [15–17]. Thus, the Wnt signaling pathway is also divided into canonical and noncanonical signal pathways.
Canonical Wnt Signaling Pathway
In the canonical Wnt signaling pathway, the Wnt protein is combined with an amboceptor molecule consisting of LRP5/6 and curled Frizzled protein with a cysteine-rich domain. After the binding signaling is transmitted from the intracellular part of the receptor, the intracellular tail of LRP5/6 binds to axin, resulting in the separation of β-catenin from its protein complex, and finally, β-catenin-mediated downstream signaling is activated [18]. The β-catenin is then moved to the nucleus where they bind to the T cell factor/lymphoid enhancer factor (TCF/LEF) family and regulate them as coactivators [19]. The canonical Wnt signaling pathway plays a crucial role in osteogenic precursor cells differentiation, calcification balance, induction of growth of potential cells and osteogenic precursor cells, and maintenance of osteoblasts and osteocytes activity [20].
Noncanonical Wnt Signaling Pathway
The planar cell polarity pathway (Wnt/PCP pathway), which is known for its polarized cell plane, is different from others to some extent. Wnt protein binds to Frizzled and its co-receptor (Ror 2 or Ryk), so the activated cytoskeletal protein Rho-associated protein kinase (ROCK) activates c-Jun N-terminal kinase (JNK) and controls the downstream transcriptional factors [21]. In the noncanonical Wnt/calcium signaling pathway, calcium ions play an important role in signal transduction [22]. When Wnt protein binds Frizzled, the trimeric protein Dsh-Axin-GSK complex can subsequently mediate the phosphorylation of transmembrane receptor 1/2 of tyrosine kinase (Ror 1/2). The formation of Wnt, Frizzled and Ror 1/2 complexes activate phospholipase C (PLC), which leads to an increase of inositol triphosphate (IP3), diacylglycerol (DAG), and intracellular calcium, and eventually changes downstream cellular functions [23].
Biological Function of miRNAs
miRNAs are conserved single-stranded short (18–25 nucleotides) RNAs that exist in eukaryotic organisms. miRNAs can specifically bind to the 3′-UTR of target mRNA, cleave the mRNA or inhibit protein translation, thereby inhibiting protein expression. Actually, there is a complex regulatory network between miRNAs and mRNAs due to the diversity of target genes and miRNAs existing in clusters. Even though there are considerable homologous sequences in many miRNAs, miRNAs can specifically and effectively recognize the target sequence of 3-UTR of mRNA. Hence, the diversity of target mRNAs complicates the regulatory network [24,25]. However, more than 36% of the miRNAs in the human genome are present in the cluster, while this proportion up to 45% in mice [26]. The transcription of the same cluster of miRNAs is also regulated by upstream sequences. The primary transcription products are located on 1 strand. Multiple mature miRNAs formed after cleaving may belong to different families and regulate mRNAs with different target sequences. The position of miRNA in the organism is also not fixed, and it is scattered in various regions of the genome. The introns of eukaryotic genes, which have been widely regarded as useless, can also be transcribed and sheared to form miRNA molecules. The online miRNA database (
