28 April 2021: Clinical Research
Multi-Phase Multiple Detector Computed Tomography (MDCT) Enhancement Patterns and Morphological Features of Chromophobe Renal Cell Carcinoma: An Analysis of 67 Cases
Min Luo12EFG, Yuting Zhu12DEF, Shaobin Chen12BC, Qilin Huang3BCD, Wei Zhang12CDF, Mingping Ma12ABD, Yongbao Wei14AG*DOI: 10.12659/MSM.929287
Med Sci Monit 2021; 27:e929287
Abstract
BACKGROUND: Chromophobe renal cell carcinoma (ChRCC) is difficult to diagnose preoperatively. We investigated multiple detector computed tomography (MDCT) plain scan and multi-phase CT enhancement features to aid ChRCC preoperative diagnosis.
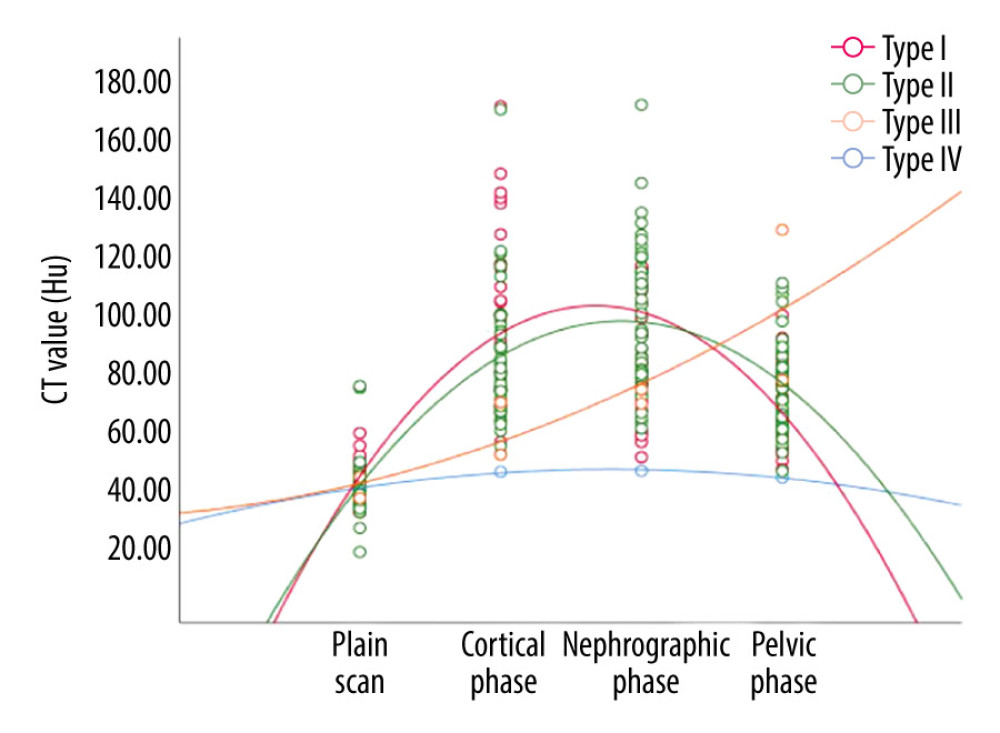
MATERIAL AND METHODS: MDCT data of patients with pathologically confirmed ChRCC were retrospectively analyzed. We calculated the ratios of the CT value for the solid part of the mass to those of the renal cortex, aorta, and inferior vena cava. These ratios were designated as L01-3 for the CT plain scan images, La1-3 for the cortical phase, Lv1-3 for the nephrographic phase, and Lp1-3 for the pelvic phase. We classified the masses into types I, II, III, and IV by type of enhancement.
RESULTS: Sixty-eight masses were included and divided into 3 groups by tumor size (groups A, B, and C). Percentages of calcification, central scars, and small vessel signs were significantly different during the cortical phase for masses in all groups (all P<0.01). Significant differences in enhancement were observed between tumors with severe and mild degrees of enhancement (P<0.01); and among La1, Lv1, and Lp1; La2, Lv2, and Lp2; and La3, Lv3, and Lp3 after enhancement during the cortical, nephrographic, and renal pelvic phases (all P<0.01). The most common type of mass enhancement was type II, followed by type I, and differences between these 2 types were significant (P<0.001).
CONCLUSIONS: Although the MDCT features for ChRCC are diverse, MDCT helped preoperatively diagnose ChRCC. Multiple MDCT features are needed to improve the accuracy of preoperative diagnosis.
Keywords: Carcinoma, Renal Cell, multidetector computed tomography, preoperative period, Diagnosis, Differential, Kidney, Kidney Neoplasms, Vena Cava, Inferior
Background
Chromophobe renal cell carcinoma (ChRCC), which was first defined by the World Health Organization in 2004, accounts for 6% to 8% of renal cell carcinoma (RCC) cases [1]. ChRCC originates from type B insert cells in the renal collecting duct cortex. Most of the cancer cells differentiate into grades I and II, and the cell proliferation index for ChRCC is lower than that of most kidney cancers [2]. The clinical stage at diagnosis is usually early, and ChRCC has a low recurrence and metastasis rate and a good prognosis [3]. The 5-year survival rate for ChRCC is as high as 90%, much better than that of clear cell RCC and papillary RCC [4].
