11 March 2021: Clinical Research
Long-Term Stability of Intraocular Lens with Trimmed or Untrimmed Haptics in Yamane Sutureless Intrascleral Fixation Technique
Hui Lin1EF, Xiancheng Ye2CD, Xinyu Huang1B, Houshuo Li1B, Zhen Wang1B, Yunli Niu1B, Yanlong Bi1AE*DOI: 10.12659/MSM.928868
Med Sci Monit 2021; 27:e928868
Abstract
BACKGROUND: In intraocular lens (IOL) sutureless intrascleral fixation using the Yamane technique, untrimmed haptics may be overlong in some cases due to varied haptic lengths and individual differences. However, whether trimming the haptic affects IOL stability remains unknown.
MATERIAL AND METHODS: Thirty-nine eyes of 39 patients who underwent Yamane sutureless intrascleral fixation surgery between October 2017 and February 2018 were analyzed retrospectively. Nineteen patients underwent surgery with untrimmed haptics and 20 patients with trimmed haptics. The slit-lamp examination, best-corrected visual acuity (BCVA), corneal endothelial cell density (ECD), IOL position, and complications were assessed.
RESULTS: The mean follow-up periods in the untrimmed and trimmed groups were 27.84±2.89 months and 27.85±2.41 months, respectively. The BCVA improved and ECD decreased in both groups postoperatively (all P<0.01). No significant differences were seen between the 2 groups in postoperative BCVA, postoperative ECD, IOL tilt at 3 months and 24 months, and IOL decentration at 3 months (all P>0.05). There were significant differences between the 2 groups in IOL decentration at 24 months (P<0.05). Compared with postoperative 3 months, both the postoperative IOL tilt and decentration at 24 months increased in the 2 groups (all P<0.01). Postoperative complications included IOL elevation (11% in the untrimmed group and 10% in the trimmed group), as well as haptic extrusion (16% in the untrimmed group).
CONCLUSIONS: The position of the IOL may change with time, but trimming the haptic to an optimum length tends to provide greater IOL stability.
Keywords: Aphakia, Lens Implantation, Intraocular, Sclera, Lenses, Intraocular, Postoperative Complications, Suture Techniques, Sutureless Surgical Procedures, Visual Acuity
Background
Located closest to the original lens, the posterior chamber intraocular lens (IOL) has some inherent advantages. For example, it is not in contact with the corneal endothelium or trabecular meshwork; thus, there is a reduced risk of corneal endothelial cell loss and secondary glaucoma [1,2]. In eyes without sufficient posterior capsular support, transscleral PC IOL fixation can be used to solve this problem.
The techniques of transscleral PC IOL fixation can be broadly classified as either sutured or sutureless. With time, however, the former reportedly has suture-related complications, such as endophthalmitis and delayed IOL dislocation owing to suture degradation or breakage [3,4]. The intrascleral IOL fixation technique was first reported by Gabor et al [5] as a sutureless technique and was further detailed by Agarwal et al [6]. There have also been reports of modified intrascleral IOL fixation techniques [7–12]. Among these, the Yamane double-needle technique with a flanged IOL fixation is an elegant approach because it bypasses the need for significant conjunctival and scleral dissection, glue, or sutures [13]. This technique retained most of the haptic length, making a flange in the end to help fixation. However, the haptic may be overlong in some cases due to the different models of IOLs and individual differences. Whether trimming the haptic affects IOL stability or visual outcome remains unknown.
Here, based on the Yamane technique, we present a method that easily achieves the appropriate length of the haptics during surgery, and we compare the long-term IOL stability and visual outcome between these 2 approaches with or without this method.
Material and Methods
SURGICAL TECHNIQUE:
In the untrimmed group, a 25-gauge pars plana vitrectomy was performed under peribulbar anesthesia. A 3-piece IOL (AR40e, Advanced Medical Optics, Santa Ana, USA) was inserted into the anterior chamber using an injector from the 3.0-mm sclerocorneal incision at 12 o’clock, and the trailing haptic was kept outside to prevent the IOL from falling into the vitreous cavity. Two scleral tunnels were created at 3 o’clock and 9 o’clock using two 27-gauge needles 2.0 mm from the limbus. The angle was 20° with respect to the limbus and 5° with respect to the surface of the iris. Then, the leading haptic was threaded into the needle using forceps, and the same process was repeated with the trailing haptic. Both haptics were externalized, and the ends of the haptics were cauterized using an ophthalmic cautery device to create flanges, which were pushed back and fixed into the scleral tunnels.