Role of miRNAs in Osteogenesis by Regulating the Wnt Signaling Pathway
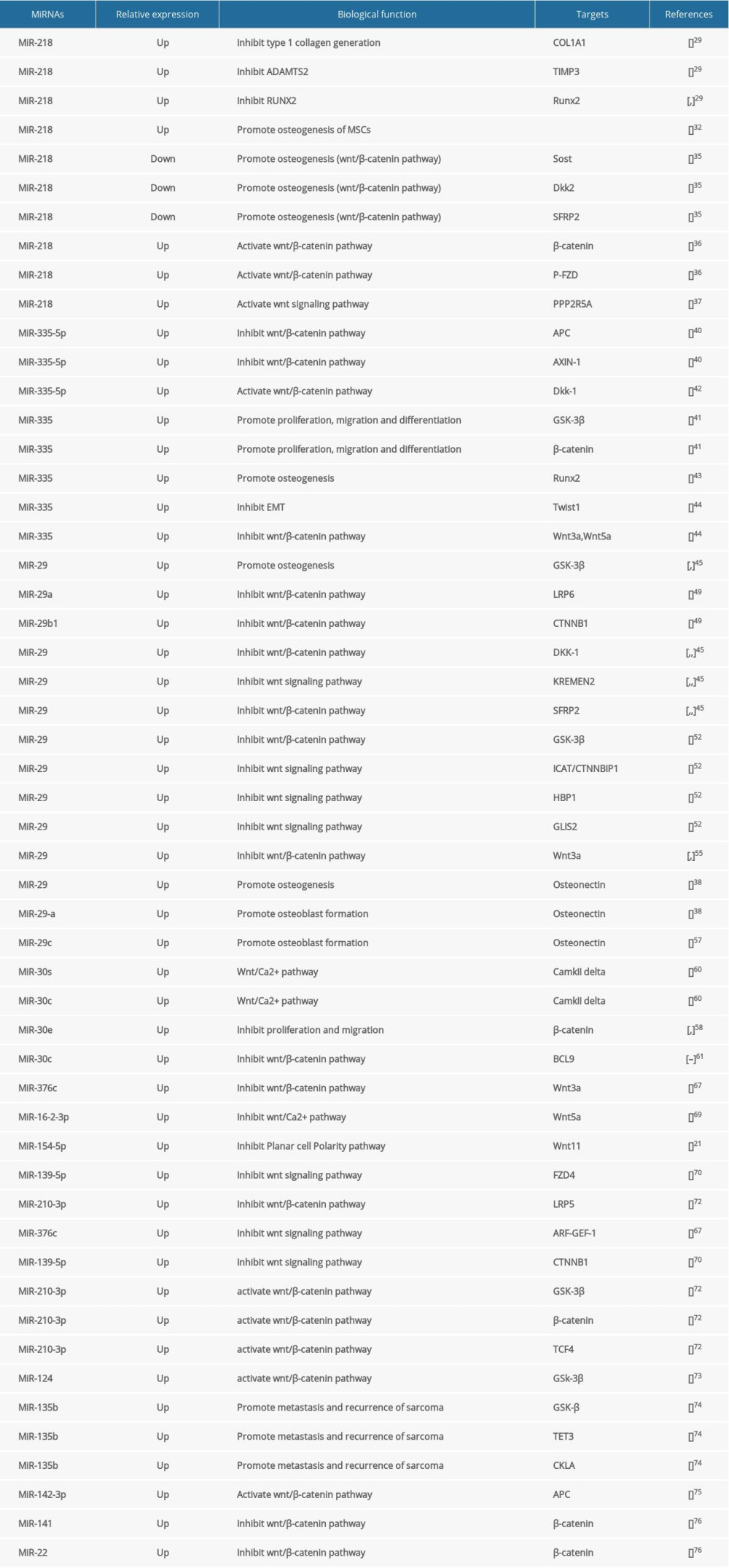
There are 3 ways in which miRNAs regulate the Wnt signaling pathway (Table 1): (1) miRNAs can target mRNA as an antagonist of the Wnt signaling pathway, such as Dickkopf-related proteins (DKKs) and SFRPs; (2) miRNAs regulate the expression of osteogenesis-related genes by affecting membrane proteins and receptors binding, and subsequently affect the formation of the tetramer APC/Axin/GSK-3β/β-catenin or independently affect the individual members of the tetramer; and (3) both of the above. Here, we review the miRNAs that regulate cellular biological activities via the Wnt signaling pathway.
miRNA-218
The role of miRNA-218 in osteogenesis is unclear. Xuxiang Liu et al reported that miRNA-218 inhibits the deposition of type 1 collagen through directly downregulating COL1A1 expression and promotes TIMP3, an inhibitor of the N-procollagenase ADAMTS2 [29]. In addition to type 1 collagen, miR-218 also target to Runx2, the key protein in osteogenesis. Yao and Zhou et al proved that miR-218 inhibits Runx2, which suggests that miR-218 inhibits the osteogenic process [30,31]. Surprisingly, miRNA-218 seems to promote osteogenic differentiation of bone marrow stem cells (BMSC) by stimulating alkaline phosphatase activity, calcium nodule formation, and osteogenic marker gene expression [32]. Furthermore, Hassan et al found the positive effect of miRNA-218 contributes to activation of the Wnt signaling pathway in the differentiation of MSCs and breast cancer cells [33,34]. Hassan et al reported that miRNA-218 can not only promote the directional differentiation of BMSCs into osteoblasts and further differentiate in maturation, but also suppress negative regulators of the Wnt signaling pathway, such as sclerostin (SOST), DKK2, and secreted Frizzled-related protein 2 (SFRP2), thus leading to activation of the Wnt pathway [35]. In addition, miRNA-218 also promotes β-catenin, phosphate-FZD, and PPP2R5A generation and affects cellular biological activity [36,37]. Moreover, the activated Wnt pathway with miRNA-218 assembles a positive feedback loop; in other words, the activated Wnt pathway has a positive effect in the expression of miRNA-218. In this way, regeneration feedback forms between miRNA-218 and the Wnt signaling pathway to facilitate differentiation of BMSCs into osteoblasts. Another study found that miRNA-218 expression can increase secretion of Wnt-targeted parathyroid hormone-related protein (PTHrP) and enhance Wnt signaling, thereby facilitating osteoclast differentiation and bone resorption [38]. Additional evidence shows miRNA-218 is an activator of the Wnt signaling pathway, but it may not regulate the osteogenic process of stem cells due to its inhibition of osteogenesis.