However, ChRCC is often difficult to diagnose with conventional imaging; thus, improving preoperative imaging diagnostic techniques may help in diagnosis and the preparation of treatment plans. Previous studies on ChRCC based on computed tomography (CT) have reported inconsistencies in plain scan CT features as well as the degrees and patterns of enhancement. One study showed that the masses were often round, with an intact capsule, and the CT scan presented uniform or low-density, and the masses were rarely accompanied by necrosis, cystic transformation (Bosniak IV classification), calcification, or bleeding. Masses often show mild to moderate uniform enhancement with a platform-like enhancement style, and the degree of enhancement is higher than that of papillary RCC, but lower than that of clear cell RCC [5]. Another study showed that CT scans are of equal or high density, and the central scar is a characteristic sign. Enhancement was mild to moderate or delayed [6]. A third study reported that CT scans show uniform or high density, combined with cystic changes of high and low mixed density [3]. These studies are particularly significant in our clinical practice; however, the number of cases in previous studies was relatively small, and CT characteristics varied greatly, lacking accuracy and generalization capability. Therefore, the imaging features of ChRCC warrant further investigation, particularly because of the recent advancements in imaging technology.
In this study, we retrospectively analyzed multi-detector computed tomography (MDCT) plain scans and multi-phase CT enhancement manifestations of ChRCC, including a larger number of cases, as compared with previous studies, that were confirmed by pathological examination to evaluate the diagnostic value of CT for ChRCC. We also aimed to increase the knowledge base of ChRCC imaging and the accuracy of the preoperative diagnosis.
Material and Methods
PATIENTS:
We collected the clinical, imaging, pathological, and follow-up data of all patients with pathologically confirmed ChRCC treated by surgical resection in our hospital between December 2009 and September 2019. The inclusion criteria were: (a) ChRCC was confirmed by postoperative pathology findings; and (b) preoperative MDCT plain scan and multi-phase enhanced CT were performed. Patients who did not meet both criteria or were unwilling to participate in this study were excluded.
This study was approved by the Ethics Committee of Fujian Provincial Hospital (K2016-10-030). All methods were carried out in accordance with relevant guidelines and regulations. Informed consent was obtained from the patients or their relatives.
IMAGING METHODS:
The multi-layer spiral CT scanning devices used were manufactured by Siemens (Erlangen, Germany). The parameters for the Sensation 64 CT were as follows: 64×0.6 mm; 3 mm layer thickness with layer spacing; 120 kV, 150 to 200 mA; 0.75 mm reconstruction layer thickness, and 0.5 mm layer spacing. The corresponding parameters for the Definition Dual-Source CT were 128×0.6 mm, 3 mm, 120 kV, 200 to 290 mA, 0.75 mm, and 0.5 mm, respectively, while those for the Somatom Force Dual-Source CT were 256×0.6 mm, 3 mm, 120 kV, 150 to 300 mA, 0.75 mm, and 0.5 mm, respectively. The procedure was performed using a standard reconstruction algorithm with breath-holding. The scan area was from the hilum level to 2 cm below the lower pole of the kidney. The enhanced CT scan was conducted after the plain CT scan. Iodine contrast was injected through the elbow vein using a double-barrelled high-pressure syringe (iodophor concentration of 320 mg/mL at a dose of 1.5 mL/kg of body weight and an injection flow rate of 3 to 4 mL/s). CT scans were performed for the renal cortex, parenchyma, and pelvis (3 phases) at 25 to 30 s, 50 to 70 s, and 300 s, respectively, after injection. The reconstructed data were transmitted to a workstation for post-processing and analysis of the coronal and sagittal sections, as well as for multiplanar reformation.
IMAGE ANALYSIS:
Using the double-blind principle, the CT images of all patients were reviewed by 2 radiologists who had more than 5 years of experience in imaging diagnosis of urological tumors. They primarily observed the basic CT characteristics of the masses including number, location, shape, size, borders, and scan density, and internal structures, such as necrosis, cystic changes, hemorrhage, calcification, uniformity, false capsule, peripheral invasion or metastasis, and lymph node enlargement. The radiologists also noted the CT enhancement features, methods, and peak phases. Also, they measured the CT values for the solid part of each mass and those of the renal cortex, renal parenchyma, aorta, and inferior vena cava on the plain scan, cortical phase, nephrographic phase, and renal pelvis phase scans. All CT features were compared with the pathological results. Disagreements between the 2 doctors were resolved by consensus. The tumors were divided into 3 groups according to the 2010 American Joint Committee on Cancer guidelines for renal cancer tumor, node, metastasis staging. Group A consisted of stage T1a tumors with a maximum diameter ≤4 cm, group B consisted of stage T1b tumors with a maximum diameter of 4 to 7 cm, and group C consisted of stage T2 tumors with a maximum diameter >7 cm. The region of interest (ROI) was defined as the area containing a substantial part of the mass, with most of the enhancement and uniform density. The selection range for the ROI was about 50% of the mass, including most of the highest density and lowest density areas [3]. We selected a small ROI area (range 8–25 mm2), measured the area 3 times at the same level, and calculated the average value. The size, shape, and position of the ROI measured for each patient on the plain and enhanced CT images were consistent. The ROI selected for masses with uneven density was in the solid part of the mass, and care was taken to avoid necrosis, cystic changes, and calcifications. To exclude the effects of factors such as differences in the rate of contrast agent passing through the body and individual renal function, we calculated the absolute enhancement values of the solid areas during the cortical phase (Z=CT cortical phase – CT plain scan), nephrographic phase (Y=CT nephrographic phase – CT plain scan), and