08 February 2021: Clinical Research
No COVID-19 Cases Detected Between April and September 2020 After Screening All 838 Admissions to a Maternity Unit in Poland
Paweł Piekarski1ABCDEF*, Małgorzata Sateja1BCF, Tomasz Maciejewski1DE, Tadeusz Issat1DEDOI: 10.12659/MSM.929123
Med Sci Monit 2021; 27:e929123
Abstract
BACKGROUND: Between April and September 2020, there were <10 000 reported cases of COVID-19 in the Masovia district, Poland, and <1000 new cases daily in Poland. During this period, all new hospital admissions to a maternity unit of a teaching hospital in Warsaw were screened for the COVID-19 infection. This retrospective study presents the findings from the reverse transcription-polymerase chain reaction (RT-PCR) test for COVID-19.
MATERIAL AND METHODS: This study included 838 women admitted for delivery between April 20 and September 20, 2020. All the admitted women were assigned to a low-risk or a high-risk group for COVID-19 and underwent RT-PCR nasopharyngeal swab tests (GeneFinder™-COVID-19-Plus-RealAmpKit. OSANG Healthcare Co., Ltd., Gyeonggi-do, Korea) for COVID-19. The testing protocol included repeated testing in case of inconclusive results or negative results in the symptomatic patients. The maternal and neonatal data from these cases were collected and analyzed.
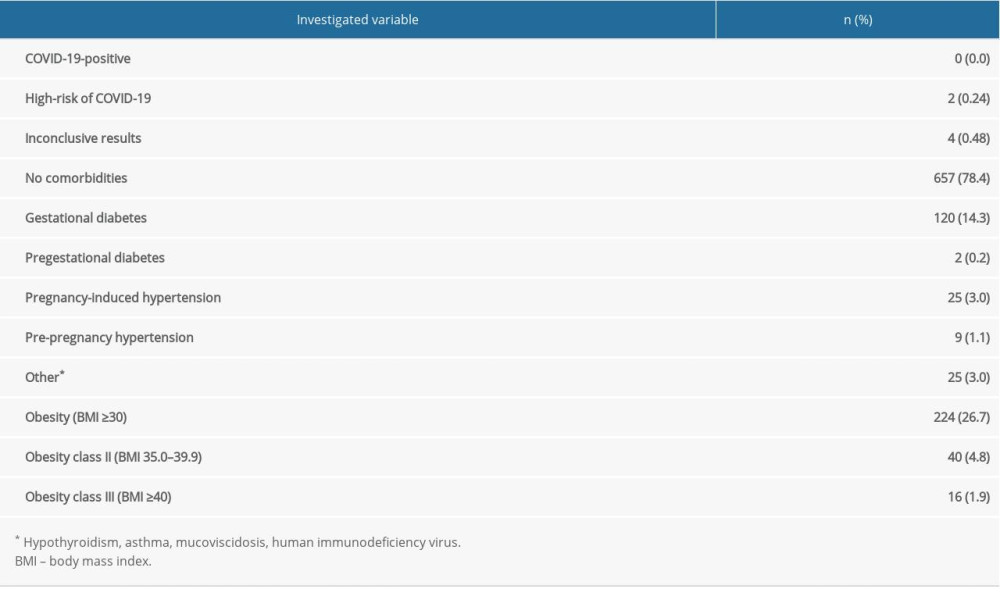
RESULTS: All of the 838 women tested negative for COVID-19. Two women (0.24%) were classified as high risk for COVID-19. For 4 (0.48%) women, the results were initially inconclusive and negative when repeated. One hundred and eighty-one (21.5%) women presented with comorbidities, and 60 (7.2%) women were ≥40 years old.
CONCLUSIONS: The findings from this study show that between April and September 2020, there were no cases of COVID-19 infections at the maternity unit of a teaching hospital in Warsaw, Poland. However, the infection rates for COVID-19 across Europe continue to change. Testing protocols have been developed and established for all hospital admissions and it is anticipated that testing methods will become more rapid and accurate.