miRNA-335
The miR-335 family consists of miR-335-3p and miR-335-5p in humans [39]. Kay [40] found that miRNA-335-3p and miRNA-5p are both interrelated with the Wnt signaling pathway. RT-PCR and luciferase assay showed that miRNA-335-5p targets the 3′UTR of APC and AXIN-1, which are the 2 main inhibitory proteins of the canonical Wnt signaling pathway. When miR-335 develop its sponge function by absorbing APC and AXIN-1, the Wnt pathway is enhanced. To a certain extent, miRNA-335 can activate the Wnt signaling pathway, but the evidence is unclear. Studies demonstrated that miRNA-335-5p controls the DKK1 expression and increases GSK-3β phosphorylation and β-catenin transcriptional activity, finally activating the Wnt signaling pathway both in vivo and in vitro, and miRNA-335 promotes osteogenesis of mouse BMSCs. Tome et al found that miR-335 can sponge Runx2 and inhibit osteogenesis [41–43]. In contrast, the Wnt signaling pathway was inhibited when miR-335-5p was reduced by anti-miR-335-5p. miR-335-suppresses HCT116 cell synthesis more than Wnt3a, Wnt5a, and β-catenin, which means knockdown miR-335 can activate the Wnt signaling pathway [44]. These experiments indicate the crucial effects of miR-335-5p in regulation of the Wnt signaling pathway and have great potential in osteogenesis.
miRNA-29
The miR-29 family of microRNAs is highly conserved among mammalian species. Its members, including miRNA-29a, miRNA-29b-1, miRNA-29b-2, and miRNA-29c, have as well been investigated for their roles in modulation of somatic cell fate reprogramming by targeting the Wnt signaling pathway [45–48]. Case et al used LiC1 to downregulate the activity of GS3-β, allowing the accumulation of β-catenin and acceleration of osteoblasts differentiation. When m0b-I2 cells were treated with LiC1, the Wnt signaling was activated and the expression of miRNA-29 was further enhanced; the expression peak of miRNA-29 occurred 3 h after treatment. A similar result was reported by Mariane Serra Fráguas et al [48], who found the miRNA-29 family is directly targeted to GSK-3β in MEF-cells. Meanwhile, miRNA-29a/b-1 was reported to have special binding sites and sponge the mRNA of Lrp6 and Ctnnb1, thus suppressing the Wnt signaling pathway [49]. Moreover, DKK1, Kremen2, and SFRP2 are also targets of miRNA-29 [39,50,51]. Indeed, 4 factors of the Wnt signaling pathway contain binding sites for miR-29 – GSK3B, ICAT/CTNNBIP1, HBP1, and GLIS2 – finally induce Wnt signaling pathway suppression [52]. In 2014, it was reported for the first time that the expression ofmiRNA-29a in peripheral blood mononuclear cells (PBMCs) of AS patients was increased and may be a useful biological marker of bone formation [53,54]. However, the ligand of the Wnt pathway has a positive effect on the generation of miRNA-29, such as Wnt3a [55,56].