pelvic phase (T=CT pelvic phase – CT plain scan). We also recorded the relative enhancement ratios for each phase (cortical phase, Za1=Z/CT plain scan; nephrographic phase, Yv1=Y/CT plain scan; and pelvic phase, Tp1=T/CT plain scan). We calculated the ratios of the CT values for the solid part of the mass to the CT values for the renal cortex, aorta, and inferior vena cava. These ratios were designated L01, L02, and L03 for the CT plain scan; La1, La2, and La3 for the cortical phase; Lv1, Lv2, and Lv3 for the nephrographic phase; and Lp1, Lp2, and Lp3 for the pelvic phase. The absolute degree of enhancement was divided into the following subgroups: (a) severe enhancement was defined when the enhanced CT value for the solid part of the mass increased by more than 40 Hounsfield units (HU) compared to the plain scan value; (b) moderate enhancement was defined as a CT value increase of 21 to 39 HU after enhancement; (c) mild enhancement was defined as a CT value increase of 6 to 20 HU after enhancement; (d) non-enhancement was defined as a CT value increase of less than 6 HU after enhancement. The degree of relative enhancement was defined as follows: (i) severe enhancement, indicating that the ratio of the difference between the enhanced CT value for the solid mass and the plain scan to the value of the plain CT was greater than 1; (ii) moderate enhancement, indicating that the ratio was 0.5 to 1; (iii) mild or non-enhancement, indicating that the ratio was less than 0.5. Finally, the density of the solid components of the mass as a percentage of the density of the renal cortex was also used to determine the degree of enhancement of the mass. Enhancement of the mass was divided into 4 types: type A, severe enhancement when La1 was more than 80%; type B, moderate enhancement when La1 was 60% to 80%; type C, mild enhancement when La1 was less than 60%; and type D, non-reinforcement when the CT value increased by less than 10 HU after enhancement. There were 4 types of mass enhancement: type I, outflow with a rapid rise and rapid decline, a gradual decrease in La1, peak enhancement during the cortical phase, and a decrease in the renal pelvis phase by more than 10% of the value during the arterial phase; type II, outflow with a slow rise and slow decline, an initial increase in La1 followed by a gradual decrease, peak enhancement at the cortex-medulla junction or nephrographic phase, and a decrease in the renal pelvis phase of more than 10% of the value during the arterial phase; type III, continuous enhancement, referring to a gradual increase in La1 and an increase in the renal pelvis phase of more than 10% of the value during the arterial phase; and type IV, enhancement presenting as a platform, with a nominal change of less than 10% in the ratio of the 3 phases. We judged the degree of tumor invasion as follows: perirenal invasion was indicated by a thickening in the prerenal fascia or masses with soft tissue density in the perirenal space; invasion of the renal capsule was indicated by an unclear boundary between the lifted renal capsule and the mass; invasion by a blood vessel was indicated by a non-smooth wall in the renal vein or inferior vena cava or by a filling defect in the lumen; and lymph node involvement was suggested by an oval lymph node with a short diameter greater than 1 cm or a round lymph node with a diameter greater than 0.8 cm. The basic CT characteristics for groups A, B, and C, with different sized tumors, were compared to determine the absolute enhancement threshold range and the relative enhancement ratio range of the solid parts of the masses in the 3 phases. The peak phase of enhancement, most common type of mass enhancement, and ratio ranges of the CT values for the solid part of the mass to that of the renal cortex, aorta, and inferior vena cava were also determined during each phase.
STATISTICAL ANALYSIS:
All data were analyzed using SPSS 25.0 software (SPSS Inc., Chicago, IL, USA). The Mann-Whitney
Results
DEMOGRAPHIC DATA:
Sixty-seven patients with 68 lesions were included in the study (36 men and 31 women). The average patient age was 52 years (range, 31–77 years). The MDCT examination was completed 3 to 42 days before surgery. The diameter of the masses ranged from 1.4 to 16.6 cm. Twenty-six masses were located in the left kidney and 42 were in the right kidney. Low back pain and/or hematuria were present in 15 patients, whereas 52 patients were asymptomatic. The average length of follow-up was 39 months (range, 2–119 months) and no recurrence or distant metastases were observed.
The main body of the tumors in 7 cases was located in the renal sinus area (1 of the cases was less than 4 cm in maximum diameter), all of which were irregular in shape, and the adjacent renal pelvis and renal calyx were compressed and deformed. The blood vessels in the renal hilum area were compressed and displaced. The remaining masses were located in the contour of the kidney or outside (the largest diameter of a mass was less than 4 cm). Only 5 masses were observed adjacent to the renal calyx and caused compression deformation of the renal pelvis. Tumors greater than 4 cm in diameter had a significantly compressed adjacent renal calyx, renal pelvis, and renal hilar vessels, compared with tumors with the largest diameter of less than 4 cm (
BASIC CT FEATURES OF THE MASSES AND SIGNS OF EXTRAVASATION:
About half of the tumors were less than 4 cm in size and classified as group A; there were fewer tumors in groups B and C (Table 1). Among the 68 tumors, 39 had more uniform or slightly higher density, while 29/68 had equal and lower or higher mixed density (Table 2). Significant differences in the proportions of mass calcification were observed among the 3 groups (group A [0%, 0/32] vs group B [27.3%, 6/22] vs group C [35.70%, 5/14], P=0.001) as well in the proportions of mass central scars (group A [6.3%, 2/32] vs group B [22.7%, 5/22] vs group C [50%, 7/14], P=0.003). Small cortical phase vascular signs were significantly different between the groups (group A [9.4%, 3/32] vs group B [63.6%, 14/22] vs group C [92.9%, 13/14], P=0.000). No differences in tumor location, shape, border, uniform density, cystic degeneration, bleeding, pseudocapsule, peripheral invasion, metastasis, or lymphadenopathy were observed among the 3 groups (all P>0.01).