In the trimmed group, most of the steps were the same except for the trimming of the haptics. Before the IOL was inserted into the anterior chamber, a 2.0-mm section was cut off the haptics (the initial length is about 9.0 mm) to reduce the difficulty in introducing the haptics caused by the overlong haptics in the eye (Figure 2A). After externalizing both haptics and adjusting the IOL position, two 1.0-mm sections were marked with calipers as the flanged positions from which the haptics passed through the sclera. The unmarked parts (approximate range: 0.5–1.0 mm) were cut off, and the 1.0-mm marks were cauterized to create flanges and fixed like the untrimmed group (Figure 2B, 2C).
STATISTICAL ANALYSES:
Statistical analyses were performed using SPSS 19.0. Data are shown as mean ± standard deviation. The preoperative and postoperative data in each group were compared using the Wilcoxon signed-rank test. The Mann-Whitney
Results
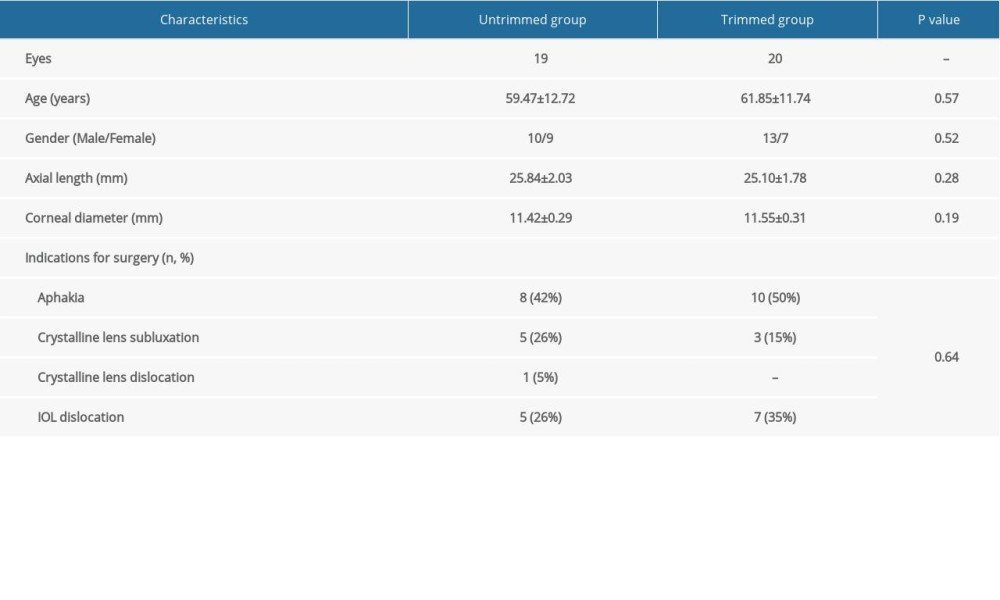
Thirty-nine eyes of 39 patients were analyzed. Nineteen patients (10 men, 9 women; mean age, 59.47±12.72 years) comprised the untrimmed group. Another 20 patients (13 men, 7 