Keywords: COVID-19, Delivery, Obstetric, Pregnancy, SARS Virus, COVID-19, COVID-19 Testing, Hospitalization, Hospitals, Maternity, Infant, Newborn, Mass Screening, Poland, SARS-CoV-2
Background
As the spread of the coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has reached pandemic status, the development of effective screening strategies, including those for pregnant women, have challenged health systems globally. A large proportion of COVID-19-positive pregnant women are asymptomatic [1–3]. Most pregnant women with COVID-19 do not have serious morbidities and the most common complication is preterm delivery [4–6]. However, the status of pregnant women can progress rapidly from being asymptomatic to having a severe COVID-19 infection during the course of the delivery [7], and several studies show maternal mortality related to COVID-19 [8–10].
COVID-19 can be transmitted by both symptomatic and asymptomatic patients [11,12]. The identification of the virus carriers is an important part of the strategy to limit the pandemic, and justifies the universal screening of pregnant women admitted to hospitals for delivery. The aim of universal screening for COVID-19 in pregnant women admitted to the hospital is to prevent or at least reduce the transmission of the virus to the other patients, neonates, and medical staff [1,2,13,14]. The pregnant women diagnosed with COVID-19 can be isolated and treated according to the recommended medical protocols [13,15,16], and safety measures can be implemented, including the use of the recommended protective equipment [17]. The universal screening of pregnant women reflects the epidemic status of the general population [1]. Routine screening with nasopharyngeal swabs is mostly accepted; however, it can be declined by some patients, mainly due to the discomfort experienced during sampling [18].
Globally, the results of universal screening for COVID-19 in pregnant women hospitalized in labor have shown the incidence of patients who tested positive for COVID-19 to be between <1% to 19.8%, with the highest rates reported in New York City (USA) in March and April 2020 [1,2,19–23]. European studies revealed the COVID-19-positive cases in women admitted for delivery to be ≤1% in Italy and Spain, 3.9% in London (United Kingdom), and 11.7% in Portugal [3,19–22]. In the study from New York City, 87.9% of cases were asymptomatic [2]. A recent study showed that the active questioning of patients can lower the rate of asymptomatic patients to 43.2% [1]. The recommendations by the World Association of Perinatal Medicine for the clinical management of COVID-19 include universal screening for COVID-19 in women admitted for delivery, especially in the high-prevalence areas [13]. This approach is confirmed by other authors, particularly in consideration of the significant percentage of asymptomatic patients who test positive for COVID-19 [1,14,16]. However, there are no clear recommendations about universal screening for the low-prevalence COVID-19 populations. Therefore, before deciding to implement universal testing, an estimation of the local needs-resources balance is advised [13–15].
The detection of COVID-19 ribonucleic acid on the nasopharyngeal swabs or other respiratory-tract specimens using the reverse transcription-polymerase chain reaction (RT-PCR) test, with an estimated sensitivity of 89% and specificity of 99%, is the criterion standard for the direct diagnosis of an active COVID-19 infection [12,24]. The optimal timing of an RT-PCR COVID-19 test with nasopharyngeal swabs begins 2 days after the infection and lasts until negativization, with the positivity peak at 7 days to 10 days after the onset of symptoms [12]. However, there are limitations of the RT-PCR COVID-19 test related to the false-positive and false-negative results [24]. The false-negative results of RT-PCR COVID-19 testing can be caused by low amounts of the sample, improper conservation during storage or transport, and the presence of inhibitors or virus mutations in the investigated territories [12]. Reports can show symptomatic patients with negative baseline results for the RT-PCR COVID-19 tests and subsequent positive results [25,26]. A study showed 15/70 (21.4%) patients from the general population with moderate or mild symptoms of COVID-19 required 3 tests before getting a positive result, and for 1 (1.4%) patient, 45 days passed after the onset of the symptoms before the test showed a positive RT-PCR result [25]. Other disadvantages of the RT-PCR COVID-19 test include its cost, the need for infrastructure and qualified staff, and the risk of incorrect sampling and transportation [12,24]. Nevertheless, due to its high sensitivity and specificity, the RT-PCR COVID-19 test remains the criterion standard for the direct diagnosis of symptomatic and asymptomatic patients.