MDCT TUMOR ENHANCEMENT FEATURES:
The CT values for most masses increased by more than 40 HU after enhancement, compared with the values on plain CT scans (Table 3). The CT value after enhancement was more than double that of the plain CT scan for most masses. Using the absolute enhancement values for the solid parts of the masses, 72.06% (49/68) of the masses had severe enhancement, 26.47% (18/68) had moderate enhancement, and 1.47% (1/68) had no enhancement (Table 4). Using the relative enhancement ratio for the solid part of the mass, 77.94% (53/68) of the masses had severe enhancement, 20.59% (14/68) had moderate enhancement, and 1.47% (1/68) had mild or no enhancement. The degree of enhancement was also classified based on the density of the solid part of the mass divided by the density of the renal cortex. Using this classification, 19.12% (13/68) of the masses had severe enhancement, while 33.82% (23/68) had moderate enhancement, 45.59 (31/68) had mild enhancement, and 1.47% (1/68) had no enhancement (Table 4). Most of the solid masses had weaker CT enhancement than did the neighboring renal cortex. Among the above 3 classifications, severely enhanced tumors in the solid part were significantly different from those with partial enhancement (all P<0.01). No significant difference was observed between tumors with moderate or mild enhancement (both P>0.01). Significant differences were observed among La1, Lv1, and Lp1 (H=14.894, P<0.01); La2, Lv2, and Lp2 (H=115.953, P<0.01) and La3, Lv3, and Lp3 (H=74.043, P<0.01) (Table 5).
The proportions of the 4 enhancement types for the solid area of the mass were as follows: 33.82% for type I (23/68), 61.76% for type II (42/68), 2.94% for type III (2/68), and 1.47% for type IV (1/68) (Table 6, Figure 1). The most common type of mass enhancement was type II, followed by type I, and significant differences were observed between these 2 groups (P=0.001). Significant differences between types I and II were found only between the relative and absolute enhancement levels during the cortical phase (P=0.001). No differences in the relative or absolute enhancement levels were observed during the nephrographic or delayed phases (all P>0.001).
Discussion
Western scholars have reported that ChRCC is most common in adult men over 50 years of age and often has no clinical symptoms [7]. Similarly, a report that included 166 cases from mainland China found that 53% (88/166) of cases were in male patients [8]. Some patients may present with low back pain, waist discomfort, or gross hematuria. The clinical onset stage is mostly early, recurrence and metastasis are rare, and the prognosis is generally good [2, 8]. However, as a malignant tumor, ChRCC has the ability to invade and metastasize; the incidence of recurrence or metastasis was 6.2% in a study that included 209 cases [9]. In our study, 54% (36/67) of the patients were men, and 78% (52/67) of patients had no obvious clinical symptoms. Only 1 case had developed lymph node metastasis at the first interview. Sixty-six patients had no signs of recurrence or metastasis during follow-up (range, 2–119 months). These findings are consistent with previous reports. In the present study, 15 patients had symptoms of low back pain or hematuria, and the symptoms had no relationship to the size of the mass. Treatments, such as radical or partial nephrectomy, as well as a ChRCC follow-up, are administered according to tumor stage; in particular, the surgical approach is based on tumor size and location [10]. In our present study, we divided the cases into 3 groups according to tumor size, and the analysis was performed according to tumor location; therefore, our results are a helpful guide for surgeons to choose the appropriate treatment plan.
The CT features of ChRCC are as follows. A single round-shaped mass with a clear boundary appears in the renal parenchyma on a plain CT scan; the main body is mostly located inside and outside the contour of the kidney; the plain scan is more uniform or higher in density, necrotic cystic degeneration, and calcification (incidence of 14% to 19%); and cases of bleeding are rare [3]. The enhancement of the tumor during the cortical phase is lower than that of the renal cortex but higher than that of the renal medulla. The enhancement of the tumor during the parenchymal and delayed phases is lower than that of the renal parenchyma [11]. Tumor enhancement is lower in renal eosinophilic adenoma and clear cell RCC [12,13]. Some of the masses may have delayed strengthening of the star-shaped central scar [6]. Invasion of the tumor into the renal capsule is common, while invasion of the renal vein or inferior vena cava, lymph nodes, or distant metastasis is rare [13]. However, we found that necrotic cysts are not uncommon in cases of ChRCC (29/68, about 42%), which was similar (48.57%) to the findings of Siva et al [3]. We also found that necrotic cysts had no relationship with the size of the mass, even though some masses were mainly cystic (3/29, and 1 with a maximum diameter of less than 4 cm). This may be because the cystic tumor has fewer solid structures and incomplete separated slender blood vessels, and the tumor cells are in cysts along the fibrous blood vessel. Some cysts may be related to insufficient blood supply because of their larger size. In addition, we discovered that calcification of the mass, the central scar, and small blood vessel signs during the cortical phase were related to the size of the mass. That is, the larger the mass, the more likely that it was calcified, had a central scar, and small blood vessel signs would appear. A larger mass has an insufficient blood supply, which can cause post-necrosis calcification and fibrosis and increase the number and thickness of blood vessels to meet tumor growth. The contrast agent takes a longer time to enter and exit thick-walled small blood vessels and it does not leak out, which allows the blood vessel sign to appear [6].
The tumors in most of our patients were located in the renal sinus area, which was often irregularly shaped; and the larger the mass, the easier it was to squeeze the adjacent renal pelvis, renal calyx, and renal hilar blood vessels. MDCT enhancement clearly showed the location of the tumor and its relationship with its surroundings, which was helpful to plan an appropriate surgical approach. The CT value of most of the solid parts of the tumors increased by more than 40 HU compared with the plain scan, and the CT values of some tumors increased by more than 100 HU during the cortical phase. In addition, the enhancement thresholds of the cortical phase, parenchymal phase, and delayed phase of the tumors were 85 HU, 90 HU, and 70 HU, respectively. The degree of enhancement of the cortical mass was lower than that of the renal cortex but higher than that of the renal medulla. The degree of enhancement of the mass in the parenchyma during the delayed phase was lower than that of the renal parenchyma. These findings were similar to those of previous studies [11,13]. Furthermore, we used 3 different methods to assess the degree of enhancement of the masses. The ratio of the lesion to the renal cortex appeared to be most effective, with an average ratio of 0.59. This method avoids the effect of individual differences in renal blood supply and operator measurement errors. The masses had 4 types of enhancement. Peak mass enhancement was most commonly observed for outflows with a slow rise and a slow flow during the skin-medullary junction or nephrographic phase. These masses were continuously enhanced, but the degree of dual-phase enhancement was lower than that of the renal cortex, which is clearly different from the rapid increase and decrease observed for clear cell RCC [14,15]. This observation might be related to the slow blood velocity and the slow contrast agent extravasation, as tumors contain mostly thick-walled small blood vessels [16]. About 30% of the masses presented rapid ascending and descending enhancement, with enhanced peaks during the cortical period, but the degree of enhancement was significantly lower than that of clear cell RCC and eosinophilic adenoma [6]. This may be due to fewer thick-walled blood vessels in the solid structure, where the thin blood vessels are not completely separated. However, we found very few MDCT images that showed features indicative of atypical ChRCC, and it was difficult to distinguish from typical clear cell RCC.