women; mean age, 61.85±11.74 years) comprised the trimmed group. The major indication for surgery in both groups was aphakia that did not complete IOL implantation in the last ocular operation, which was 42% in the untrimmed group and 50% in the trimmed group. Other indications included crystalline lens subluxation/dislocation and IOL dislocation (Table 1). The baseline data, including age, sex, axial length, corneal diameter, and indications for surgery, were not statistically significantly different between the 2 groups (
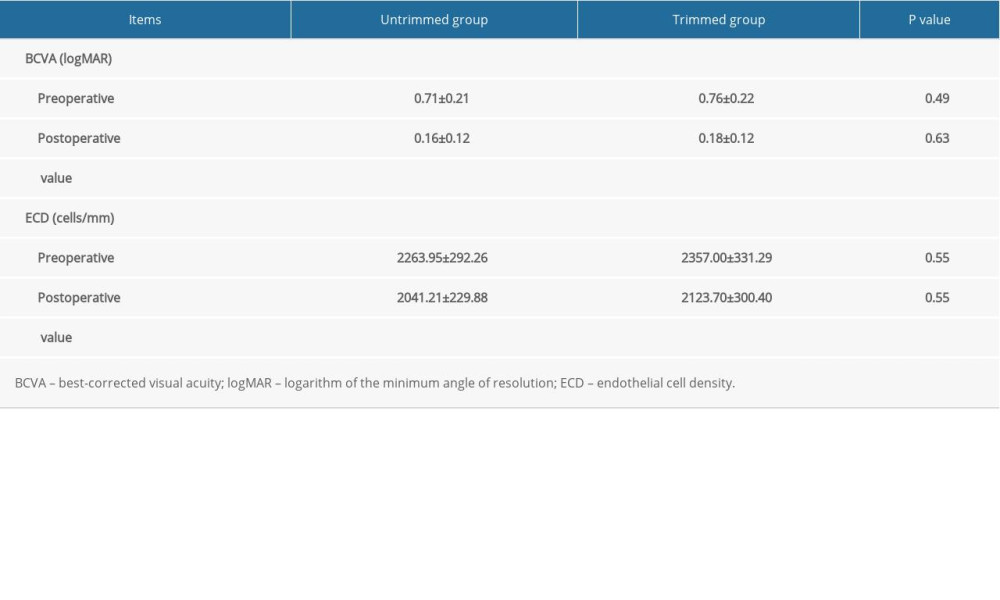
As shown in Table 2, the postoperative BCVA improved from 0.71±0.21 (logMAR) to 0.16±0.12 (logMAR) in the untrimmed group (
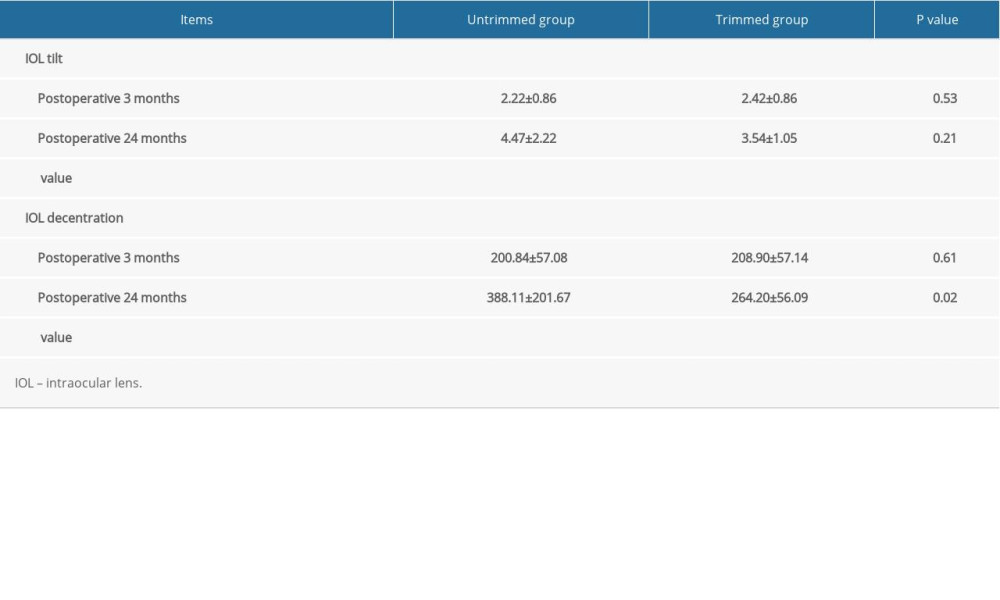
As shown in Table 3, compared with postoperative values at 3 months, the postoperative IOL tilt at 24 months increased from 2.22±0.86° to 4.47±2.22° in the untrimmed group (
The IOL haptics location detected by AS-OCT at 24 months showed different states of haptics between the untrimmed group (Figure 3A) and the trimmed group (Figure 3B). The untrimmed haptic was overlong and would push out of the scleral tunnel as time went by.