The miRNA-29 family can affect other key proteins in osteogenesis. The complementary sequences of miRNA-29a can target the 3′-UTR regions of osteonectin to regulate the expression of osteonectin, which in turn affects maturation and mineralization of the extracellular matrix. In addition to miRNA-29a, miRNA-29c also significantly suppresses the expression of osteonectin and further promotes osteoblast differentiation [38]. However, there is a positive effect on mineral deposition, which occurs in the late stage of bone formation [57]. Above all, the miRNA-29 family promotes osteogenesis, but it is unclear whether the positive effect is related to the Wnt signaling pathway, as there are conflicting reports on the effect of miRNA-29 family on the Wnt signaling pathway.
miRNA-30
miR-30, a family of calcium/calcineurin signaling miRs, is evolutionarily conserved and consists of 5 members: miR-30a, miR-30b, miR-30c, miR-30d, and miR-30e [58,59]. miR-30s are essential regulators of calcium/calcineurin signaling, but whether they affect the Wnt/Ca2+ signal pathway is still unclear. CaMKIIδ is the key protein downstream of the Wnt/Ca2+ pathway, whose mRNA is sponged by miR-30c or miRNA-30e, which inhibits VSM cell proliferation and migration [60]. B cell lymphoma-9 (BCL-9) is a transcription activator of β-catenin [61]. Zhao et al [62,63] found that BCL-9 is a target of miRNA-30c. The expression of BCL-9 was increased and the Wnt signaling pathway was activated. It seems that miRNA-30 is an inhibitor of osteogenesis, while the positive effector in the osteogenic process transforming growth factor (TGF) -β1 harms miRNA-30 [64–66]. miRNA-30 appears to have an insignificant effect on the Wnt signaling pathway, but the negative effect on osteogenesis is evident. Thus, there is a need for extensive and in-depth research on the potential links among the Wnt signaling pathway, osteogenesis, and miR-30 family to reveal new view information on bone regeneration.
Other MicroRNAs
Among the whole Wnt signaling pathway, the ligand, receptor, key protein, and transcription factor are affected by different miRNAs. The canonical and noncanonical Wnt signaling pathway ligands, such as Wnt3, Wnt5a, and Wnt 11, are affected by miR-376c, miRNA-26a-5p/miRNA-16-2*, and miRNA-154-5p, respectively [21,67–69]. Once these ligands of the Wnt signaling pathway decrease or are deficient, there is downstream inhibition. The receptors FZD4, ROR1/2, and LRP5 are also sponged by miRNA-139-5p, miRNA-214, and miR-210-3p, respectively [70,71]. Meanwhile, these MicroRNAs seem to have multiple target genes. ARF guanine nucleotide exchange factor-1 (ARF-GEF-1) is also targeted by miRNA-376c and further inhibits transactivation of β-catenin, finally inhibiting osteogenesis [21], and CTNNB1 (the gene name of β-catenin) is sponged by miR-139-5p [70]. When miR-210-3p is overexpressed, the key regulatory factors of the Wnt signaling pathway – GSK-3β, β-catenin, and TCF4 – are significantly increased, and finally promote bone formation [72]. Meanwhile, the expression of miRNA-124 was increased in ligament tissue of AS patients, and the expression of GSK3β was inhibited, thereby enhancing Wnt/β-catenin signaling pathway activity and accelerating the osteogenesis from fibroblasts [73]. GSK-3β, TET3, and CKla are suppressed by miRNA-135b, which leads to metastasis and recurrence of sarcoma [74]. miRNA-142-3p is found to increase in MSCs differentiated into osteoblasts and inhibit the expression of APC and activate the Wnt signaling pathway [75]. Moreover, miRNA-141 and miRNA-22 directly decrease β-catenin expression and decrease the osteogenic marker, finally inhibiting osteogenesis of BMSCs [76].
Future Research
Those microRNAs that we highlighted may help or hinder the osteogenesis of stem cells. The present review shows that microRNAs can target and bind to several genes, as shown by Suh Yee Goh, making miRNA a potential regulator of osteogenesis. Therefore, understanding the mechanism involved in the imbalance of miRNA in bone metastatic diseases or bone formation would promote the development of bone tissue engineering. The key pathways that regulate osteoblast differentiation of stem cells include Wnt/β-catenin, PI3k/Akt, TGF-β/Smad, BMP/Smad [23], and MAPK pathways, which are reported to be targets of diverse miRNAs. The Wnt signaling pathway is the most important pathway involved in osteogenesis. As reported, these miRNAs activate or inhibit the Wnt pathway and thus affect bone metastasis. However, in different cell lines, microRNAs may have different targets, which can influence the final results, and can even have completely opposite results. So, miRNAs in different fields may not have a common outcome. Furthermore, miRNA research may be a double-edged sword. miRNAs are powerful regulators of gene expression and can modulate the process of stem cells differentiation by regulating different pathways, with or without interaction with other modulatory molecules. Although the Wnt signaling pathway is targeted by many microRNAs, the evidence is scant and there is a shortage of animal experiments. The local microenvironment is very different from the systemic environment, and diverse factors are involved in media, which may promote or inhibit stem cells osteogenesis. It is undeniable that microRNA strongly affects osteogenesis by targeting the Wnt signaling pathway. MicroRNA has strong potential for use in bone formation, but the disadvantages of miRNA therapy are that it is easily degraded and hard to transport, and the diverse targets of a single microRNA may have different outcomes. Finally, the current experimental approaches used in miRNA research are still limited. Hence, we need to focus more on miRNAs research.