To have a confirmed positive PCR test for the SARS virus, the World Health Organization (WHO) requires 2 samples collected from an individual at 1 time, or samples collected from the individual 2 or more times on 2 or more days, or different assays run on the same sample, or repeated PCR testing on the same sample [27]. Repeated testing is recommended in case of a negative result for the RT-PCR COVID-19 test in patients who meet the clinical criteria for COVID-19, including serum collection for antibody detection with a serological assay in the acute phase, and 2–4 weeks later in case of a persistent negative RT-PCR COVID-19 result [24]. According to the International Society of Infectious Disease in Obstetrics and Gynecology (ISIDOG), pregnant women are considered to be at high risk for COVID-19 and a low threshold for RT-PCR testing is advised [16]. ISIDOG recommends a repeat of the test in 24 hours in case of a negative result in patients suspicious for COVID-19 [16].
Between April and September 2020, there were <10 000 reported cases of COVID-19 in Warsaw, Masovia District, Poland, and <1000 new cases daily in Poland. During this time, all new hospital admissions were screened for COVID-19. This retrospective study presents the findings from the RT-PCR testing for COVID-19 for all pregnant women admitted to the maternity unit of a teaching hospital in Warsaw, Poland between April and September 2020.
Material and Methods
STATISTICAL ANALYSIS:
A descriptive statistical analysis was performed, using absolute frequencies (percentages) and means (standard deviations). As no positive cases were found in the study, a comparative analysis could not be performed. The calculations were performed using Excel 2011 for Mac, Version 14.7.3 (Microsoft Corporation, Redmond, USA).
This study was evaluated and approved by the Ethics Committee of the Institute of Mother and Child, Warsaw, Poland. Written informed consent from the pregnant women was waived by the Ethics Committee for this retrospective analysis.
Results
All 838 women tested during this study showed a negative result on the RT-PCR test (Table 1). At admission, 2 women (0.24%) were evaluated as high risk for COVID-19 and were isolated until the negative results of their tests were available. One of these 2 women considered at high risk was 25 years old and in her third pregnancy (30.2 weeks). She presented with a high temperature (39.4°C) and oligohydramnios. She did not complain of cough or dyspnea. The fetal heart rate was 180 beats per minute. Her C-reactive protein was 184 mg/L. She was suspected to have an intrauterine infection and was administered intravenous ampicillin (2.0 g initially and then 1.0 g every 4 hours). She had a cesarean section on the same day as her hospital admission because of a suspected intrauterine infection. She delivered a male neonate, with a birth weight of 1270 g and a 5-minute Apgar score of 8. He was intubated on the same day for 4 days and required intensive care as he had neonatal respiratory distress without any symptoms of infection. Her temperature returned to normal the next day, and she was discharged on day 4 in good condition. The neonate was discharged after 13 weeks and 4 days of hospitalization.
The second woman considered at high risk for COVID-19 was 36 years old and in her third pregnancy (37.3 weeks). She presented with a cough and a history of contact with a family member who had tested positive for COVID-19. She did not have fever or dyspnea and her C-reactive protein was 13.9 mg/L. She had a vaginal delivery on the day of her admission and gave birth to a healthy male neonate weighing 3210 g with a 5-minute Apgar score of 10. She was discharged in good condition on the same day at her own request. All the other women were non-symptomatic during their hospitalization. For 4 (0.48%) women, the results of the initial RT-PCR tests were inconclusive and were negative when the test was repeated after 48 hours.
The characteristics of all the women are shown in Table 2. There were 60 (7.2%) women aged ≥40 years. Sixty-four (7.6%) women had premature deliveries, 354 (42.2%) women had cesarean sections, and 419 (50.0%) women delivered for the first time. One hundred and eighty-one (21.5%) women had comorbidities; the most common were gestational diabetes 120 (14.3%), followed by pregnancy-induced hypertension 25 (3.0%). Two hundred and twenty-four (26.7%) women were obese, including class II and class III obesity in 40 (4.8%) and 16 (1.9%) women, respectively (Table 1).
Discussion
This is one of the first studies showing the results of the routine RT-PCR test for COVID-19 in pregnant women admitted for delivery at a teaching hospital’s maternity unit in Eastern Europe, at a time of low incidence of the virus in the general population.