Postoperative complications included IOP elevation in 2 eyes (11%) and haptic extrusion in 3 eyes (16%) in the untrimmed group (Figure 4) and IOP elevation in 2 eyes (10%) in the trimmed group. The IOP elevation was treated with eye drops, and the IOP recovered to normal in these eyes within 1 month. The haptic extrusion in 3 eyes has not interfered with visual acuity or caused other complications; therefore, these cases continue to be observed.
Discussion
Since the sutureless intrascleral technique was introduced, it has been modified by many surgeons. The main differences among the modified techniques are the creation of scleral tunnels, externalization of the haptics, and fixation of the haptics.
First, scleral tunnels can be created by any gauge cannula, but smaller ones (such as 27-gauge and 30-gauge) are more suitable as they create smaller and tighter scleral tunnels, produce less tissue injury, and have a reduced risk of postoperative hypotony [13,14].
Second, externalization of the haptics can be completed using intraocular forceps [5], sutures [11], and needles [7,13]. Although using forceps is simple and convenient, it can cause haptic deformation or fracture when the direction of traction is different from the haptic. Using sutures tied to the haptics as a guidance can reduce anterior segment manipulations. However, due to the limited traction of sutures, the scleral tunnels in this technique are short, although the long-term IOL stability remains to be assessed. The double-needle technique reported by Yamane can guide haptic externalization under relatively uniform stress, and the length of scleral tunnels would be sufficient.
Lastly, there are many ways to fix haptics, such as scleral flaps with sutures or fibrin glue [6,11], Y-fixation [8], hook-shaped haptics [12], and flanged haptics [13]. Kumar et al reported that the degree of scleral erosion by the haptic was associated with the depth of scleral tunnels, and a subconjunctival haptic extrusion was seen in 3 eyes (1.4%) with thin scleral flaps [14,15]. The technique requires sclera thick enough to create scleral flaps and prevent the haptic extrusion, although it is not easy to achieve this in some cases, such as Marfan’s syndrome. The Y-fixation creates scleral tunnels parallel to the limbus, which makes the haptics bend abnormally and increases the risk of haptic extrusion [16]. The hook-shaped haptic reported by Yoshida is fixated within the tunnels created with a 23-gauge MVR blade. In this technique, the tunnels might be too large for the haptics, which could result in haptic extrusion or hypotension after surgery. The flanged haptic reported by Yamane is easy to achieve, and the flange at the end of the haptic also plays a plug-like role in making the scleral tunnel watertight.
Overall, the Yamane technique is comparatively better for sutureless intrascleral fixation, although it requires skill and experience in externalizing of the haptics. However, due to the varying models of IOLs and individual differences, the untrimmed haptics may be overlong in some cases that use the Yamane technique. Merely pushing the haptics back into the scleral tunnels can place an unnatural tension on the haptics and the reactive force produced by the haptics themselves would push them out, causing haptic extrusion. Thus, we presented a method that easily achieves an optimum haptic length during surgery and compared the long-term IOL stability and visual outcome between these 2 approaches with or without this method.
In this study, we used a 27-gauge needle because the haptics of routinely used lenses, such as Tecnis ZA9003 (Abbott Medical Optics, Santa Ana, America) and Sensar AR40e (Abbott Medical Optics, Santa Ana, America), cannot be threaded into the routine 30-gauge needle, and the thin-walled 30-gauge needle reported by Yamane is not readily available. We even found that the haptic was sometimes difficult to thread through the 27-gauge needle, and distending the needle lumen by another needle was feasible. The scleral tunnel can be watertight and the haptic can also be fixed well, as long as the flange is made into a proper size.
In the trimmed group, the haptic was trimmed twice. The first time before the IOL insertion into the anterior was to reduce the difficulty in introducing the haptics caused by the overlong haptics in the eye, especially in the introduction of the trailing haptic. This problem has also been pointed out by some surgeons [11,17]. In our experience, the trimmed haptic is easier threaded into the needle. The second trimming was performed after externalizing and adjusting the IOL position to finally achieve the optimum haptic length, which slightly stretched the haptics to avoid haptic extrusion. We marked off two 1-mm points from which the haptics passed through the sclera as the flanged positions to achieve better IOL centration. Given that the haptic length was different in varying models of IOLs, such as 8.6 mm in NX-70 (Santen, Osaka, Japan) and 8.2 mm in PN6A (Kowa, Tokyo, Japan) [18], the first step can be skipped when using IOLs with shorter haptics.