ChRCC is usually easy to distinguish from typical clear cell RCC and papillary RCC. The degree of enhancement of ChRCC is higher than that of inflow-enhanced papillary RCC [17] but lower than that of outflow-enhanced clear cell RCC [13,18]. ChRCC is often difficult to differentiate from renal oncocytoma (RO) and low-fat angiomyolipoma (lfAML). RO often occurs in the renal cortex and is mostly homogeneous or slightly less dense on a plain scan; necrosis, cystic transformation and calcification are rare; and the degree of enhancement of cortical tumors is significantly higher than that of ChRCC [12]. Some cases of RO may have characteristic enhancement reversal signs; others may have delayed enhancement of central scars, which are difficult to distinguish from eosinophilic ChRCC, but immunohistochemistry can be used to distinguish RO from ChRCC, for instance, where there is positive S100A1 in RO but negative S100A1 in ChRCC [12]. lfAML commonly occurs in women, is usually equally or slightly denser than ChRCC on a plain scan, and necrosis, hemorrhage, and calcification are rare. No central scar or pseudocapsule is observed inside the mass when the moderately or significantly enhanced mass is small [19]. We found that the degree of enhancement of lfAML was slightly higher than that of ChRCC, and spot-shaped blood vessels are common in lfAML. Magnetic resonance imaging can be performed when identification is difficult. lfAML has mostly equal or low signal intensity on T2WI images, whereas ChRCC has slightly high signal intensity.
This study had some limitations. As a single-arm retrospective study, we did not include other types of renal tumors for comparative and prospective analyses, such as clear cell RCC and papillary RCC. Furthermore, it was a single-center study and therefore the imaging features we used may not be fully recognized by other centers. Other important features were not found or analyzed in this study. However, this study had a larger sample size, compared with a previous study, and it may help physicians make a preoperative diagnosis with more confidence. In the future, multi-center, prospective, case-controlled studies are needed to further explain the imaging characteristics of ChRCC and allow doctors to make more accurate preoperative decisions.
Conclusions
MDCT helped us diagnose ChRCC before surgery. The MDCT plain scan and enhanced performance features of ChRCC are diverse. The tumors were homogeneous or slightly dense with a round-like shape, clear boundaries, and complete capsules; the degree of tumor enhancement during the different phases seemed lower than those of surrounding tissues; calcification, necrosis, and cystic changes were not uncommon; and central scars and small cortical signs in the homogeneous mass were also helpful for making diagnoses. In short, it is necessary to synthesize the multiple MDCT features of a mass to improve accuracy of the preoperative diagnosis.
Tables
Table 1. Clinical characteristics of the 3 groups of patients with masses of different sizes.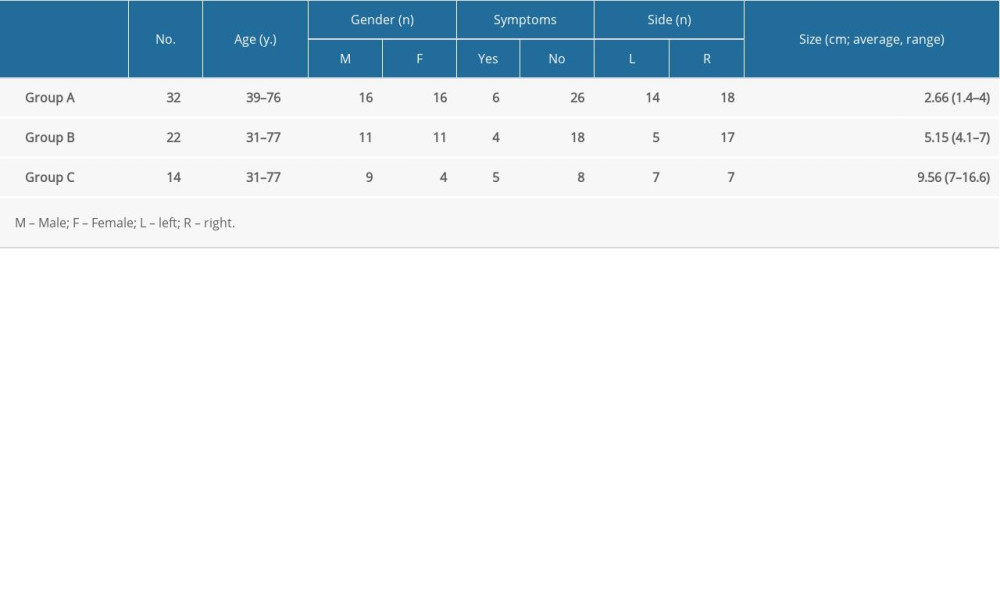 Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes.
Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes.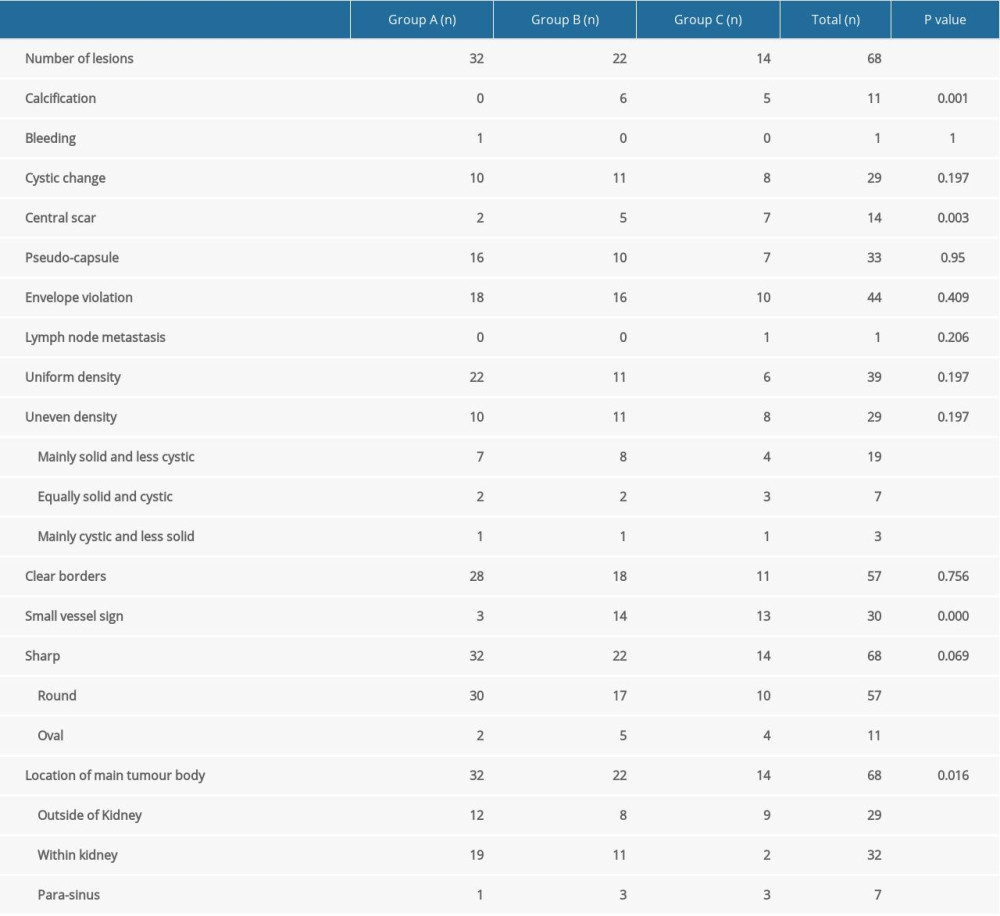 Table 3. Computed tomography value for the mass in each stage.
Table 3. Computed tomography value for the mass in each stage.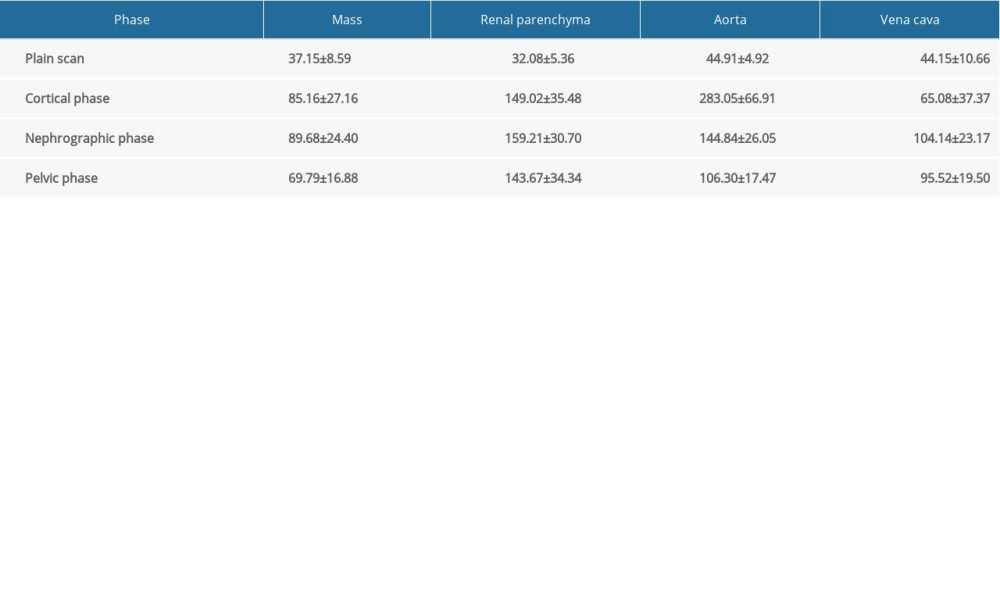 Table 4. The number and degree of enhancement of the solid areas of the 68 masses.