Conclusions
This review has presented an update on the current status of miRNAs that target the Wnt signaling pathway in the regulation of osteogenesis and bone metabolism. The key miRNAs that regulate the function of the miRNA receptors and inhibitors include miRNA-218, miRNA-335, miRNA-29, and miRNA-30. However, ongoing studies on osteogenesis, bone metabolism, and the Wnt signaling pathway continue to identify roles of miRNAs, which may contribute to miRNA therapy for some metabolic bone diseases.
References
1. Hata A, Kang H, Functions of the bone morphogenetic protein signaling pathway through microRNAs (review): Int J Mol Med, 2015; 35(3); 563-68
2. Kang H, Hata A, The role of microRNAs in cell fate determination of mesenchymal stem cells: balancing adipogenesis and osteogenesis: BMB Rep, 2015; 48(6); 319-23
3. Arfat Y, Xiao WZ, Ahmad M, Role of microRNAs in osteoblasts differentiation and bone disorders: Curr Med Chem, 2015; 22(6); 748-58
4. Mahmood S, Bhatti A, Syed NA, John P, The microRNA regulatory network: A far-reaching approach to the regulate the Wnt signaling pathway in number of diseases: J Recept Signal Transduct Res, 2016; 36(3); 310-18
5. Hassan MQ, Tye CE, Stein GS, Lian JB, Non-coding RNAs: Epigenetic regulators of bone development and homeostasis: Bone, 2015; 81; 746-56
6. Duan P, Bonewald LF, The role of the wnt/β-catenin signaling pathway in formation and maintenance of bone and teeth: Int J Biochem Cell Biol, 2016; 77(Pt A); 23-29
7. Kobayashi Y, Uehara S, Udagawa N, Takahashi N, Regulation of bone metabolism by Wnt signals: J Biochem, 2016; 159(4); 387-92
8. Gao Y, Patil S, Qian A, The role of microRNAs in bone metabolism and disease: Int J Mol Sci, 2020; 21(17); 6081
9. Amjadi-Moheb F, Akhavan-Niaki H, Wnt signaling pathway in osteoporosis: Epigenetic regulation, interaction with other signaling pathways, and therapeutic promises: J Cell Physiol, 2019 [Online ahead of print]
10. Fang S, Deng Y, Gu P, Fan X, MicroRNAs regulate bone development and regeneration: Int J Mol Sci, 2015; 16(4); 8227-53
11. Xie Y, Zhang L, Gao Y, The multiple roles of microrna-223 in regulating bone metabolism: Molecules, 2015; 20(10); 19433-48
12. Chen J, Qiu M, Dou C, MicroRNAs in bone balance and osteoporosis: Drug Dev Res, 2015; 76(5); 235-45
13. Song JL, Nigam P, Tektas SS, Selva E, microRNA regulation of Wnt signaling pathways in development and disease: Cell Signal, 2015; 27(7); 1380-91
14. Kikuchi A, Yamamoto H, Sato A, Matsumoto S, Wnt5a: Its signalling, functions and implication in diseases: Acta Physiol (Oxf), 2012; 204(1); 17-33
15. Offermanns S, Rosenthal W, Planar Cell Polarity Pathway: Encyclopedia of molecular pharmacology, 2008; 984, Berlin, Heidelberg:, Springer Berlin Heidelberg
16. Okerlund ND, Cheyette BNR, Wnt Signaling: Encyclopedia of molecular pharmacology, 2008; 1316-22, Berlin, Heidelberg, Springer Berlin Heidelberg
17. Cheng CW, Yeh JC, Fan TP, Wnt5a-mediated non-canonical Wnt signalling regulates human endothelial cell proliferation and migration: Biochem Biophys Res Commun, 2008; 365(2); 285-90
18. Xie W, Zhou L, Li S, Wnt/beta-catenin signaling plays a key role in the development of spondyloarthritis: Ann NY Acad Sci, 2016; 1364; 25-31
19. Behrens J, von Kries JP, Kuhl M, Functional interaction of beta-catenin with the transcription factor LEF-1: Nature, 1996; 382(6592); 638-42