In this study, there were no significant differences between the 2 groups in postoperative BCVA, postoperative ECD, IOL tilt at 3 months and 24 months, and IOL decentration at 3 months. However, IOL decentration between the 2 groups was significantly different at 24 months, and haptic extrusion was seen in 3 eyes (16%) in the untrimmed group, suggesting that the trimmed group had greater IOL stability. Similar to our study, Kurimori et al reported 2 cases of severe tilting of an IOL after using the Yamane technique and proposed that the tilt can be reduced by shortening the length of the haptics [19]. Additionally, both IOL tilt and IOL decentration between 3 months and 24 months were significantly different in the 2 groups, suggesting that the position of the IOL can change with time. This may be related to the haptic distortion or slide caused by daily oculogyration and eye rubbing.
In our cases, IOP elevation appeared in both groups, which may be caused by mild viscoelastic material retention. Transient IOP elevation was controlled by antiglaucoma medications, without affecting the final visual outcome [20]. The haptic extrusion was only seen in the untrimmed group; fortunately, the exposed part was symmetric and subconjunctival, and visual acuity was not severely affected, nor did other complications appear. Long-term observation is still needed. If the exposed part becomes asymmetric, the IOL may tilt and be severely off-center, which can have a strong impact on visual acuity. More Severe complications, such as endophthalmitis, may appear if the exposed part is out of the conjunctiva.
In summary, achieving an optimum haptic length is necessary when proceeding with sutureless intrascleral fixation using the Yamane technique. The personalized method of trimming can slightly stretch the haptic, generating a pull force to provide a more stable fixation.
There are limitations to our study, including the limited sample size and the single model of IOL used. Although the baseline data were not significantly different between the 2 groups, eyes with haptic extrusion were highly myopic, suggesting that the haptic is easily exposed in thinner sclera. Larger samples with more high-myopia cases are needed for further study. More stable fixations of the IOL in eyes without sufficient posterior capsular support still need to be explored in the future.
Conclusions
In this study, we provide an easy method to achieve the optimum length of the haptic by trimming the haptic at the point at which it passes through the sclera after adjusting the IOL position and centration. Although the position of the IOL can change with time, trimming the haptic to an optimum length tends to provide greater IOL stability.
Figures
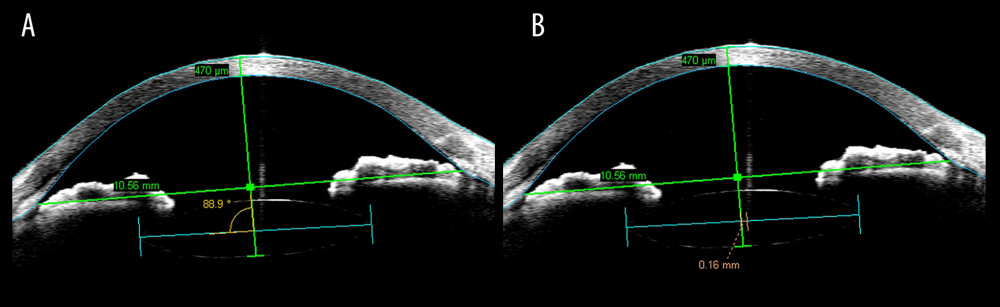 Figure 1. Anterior segment optical coherence tomography (AS-OCT) images show the method used to measure IOL tilt (A) and IOL decentration (B). The angle shown in (A) is the supplementary angle of the angle of IOL tilt.