Table 4. The number and degree of enhancement of the solid areas of the 68 masses.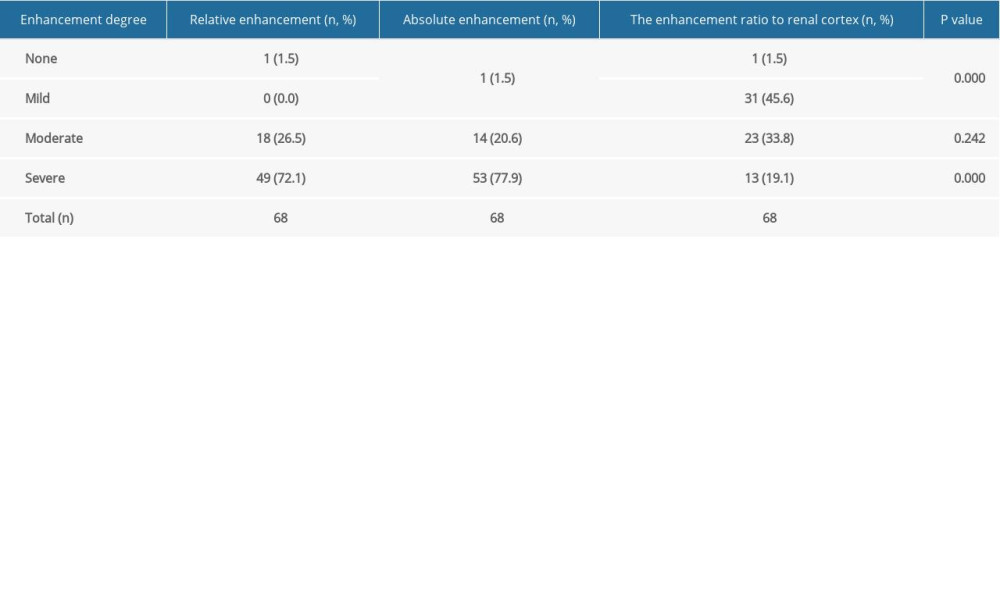 Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios.
Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios.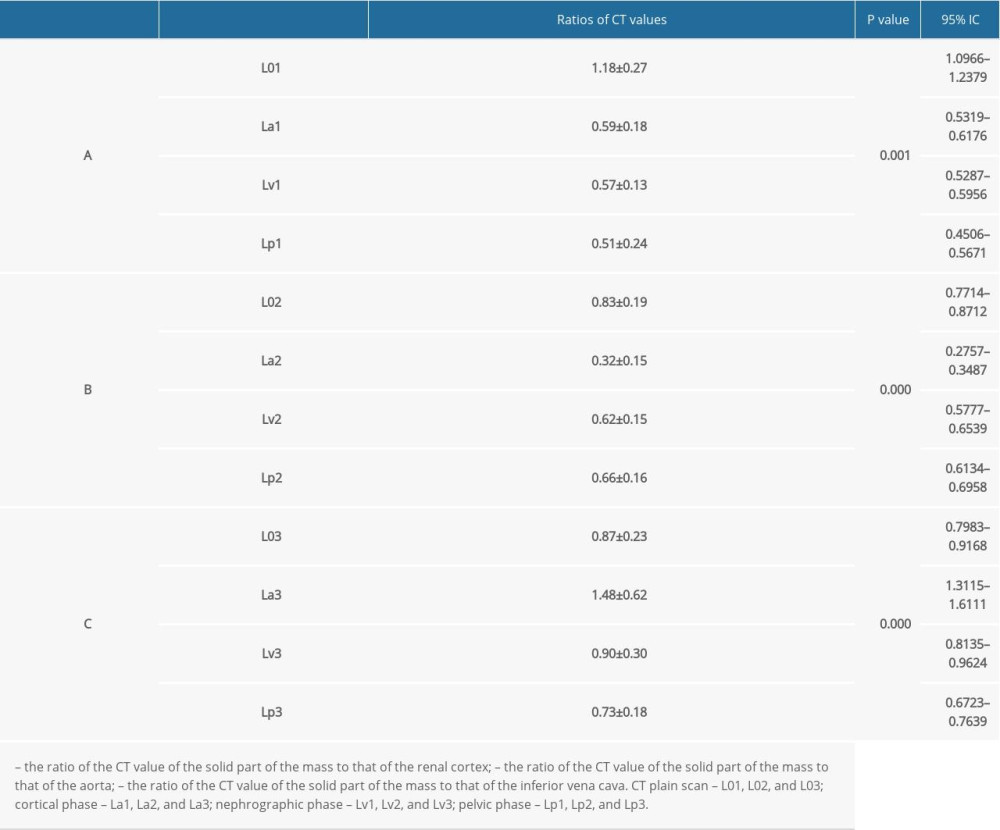 Table 6. Absolute and relative enhancement of 65 masses.