None of the 838 women admitted for delivery in this study (April to September 2020) tested positive for COVID-19. Other studies showed the rate of COVID-19-positive pregnant women ranging from <1% to 19.8% [1–3,19–23]. Sutton et al reported that 33/215 (15.4%) women admitted for delivery in New York City were positive for COVID-19 in March and April 2020 [2]. During the 2 weeks of their observation, there were 85 056 new positive COVID-19 cases and 5037 COVID-19-related deaths in the general population in New York City [33]. Other cities in the USA with a lower incidence of COVID-19 in the general population showed much lower rates of COVID-19 in pregnant women [34,35]. In Europe, high rates (11.7%) of positive COVID-19 pregnant women were reported from the most affected regions of Portugal during the national lockdown [3]. The total number of positive COVID-19 cases in Poland until the end of our study was 78 330, and the first day that the number of new positive COVID-19 cases in Poland reached 1000 (1002 cases) was the last day of observation (September 20, 2020) for this study [31]. For most of the present study duration, the rates of positive COVID-19 cases in the general population per 100 000 people in Poland and the Masovia district were below 200, reaching this number on September 16, 2020 and September 18, 2020, respectively. The results of our study correspond with the findings from Slovenia, where they found no positive COVID-19 cases in 202 pregnant women between March and May 2020 [36]. In another European study, Herraiz et al [19] compared the results of universal screening for COVID-19 in women admitted for delivery in Madrid with the other studies from Italy and USA. They concluded that the incidence of positive COVID-19 cases in pregnant women corresponds mostly to the incidence of COVID-19 in the general population. A positive correlation was reported between the daily rate of positive COVID-19 cases in a maternity ward and the daily incidence of COVID-19 in the general population of an investigated area [1].
In the present study, the RT-PCR nasopharyngeal swab test for COVID-19 used for routine screening, in compliance with the manufacturer’s instructions [28] and WHO guidelines [29] with its high specificity and sensitivity, was an advantage [37]. The disadvantage was the time (up to 48 hours) required to obtain the results as the samples were sent to an external laboratory used by a number of public and private health facilities in Warsaw. Soon after the screening had started, it became clear that some of the patients could have the results of their tests while they were on the way home. This was particularly true in cases where repeated testing was needed. As the results were ready within 48 hours, patients scheduled for elective cesarean sections or induction of labor were tested in our maternity unit 48 hours before admission, so the results were available at the time of their admission. The acceptance of the results of the testing done within 48 hours before admission was presented in other publications [1]. Our study protocol included repeated testing in 24 hours for symptomatic women with negative results, and repeated testing in 48 hours for asymptomatic women with inconclusive results, as suggested by the ISIDOG and WHO recommendations [16,24]. In this study, we repeated the test for the 4 patients whose initial results were inconclusive and their repeated tests showed negative results. Of the 2 symptomatic women, neither underwent repeated testing. The woman diagnosed with the intrauterine infection had symptoms that resolved quickly after the cesarean section and the first RT-PCR nasopharyngeal swab test had a negative result. Therefore, no repeated testing was performed. The second woman left hospital at her own request soon after a vaginal delivery, on the day of admission.
In this study, none of the subjects refused testing. Kernberg et al tested 223 asymptomatic women 3–5 days before or on admission to the labor unit using the RT-PCR nasopharyngeal swab test for COVID-19 and found that 17% of women declined testing, mostly because of the discomfort during testing [18]. As the screening progressed in our institution, we noticed that patient discomfort during testing was an issue; however, we did not include this parameter in the study protocol. Another reason, reported by Kernberg et al, for refusing testing was the apprehension that the positive results in asymptomatic pregnant women could result in a separation from their babies and/or their families [18].
The results of our study support that the incidence of COVID-19 in pregnant women is related to its incidence in the general population. As the substantial proportion of COVID-19-positive pregnant carriers of the virus are asymptomatic or do not mention any symptoms on admission [1,2,34], routine screening of all women admitted for delivery could be useful in limiting the spread of the virus. This is acknowledged in a number of recommendations [13–16]. However, this approach is beneficial when the number of positive COVID-19 cases in the general population is large enough to be reflected in the population of pregnant women admitted to hospitals. Whether there is a cutoff related to the number of daily new cases of COVID-19 or to the rate of COVID-19 per 100 000 people in the general population needs further research. As the rates of COVID-19 cases change with time and populations [38], a flexible approach to universal testing for the virus would be favorable for both the patients in need and the overloaded healthcare systems.