Figure 1. Anterior segment optical coherence tomography (AS-OCT) images show the method used to measure IOL tilt (A) and IOL decentration (B). The angle shown in (A) is the supplementary angle of the angle of IOL tilt. 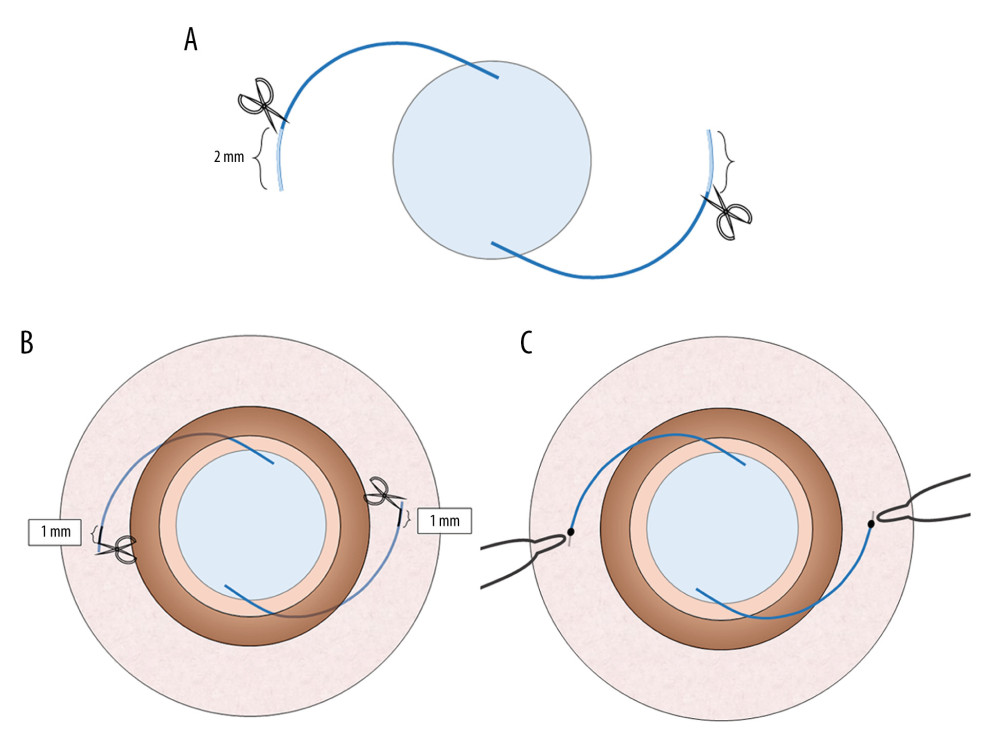 Figure 2. The diagrams show the double trimming of the haptics in the trimmed group. A 2.0-mm section was cut off the haptics before the IOL was inserted into the eye (A). Two 1.0-mm sections were marked at points from which the haptics passed through the sclera, and the unmarked parts were cut off (B). The 1.0-mm marks were cauterized to create flanges and fixed into the scleral tunnels (C).
Figure 2. The diagrams show the double trimming of the haptics in the trimmed group. A 2.0-mm section was cut off the haptics before the IOL was inserted into the eye (A). Two 1.0-mm sections were marked at points from which the haptics passed through the sclera, and the unmarked parts were cut off (B). The 1.0-mm marks were cauterized to create flanges and fixed into the scleral tunnels (C). 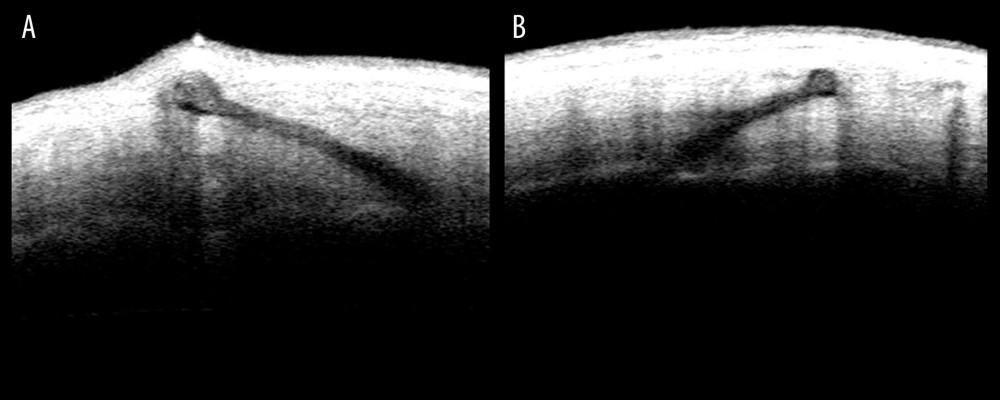 Figure 3. Anterior segment optical coherence tomography (AS-OCT) images show the location of the haptics. The flange of the untrimmed haptic is seen pushing up the partial sclera in (A). The flange of the trimmed haptic was well-fixed in the sclera in (B).