20. Khosla S, Westendorf JJ, Oursler MJ, Building bone to reverse osteoporosis and repair fractures: J Clin Invest, 2008; 118(2); 421-28
21. Li J, Hu C, Han L, MiR-154-5p regulates osteogenic differentiation of adipose-derived mesenchymal stem cells under tensile stress through the Wnt/PCP pathway by targeting Wnt11: Bone, 2015; 78; 130-41
22. De A, Wnt/Ca2+ signaling pathway: A brief overview: Acta Biochim Biophys Sin (Shanghai), 2011; 43(10); 745-56
23. Spreafico A, Tentler JJ, Pitts TM, Rational combination of a MEK inhibitor, selumetinib, and the Wnt/calcium pathway modulator, cyclosporin A, in preclinical models of colorectal cancer: Clin Cancer Res, 2013; 19(15); 4149-62
24. Small EM, Olson EN, Pervasive roles of microRNAs in cardiovascular biology: Nature, 2011; 469(7330); 336-42
25. Heinrich EM, Dimmeler S, MicroRNAs and stem cells: Control of pluripotency, reprogramming, and lineage commitment: Circ Res, 2012; 110(7); 1014-22
26. Griffiths-Jones S, Saini HK, van Dongen S, Enright AJ, miRBase: Tools for microRNA genomics: Nucleic Acids Res, 2008; 36(Database issue); D154-58
27. Kozomara A, Birgaoanu M, Griffiths-Jones S, miRBase: From microRNA sequences to function: Nucleic Acids Research, 2018; 47(D1); D155-62
28. Jiang Q, Wang Y, Hao Y, miR2Disease: A manually curated database for microRNA deregulation in human disease: Nucleic Acids Research, 2009; 37; D98-104
29. Liu X, Cao M, Palomares M, Metastatic breast cancer cells overexpress and secrete miR-218 to regulate type I collagen deposition by osteoblasts: Breast Cancer Res, 2018; 20(1); 127
30. Zhou MH, Zhang L, Song MJ, Sun WJ, MicroRNA-218 prevents lung injury in sepsis by inhibiting RUNX2: Eur Rev Med Pharmacol Sci, 2018; 22(23); 8438-46
31. Yao R, Yao X, Liu R, Glucose-induced microRNA-218 suppresses the proliferation and promotes the apoptosis of human retinal pigment epithelium cells by targeting RUNX2: Biosci Rep, 2019; 39(12); BSR20192580
32. Shi L, Feng L, Liu Y, MicroRNA-218 promotes osteogenic differentiation of mesenchymal stem cells and accelerates bone fracture healing: Calcif Tissue Int, 2018; 103(2); 227-36
33. Hassan MQ, Maeda Y, Taipaleenmaki H, miR-218 directs a Wnt signaling circuit to promote differentiation of osteoblasts and osteomimicry of metastatic cancer cells: J Biol Chem, 2012; 287(50); 42084-92
34. Taipaleenmaki H, Farina NH, van Wijnen AJ, Antagonizing miR-218-5p attenuates Wnt signaling and reduces metastatic bone disease of triple negative breast cancer cells: Oncotarget, 2016; 7(48); 79032-46
35. Qin Y, Peng Y, Zhao W, Myostatin inhibits osteoblastic differentiation by suppressing osteocyte-derived exosomal microRNA-218: A novel mechanism in muscle-bone communication: J Biol Chem, 2017; 292(26); 11021-33
36. Hu F, Sun B, Xu P, MiR-218 induces neuronal differentiation of ASCs in a temporally sequential manner with fibroblast growth factor by regulation of the Wnt signaling pathway: Sci Rep, 2017; 7; 39427
37. Zhuang Z, Hu F, Hu J, MicroRNA-218 promotes cisplatin resistance in oral cancer via the PPP2R5A/Wnt signaling pathway: Oncol Rep, 2017; 38(4); 2051-61
38. Kapinas K, Kessler CB, Delany AM, miR-29 suppression of osteonectin in osteoblasts: Regulation during differentiation and by canonical Wnt signaling: J Cell Biochem, 2009; 108(1); 216-24
39. Landgraf P, Rusu M, Sheridan R, A mammalian microRNA expression atlas based on small RNA library sequencing: Cell, 2007; 129(7); 1401-14