Conclusions
The findings from this study showed that between April and September 2020, there were no cases of COVID-19 infections at the teaching hospital’s maternity unit in Warsaw, Poland. However, the infection rates for COVID-19 across Europe continue to change. The establishment of universal testing protocols for all hospital admissions have now been developed and it is hoped that testing methods will become more rapid and accurate.
References
1. Díaz-Corvillón P, Mönckeberg M, Barros A, Routine screening for SARS-CoV-2 in unselected pregnant women at delivery: PLoS One, 2020; 15(9); e0239887
2. Sutton D, Fuchs K, D’Alton M, Goffman D, Universal screening for SARS-CoV-2 in women admitted for delivery: N Engl J Med, 2020; 382(22); 2163-64
3. Dória M, Peixinho C, Laranjo M, Covid-19 during pregnancy: A case series from an universally tested population from the north of Portugal: Eur J Obstet Gynecol Reprod Biol, 2020; 250; 261-62
4. Mullins E, Evans D, Viner RM, Coronavirus in pregnancy and delivery: Rapid review: Ultrasound Obstet Gynecol, 2020; 55(5); 586-92
5. Di Mascio D, Khalil A, Saccone G, Outcome of coronavirus spectrum infections (SARS, MERS, COVID-19) during pregnancy: A systematic review and meta-analysis: Am J Obstet Gynecol, 2020; 2(2); 100107
6. Huntley BJF, Huntley ES, Di Mascio D, Rates of maternal and perinatal mortality and vertical transmission in pregnancies complicated by severe acute respiratory syndrome coronavirus 2 (SARS-Co-V-2) infection: A systematic review: Obstet Gynecol, 2020; 136(2); 303-12
7. Breslin N, Baptiste C, Gyamfi-Bannerman C, COVID-19 infection among asymptomatic and symptomatic pregnant women: Two weeks of confirmed presentations to an affiliated pair of New York City hospitals: Am J Obstet Gynecol, 2020; 2(2); 100118
8. Hantoushzadeh S, Shamshirsaz AA, Aleyasin A, Maternal death due to COVID-19: Am J Obstet Gynecol, 2020; 223(1); 109.e1-16
9. Nakamura-Pereira M, Betina Andreucci C, de Oliveira Menezes M, Worldwide maternal deaths due to COVID-19: A brief review: Int J Gynecol Obstet, 2020; 151(1); 148-50
10. Takemoto MLS, Menezes MO, Andreucci CB, Maternal mortality and COVID-19: J Matern Neonatal Med, 2020; 16; 1-7
11. World Health Organization, Transmission of SARS-CoV-2: Implications for infection prevention precautions: Scientific brief, 2020 https://www.who.int/news-room/commentaries/detail/transmission-of-sars-cov-2-implications-for-infection-prevention-precautions
12. Russo A, Minichini C, Starace M, Current status of laboratory diagnosis for COVID-19: A narrative review: Infect Drug Resist, 2020; 13; 2657
13. Api O, Sen C, Debska M, Clinical management of coronavirus disease 2019 (COVID-19) in pregnancy: Recommendations of WAPM-World Association of Perinatal Medicine: J Perinat Med, 2020; 48(9); 857-66
14. Metz TD, Is universal testing for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) needed on all labor and delivery units?: Obstet Gynecol, 2020; 136(2); 227-28
15. López M, Gonce A, Meler E, Coronavirus disease 2019 in pregnancy: A Clinical management protocol and considerations for practice: Fetal Diagn Ther, 2020; 47(7); 519-28
16. Donders F, Lonnée-Hoffmann R, Tsiakalos A, ISIDOG recommendations concerning COVID-19 and pregnancy: Diagnostics, 2020; 10(4); 243
17. World Health Organization: Personal protective equipment for COVID-19, 2020 https://www.who.int/teams/health-product-and-policy-standards/access-to-assistive-technology-medical-devices/priority-medical-devices-for-covid/ppe-covid
18. Kernberg A, Kelly J, Nazeer S, Universal severe acute respiratory syndrome coronavirus 2 (SARS-COV-2) testing uptake in the labor and delivery unit: Obstet Gynecol, 2020; 136(6); 1103-8