Figure 3. Anterior segment optical coherence tomography (AS-OCT) images show the location of the haptics. The flange of the untrimmed haptic is seen pushing up the partial sclera in (A). The flange of the trimmed haptic was well-fixed in the sclera in (B). 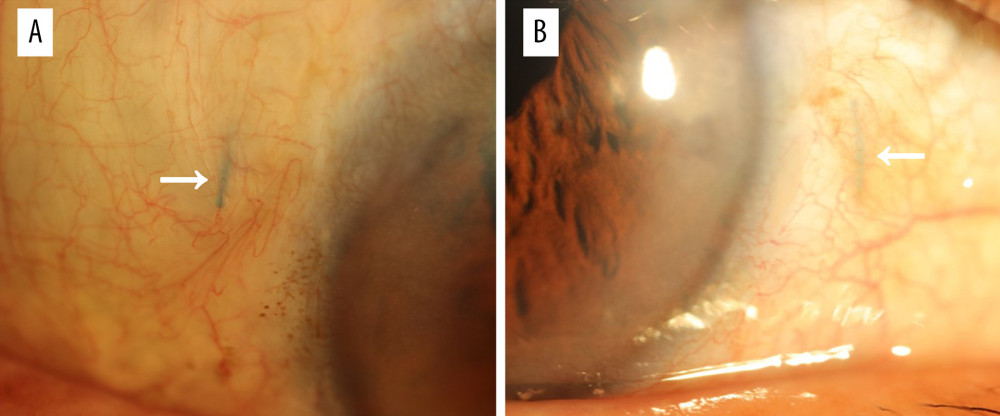 Figure 4. A slit-lamp microscopy image shows the subconjunctival haptic extrusion at the temporal side (A, arrow) and the nasal side (B, arrow).
Figure 4. A slit-lamp microscopy image shows the subconjunctival haptic extrusion at the temporal side (A, arrow) and the nasal side (B, arrow). References
1. Drolsum L, Long-term follow-up of secondary flexible, open-loop, anterior chamber intraocular lenses: J Cataract Refract Surg, 2003; 29; 498-503
2. Evereklioglu C, Er H, Bekir NA, Comparison of secondary implantation of flexible open-loop anterior chamber and scleral-fixated posterior chamber intraocular lenses: J Cataract Refract Surg, 2003; 29; 301-8
3. Lockington D, Ali NQ, Al-Taie R, Outcomes of scleral-sutured conventional and aniridia intraocular lens implantation performed in a university hospital setting: J Cataract Refract Surg, 2014; 40; 609-17
4. McAllister AS, Hirst LW, Visual outcomes and complications of scleral-fixated posterior chamber intraocular lenses: J Cataract Refract Surg, 2011; 37; 1263-69
5. Gabor SGB, Pavlidis MM, Sutureless intrascleral posterior chamber intraocular lens fixation: J Cataract Refract Surg, 2007; 33; 1851-54
6. Agarwal A, Kumar DA, Jacob S, Fibrin glue-assisted sutureless posterior chamber intraocular lens implantation in eyes with deficient posterior capsules: J Cataract Refract Surg, 2008; 34; 1433-38
7. Yamane S, Inoue M, Arakawa A, Kadonosono K, Sutureless 27-gauge needle-guided intrascleral intraocular lens implantation with lamellar scleral dissection: Ophthalmology, 2014; 121; 61-66
8. Ohta T, Toshida H, Murakami A, Simplified and safe method of sutureless intrascleral posterior chamber intraocular lens fixation: Y-fixation technique: J Cataract Refract Surg, 2014; 40; 2-7
9. Takayama K, Akimoto M, Taguchi H, Transconjunctival sutureless intrascleral intraocular lens fixation using intrascleral tunnels guided with catheter and 30-gauge needles: Br J Ophthalmol, 2015; 99; 1457-59
10. Zhang Y, He F, Jiang J, Modified technique for intrascleral fixation of posterior chamber intraocular lens without scleral flaps: J Cataract Refract Surg, 2017; 43; 162-66
11. Yang Y, Yao T-t, Zhou Y-l, A modified intrascleral intraocular lens fixation technique with fewer anterior segment manipulations: 27-gauge needle-guided procedure with built-in 8-0 absorbable sutures: BMC Ophthalmol, 2019; 19; 234
12. Yoshida N, Kojima T, Yokoyama S, New surgical approach for intrascleral fixation using an intraocular lens with hook-shaped haptics: J Cataract Refract Surg, 2018; 44; 129-33
13. Yamane S, Sato S, Maruyama-Inoue M, Kadonosono K, Flanged intrascleral intraocular lens fixation with double-needle technique: Ophthalmology, 2017; 124; 1136-42
14. Todorich B, Thanos A, Woodward MA, Wolfe JD, Sutureless intrascleral fixation of secondary intraocular lens using 27-gauge vitrectomy system: Ophthalmic Surg Lasers Imaging Retina, 2016; 47(4); 376-79