40. Kay M, Soltani BM, Aghdaei FH, Hsa-miR-335 regulates cardiac mesoderm and progenitor cell differentiation: Stem Cell Res Ther, 2019; 10(1); 191
41. Tome M, Lopez-Romero P, Albo C, miR-335 orchestrates cell proliferation, migration and differentiation in human mesenchymal stem cells: Cell Death Differ, 2011; 18(6); 985-95
42. Zhang J, Tu Q, Bonewald LF, Effects of miR-335-5p in modulating osteogenic differentiation by specifically downregulating Wnt antagonist DKK1: J Bone Miner Res, 2011; 26(8); 1953-63
43. Zhang L, Tang Y, Zhu X, Overexpression of MiR-335-5p promotes bone formation and regeneration in mice: J Bone Miner Res, 2017; 32(12); 2466-75
44. Wang J, Wang X, Liu F, Fu Y, microRNA-335 inhibits colorectal cancer HCT116 cells growth and epithelial-mesenchymal transition (EMT) process by targeting Twist1: Pharmazie, 2017; 72(8); 475-81
45. Kapinas K, Kessler C, Ricks T, miR-29 modulates Wnt signaling in human osteoblasts through a positive feedback loop: J Biol Chem, 2010; 285(33); 25221-31
46. Le LT, Swingler TE, Crowe N, The microRNA-29 family in cartilage homeostasis and osteoarthritis: J Mol Med (Berl), 2016; 94(5); 583-96
47. Li SC, Wang FS, Yang YL, Microarray study of pathway analysis expression profile associated with MicroRNA-29a with regard to murine cholestatic liver injuries: Int J Mol Sci, 2016; 17(3); 324
48. Fraguas MS, Eggenschwiler R, Hoepfner J, MicroRNA-29 impairs the early phase of reprogramming process by targeting active DNA demethylation enzymes and Wnt signaling: Stem Cell Res, 2017; 19; 21-30
49. Ge M, Liu C, Li L, miR-29a/b1 inhibits hair follicle stem cell lineage progression by spatiotemporally suppressing WNT and BMP signaling: Cell Rep, 2019; 29(8); 2489-504.e4
50. Li C, Zhang P, Gu J, miR-29a modulates tumor necrosis factor-alpha-induced osteogenic inhibition by targeting Wnt antagonists: Dev Growth Differ, 2015; 57(3); 264-73
51. Huang J, Song G, Yin Z, MiR-29a and messenger RNA expression of bone turnover markers in canonical Wnt pathway in patients with ankylosing spondylitis: Clin Lab, 2017; 63(5); 955-60
52. Sassi Y, Avramopoulos P, Ramanujam D, Cardiac myocyte miR-29 promotes pathological remodeling of the heart by activating Wnt signaling: Nat Commun, 2017; 8(1); 1614
53. Huang J, Song G, Yin Z, Altered expression of microRNAs targeting Dkk-1 in peripheral blood mononuclear cells of patients with ankylosing spondylitis: Cent Eur J Immunol, 2019; 44(1); 59-64
54. Huang J, Song G, Yin Z, Elevated miR-29a expression is not correlated with disease activity index in PBMCs of patients with ankylosing spondylitis: Mod Rheumatol, 2014; 24(2); 331-34
55. Hu Z, Klein JD, Mitch WE, MicroRNA-29 induces cellular senescence in aging muscle through multiple signaling pathways: Aging (Albany NY), 2014; 6(3); 160-75
56. Case N, Ma M, Sen B, Beta-catenin levels influence rapid mechanical responses in osteoblasts: J Biol Chem, 2008; 283(43); 29196-205
57. Roberto VP, Tiago DM, Silva IA, Cancela ML, MiR-29a is an enhancer of mineral deposition in bone-derived systems: Arch Biochem Biophys, 2014; 564; 173-83
58. Shi M, Mu Y, Zhang H, MicroRNA-200 and microRNA-30 family as prognostic molecular signatures in ovarian cancer: A meta-analysis: Medicine (Baltimore), 2018; 97(32); e11505
59. Wu J, Zheng C, Wang X, MicroRNA-30 family members regulate calcium/calcineurin signaling in podocytes: J Clin Invest, 2015; 125(11); 4091-106