Table 6. Absolute and relative enhancement of 65 masses.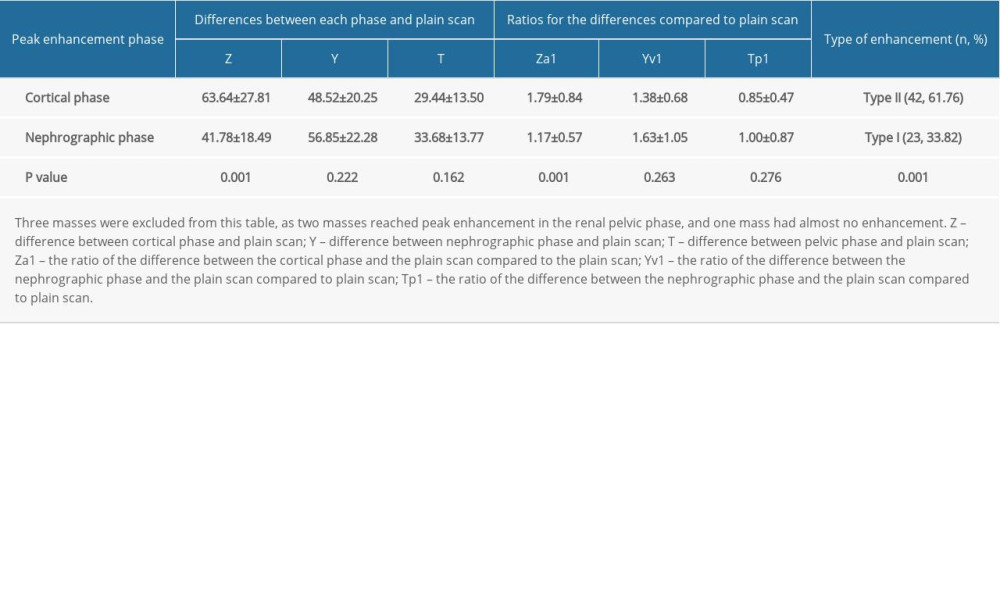
References
1. Moch H, Cubilla AL, Humphrey PA, The 2016 WHO Classification of tumors of the urinary system and male genital organs – part A: Renal, penile, and testicular tumors: Eur Urol, 2016; 70; 93-105
2. May M, Zigeuner R, Aziz APostoperative prognosis of chromophobic renal cell carcinoma: Comparative analysis of the multinational CORONA database: Urologe A, 2014; 53; 228-35 [in German]
3. Raman SP, Johnson PT, Allaf ME, Chromophobe renal cell carcinoma: multiphase MDCT enhancement patterns and morphologic features: Am J Roentgenol, 2013; 201; 1268-76
4. Amin MB, Paner GP, Alvarado-Cabrero I, Chromophobe renal cell carcinoma: Histomorphologic characteristics and evaluation of conventional pathologic prognostic parameters in 145 cases: Am J Surg Pathol, 2008; 32; 1822-34
5. Wu J, Zhu Q, Zhu W, Comparative study of CT appearances in renal oncocytoma and chromophobe renal cell carcinoma: Acta Radiol, 2016; 57; 500-6
6. Galmiche C, Bernhard JC, Yacoub M, Is multiparametric MRI useful for differentiating oncocytomas from chromophobe renal cell carcinomas?: Am J Roentgenol, 2017; 208; 343-50
7. Moch H, Artibani W, Delahunt B, Reassessing the current UICC/AJCC TNM staging for renal cell carcinoma: Eur Urol, 2009; 56; 636-43
8. Jiang HM, Wei JH, Zhang ZL, Does chromophobe renal cell carcinoma have better survival than clear cell renal cell carcinoma? A clinical-based cohort study and meta-analysis: Int Urol Nephrol, 2016; 48; 191-99
9. Xie Y, Ma X, Li H, Prognostic value of clinical and pathological features in chinese patients with chromophobe renal cell carcinoma: A 10-year single-center study: J Cancer, 2017; 8; 3474-79
10. Motzer RJ, Jonasch E, Agarwal N, Kidney cancer, Version 2.2017, NCCN clinical practice guidelines in oncology: J Natl Compr Canc Netw, 2017; 15; 804-34
11. Kopp RP, Aganovic L, Palazzi KL, Differentiation of clear from non-clear cell renal cell carcinoma using CT washout formula: Can J Urol, 2013; 20; 6790-97
12. Rosenkrantz AB, Hindman N, Fitzgerald EF, MRI features of renal oncocytoma and chromophobe renal cell carcinoma: Am J Roentgenol, 2010; 195; W421-27
13. Young JR, Margolis D, Sauk S, Clear cell renal cell carcinoma: Discrimination from other renal cell carcinoma subtypes and oncocytoma at multiphasic multidetector CT: Radiology, 2013; 267; 444-53
14. Kim JK, Kim TK, Ahn HJ, Differentiation of subtypes of renal cell carcinoma on helical CT scans: Am J Roentgenol, 2002; 178; 1499-506
15. Alshumrani G, O’Malley M, Ghai S, Small (≤4 cm) cortical renal tumors: Characterization with multidetector CT: Abdom Imaging, 2010; 35; 488-93
16. Sun MR, Ngo L, Genega EM, Renal cell carcinoma: Dynamic contrast-enhanced MR imaging for differentiation of tumor subtypes – correlation with pathologic findings: Radiology, 2009; 250; 793-802
17. Young JR, Coy H, Douek M, Type 1 papillary renal cell carcinoma: Differentiation from Type 2 papillary RCC on multiphasic MDCT: Abdom Radiol (NY), 2017; 42; 1911-18
18. Lee-Felker SA, Felker ER, Tan N, Qualitative and quantitative MDCT features for differentiating clear cell renal cell carcinoma from other solid renal cortical masses: Am J Roentgenol, 2014; 203; W516-24
19. Sung CK, Kim SH, Woo S, Angiomyolipoma with minimal fat: Differentiation of morphological and enhancement features from renal cell carcinoma at CT imaging: Acta Radiol, 2016; 57; 1114-22
Tables
 Table 1. Clinical characteristics of the 3 groups of patients with masses of different sizes.
Table 1. Clinical characteristics of the 3 groups of patients with masses of different sizes. Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes.
Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes. Table 3. Computed tomography value for the mass in each stage.
Table 3. Computed tomography value for the mass in each stage. Table 4. The number and degree of enhancement of the solid areas of the 68 masses.
Table 4. The number and degree of enhancement of the solid areas of the 68 masses. Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios.
Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios. Table 6. Absolute and relative enhancement of 65 masses.
Table 6. Absolute and relative enhancement of 65 masses. Table 1. Clinical characteristics of the 3 groups of patients with masses of different sizes.
Table 1. Clinical characteristics of the 3 groups of patients with masses of different sizes. Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes.
Table 2. Multiple detector computed tomography plain scan and enhancement features among the 3 groups with different tumor sizes. Table 3. Computed tomography value for the mass in each stage.
Table 3. Computed tomography value for the mass in each stage. Table 4. The number and degree of enhancement of the solid areas of the 68 masses.
Table 4. The number and degree of enhancement of the solid areas of the 68 masses. Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios.
Table 5. Tumor plain scan and multi-phase enhanced CT values and their corresponding ratios. Table 6. Absolute and relative enhancement of 65 masses.
Table 6. Absolute and relative enhancement of 65 masses. In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952