15. Kumar DA, Agarwal A, Glued intraocular lens: A major review on surgical technique and results: Curr Opin Ophthalmol, 2013; 24; 21-29
16. Matsui Y, Matsubara H, Hanemoto T, Kondo M, Exposure of haptic of posterior chamber intraocular lens after sutureless intrascleral fixation: BMC Ophthalmol, 2015; 15; 104
17. Kim DB, Trailing-haptic-first modification of double-needle intrascleral haptic fixation technique: J Cataract Refract Surg, 2018; 44; 424-28
18. Miura Y, Harada Y, Kiuchi Y, Comparison of different IOL types in the flanged IOL fixation technique: J Ophthalmol, 2020; 2020 8534028
19. Kurimori HY, Inoue M, Hirakata A, Adjustments of haptics length for tilted intraocular lens after intrascleral fixation: Am J Ophthalmol Case Rep, 2018; 10; 180-84
20. Aaltonen P, Oskala P, Immonen I, Outcomes of intraocular lens scleral fixation with the friction knot technique: Acta Ophthalmol (Copenh), 2019; 97; e506-13
Figures
 Figure 1. Anterior segment optical coherence tomography (AS-OCT) images show the method used to measure IOL tilt (A) and IOL decentration (B). The angle shown in (A) is the supplementary angle of the angle of IOL tilt.
Figure 1. Anterior segment optical coherence tomography (AS-OCT) images show the method used to measure IOL tilt (A) and IOL decentration (B). The angle shown in (A) is the supplementary angle of the angle of IOL tilt. Figure 2. The diagrams show the double trimming of the haptics in the trimmed group. A 2.0-mm section was cut off the haptics before the IOL was inserted into the eye (A). Two 1.0-mm sections were marked at points from which the haptics passed through the sclera, and the unmarked parts were cut off (B). The 1.0-mm marks were cauterized to create flanges and fixed into the scleral tunnels (C).
Figure 2. The diagrams show the double trimming of the haptics in the trimmed group. A 2.0-mm section was cut off the haptics before the IOL was inserted into the eye (A). Two 1.0-mm sections were marked at points from which the haptics passed through the sclera, and the unmarked parts were cut off (B). The 1.0-mm marks were cauterized to create flanges and fixed into the scleral tunnels (C). Figure 3. Anterior segment optical coherence tomography (AS-OCT) images show the location of the haptics. The flange of the untrimmed haptic is seen pushing up the partial sclera in (A). The flange of the trimmed haptic was well-fixed in the sclera in (B).
Figure 3. Anterior segment optical coherence tomography (AS-OCT) images show the location of the haptics. The flange of the untrimmed haptic is seen pushing up the partial sclera in (A). The flange of the trimmed haptic was well-fixed in the sclera in (B). Figure 4. A slit-lamp microscopy image shows the subconjunctival haptic extrusion at the temporal side (A, arrow) and the nasal side (B, arrow).
Figure 4. A slit-lamp microscopy image shows the subconjunctival haptic extrusion at the temporal side (A, arrow) and the nasal side (B, arrow). In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952