60. Liu YF, Spinelli A, Sun LY, MicroRNA-30 inhibits neointimal hyperplasia by targeting Ca(2+)/calmodulin-dependent protein kinase IIdelta (CaMKIIdelta): Sci Rep, 2016; 6; 26166
61. Kramps T, Peter O, Brunner E, Wnt/wingless signaling requires BCL9/legless-mediated recruitment of pygopus to the nuclear beta-catenin-TCF complex: Cell, 2002; 109(1); 47-60
62. Zhao JJ, Lin J, Zhu D, miR-30-5p functions as a tumor suppressor and novel therapeutic tool by targeting the oncogenic Wnt/beta-catenin/BCL9 pathway: Cancer Res, 2014; 74(6); 1801-13
63. Zhao JJ, Carrasco RD, Crosstalk between microRNA30a/b/c/d/e-5p and the canonical Wnt pathway: Implications for multiple myeloma therapy: Cancer Res, 2014; 74(19); 5351-58
64. Zhang Z, Zhang X, Zhao D, TGF-β1 promotes the osteoinduction of human osteoblasts via the PI3K/AKT/mTOR/S6K1 signalling pathway: Mol Med Rep, 2019; 19(5); 3505-18
65. Asparuhova MB, Caballé-Serrano J, Buser D, Chappuis V, Bone-conditioned medium contributes to initiation and progression of osteogenesis by exhibiting synergistic TGF-β1/BMP-2 activity: Int J Oral Sci, 2018; 10(2); 20
66. Zhang X, Dong S, Jia Q, The microRNA in ventricular remodeling: The miR-30 family: Biosci Rep, 2019; 39(8); BSR20190788
67. Kureel J, John AA, Prakash R, Singh D, MiR 376c inhibits osteoblastogenesis by targeting Wnt3 and ARF-GEF-1 -facilitated augmentation of beta-catenin transactivation: J Cell Biochem, 2018; 119(4); 3293-303
68. Zhang C, Hu Y, Wan J, He H, MicroRNA-124 suppresses the migration and invasion of osteosarcoma cells via targeting ROR2-mediated non-canonical Wnt signaling: Oncol Rep, 2015; 34(4); 2195-201
69. Duan L, Zhao H, Xiong Y, miR-16-2* interferes with WNT5A to regulate osteogenesis of mesenchymal stem cells: Cell Physiol Biochem, 2018; 51(3); 1087-102
70. Long H, Sun B, Cheng L, miR-139-5p represses BMSC osteogenesis via targeting Wnt/beta-catenin signaling pathway: DNA Cell Biol, 2017; 36(8); 715-24
71. Wei R, Deng Z, Su J, miR-217 targeting Wnt5a in osteosarcoma functions as a potential tumor suppressor: Biomed Pharmacother, 2015; 72; 158-64
72. Li X, Peng B, Zhu X, MiR-210-3p inhibits osteogenic differentiation and promotes adipogenic differentiation correlated with Wnt signaling in ERalpha-deficient rBMSCs: J Cell Physiol, 2019; 234(1); 23475-84
73. Tang SL, Huang QH, Wu LG, MiR-124 regulates osteoblast differentiation through GSK-3beta in ankylosing spondylitis: Eur Rev Med Pharmacol Sci, 2018; 22(20); 6616-24
74. Jin H, Luo S, Wang Y, miR-135b stimulates osteosarcoma recurrence and lung metastasis via Notch and Wnt/beta-catenin signaling: Mol Ther Nucleic Acids, 2017; 8; 111-22
75. Hu W, Ye Y, Zhang W, miR1423p promotes osteoblast differentiation by modulating Wnt signaling: Mol Med Rep, 2013; 7(2); 689-93
76. Liang WC, Fu WM, Wang YB, H19 activates Wnt signaling and promotes osteoblast differentiation by functioning as a competing endogenous RNA: Sci Rep, 2016; 6; 20121
In Press
05 Mar 2024 : Clinical Research
Muscular Function Recovery from General Anesthesia in 132 Patients Undergoing Surgery with Acceleromyograph...Med Sci Monit In Press; DOI: 10.12659/MSM.942780
05 Mar 2024 : Clinical Research
Effects of Thermal Insulation on Recovery and Comfort of Patients Undergoing Holmium Laser LithotripsyMed Sci Monit In Press; DOI: 10.12659/MSM.942836
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952