19. Herraiz I, Folgueira D, Villalaín C, Universal screening for SARS-CoV-2 before labor admission during Covid-19 pandemic in Madrid: J Perinat Med, 2020; 48(9); 981-84
20. Abeysuriya S, Wasif S, Counihan C, Universal screening for SARS-CoV-2 in pregnant women at term admitted to an East London maternity unit: Eur J Obstet Gynecol Reprod Biol, 2020; 252; 444-46
21. Tanacan A, Erol SA, Turgay B, The rate of SARS-CoV-2 positivity in asymptomatic pregnant women admitted to hospital for delivery: Experience of a pandemic center in Turkey: Eur J Obstet Gynecol Reprod Biol, 2020; 253; 31-34
22. Gagliardi L, Danieli R, Suriano G, Universal SARS-CoV-2 testing of pregnant women admitted for delivery in two Italian regions: Am J Obstet Gynecol, 2020; 223(2); 291-92
23. Vintzileos WS, Muscat J, Hoffmann E, Screening all pregnant women admitted to labor and delivery for the virus responsible for coronavirus disease 2019: Am J Obstet Gynecol, 2020; 223(2); 284-86
24. World Health Organization: Diagnostic testing for SARS-CoV-2, 2020 Interim guidance. https://www.who.int/publications/i/item/diagnostic-testing-for-sars-cov-2
25. Xiao AT, Tong YX, Zhang S, False-negative of RT-PCR and prolonged nucleic acid conversion in COVID-19: Rather than recurrence: J Med Virol, 2020; 92(10); 1755-56
26. Kelly JC, Dombrowksi M, O’Neil-Callahan M, False-negative COVID-19 testing: Considerations in obstetrical care: Am J Obstet Gynecol, 2020; 2(3); 100130
27. World Health Organization: Use of laboratory methods for SARS diagnosis, 2020 https://www.who.int/csr/sars/labmethods/en/
28. OSANG Healthcare Co Ltd: Instructions for Use GeneFinder™ COVID-19 PLUS RealAmp Kit Available from http://www.osanghc.com/en/customer_en/archive/?uid=51&mod=document
29. World Health Organization: Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases, Interim guidance 17 January 2020, 2020 https://www.who.int/publications/i/item/laboratory-testing-of-2019-novel-coronavirus-(2019-ncov)-in-suspected-human-cases-interim-guidance-17-january-2020
30. World Health Organization, Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases: Interim guidance March 2, 2020 https://www.who.int/publications/i/item/10665-331501
31. World Health Organization: Overview of coronavirus disease (COVID-19) by date 2020 https://covid19.who.int/region/euro/country/pl
32. : SARS-CoV-2 positive cases rate per 100.000 in Poland and Masovia, 2020 (in Polish)https://koronawirusunas.pl/wojewodztwo-mazowieckie
33. Worldometer: Total Coronavirus Cases in New York Total Coronavirus Deaths in New York, 2020 https://www.worldometers.info/coronavirus/usa/new-york/
34. Fassett MJ, Lurvey LD, Yasumura L, Universal SARS-Cov-2 screening in women admitted for delivery in a large managed care organization: Am J Perinatol, 2020; 37(11); 1110
35. LaCourse SM, Kachikis A, Blain M, Low prevalence of SARS-Cov-2 among pregnant and postpartum patients with universal screening in Seattle, Washington: Clin Infect Dis, 2020 [Online ahead of print]
36. Šterbenc A, Sršen TP, Lučovnik M, Usefulness of COVID-19 screen-and-test approach in pregnant women: An experience from a country with low COVID-19 burden: J Perinat Med, 2020 [Online ahead of print]
37. Farfour E, Lesprit P, Visseaux B, The Allplex 2019-nCoV (Seegene) assay: Which performances are for SARS-CoV-2 infection diagnosis?: Eur J Clin Microbiol Infect Dis, 2020; 39(10); 1997-2000
38. Worldometer: COVID-19 CORONAVIRUS PANDEMIC. Reported Cases and Deaths by Country, Territory, or Conveyance, 2020 https://www.worldometers.info/coronavirus/
In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952