23 February 2021: Animal Study
Different Responses to Identical Trauma Between BALB/C and C57BL/6 Mice
Xiuying Chen1CDEF, Chang Cheng1BCD, Wenchao Cheng2EF, Yuhan Wang1EF, Xuzheng Zuo1B, Weiju Tang1B, Zhanyang Yu3CD, Zhihuan Yang4B, Zhengguo Wang4AD, Peifang Zhu4AG, Wen Huang1ADG*DOI: 10.12659/MSM.928676
Med Sci Monit 2021; 27:e928676
Abstract
BACKGROUND: Different responses to identical trauma may be related to the genetic background of individuals, but the molecular mechanism is unclear. In this study we investigated the heterogeneity of trauma in mice and the potential biological explanations for the differences.
MATERIAL AND METHODS: Compared with other organs, the pathological response of the lung after injury is the earliest and most serious. We used C57BL/6 and BALB/C mice to explore the genetic background of different responses to trauma in the lung. We measured mortality rate, pulmonary microvascular permeability, and Cxcl15 gene expression in BALB/C and C57BL/6 mice before and after blast-wave injury. Microvascular permeability was measured using a fluorescent tracer, and Cxcl15 gene expression level and expression distribution were measured using fluorogenic probe quantitative polymerase chain reaction and northern blot.
RESULTS: C57BL/6 mice showed lower mortality rates and pulmonary microvascular permeability than BALB/C mice after blast-wave injury; there was no significant difference in the permeability before blast-wave injury. The Cxcl15 gene was expressed specifically in the lung tissue of mice. The level of Cxcl15 expression in BALB/C mice was higher than in C57BL/6 mice before and after injury, and the variation trend of Cxcl15 expression level after injury was significantly different between BALB/C and C57BL/6 mice.
CONCLUSIONS: Our results indicated that BALB/C and C57BL/6 mice had significant heterogeneity in posttraumatic response in terms of mortality and degree of lung damage. The differences in genetic factors such as Cxcl15 may have played a role in this heterogeneity.
Keywords: Genetic Heterogeneity, Lung Injury, Blast Injuries, Capillary Permeability, Chemokines, CXC, Lung, Wounds and Injuries
Background
Individuals have differential responses to similar traumatic events, a phenomenon known as the heterogeneity of traumatic responses. Most studies have used differences in nutritional, physical, or mental state to explain this phenomenon; however, the molecular mechanism remains unclear.
Increasing evidence [1–3] suggests that individual differences in genetic background may be an important reason for the heterogeneity of traumatic responses. Previous investigation in our laboratory [4] revealed that the pathological response in the lung, compared with other organs, is the earliest and most serious after injury. In our study, we randomly anatomized surviving mice 72 h after injury. Upon visual examination, we found that the degree of lung injury in BALB/c mice appeared more severe than in C57BL/6, and this finding was confirmed by microscopic imaging of pathological sections. In addition, our previous gene chip results suggested that the expression of the
Given the previous findings, in addition to acute lung injury accounting for high rates of patient morbidity and mortality after physically traumatic events [5] and pulmonary microvascular permeability being an early impairment event after trauma that triggers further inflammatory responses, we speculated that anatomical structures associated with the microvascular permeability (such as vascular diameter, vascular compliance, and cell conjunction), among other factors, were associated with the trauma response differences. In order to explore the genetic background of different responses to trauma in the lung, we tested pulmonary vascular permeability before and after injury in C57BL/6 and BALB/C mice. We additionally detected
Material and Methods
ANIMAL MODEL AND INJURY CONDITION:
In this study, we used inbred 8- to 9-week-old specific pathogen-free BALB/C and C57BL/6 mice (Shanghai Laboratory Animal Centre of the Chinese Academy of Sciences, Beijing, China). The average weight of the mice was 20.78±1.25 g, and the weight difference in the same batch of experimental animals was <1.00 g. We used pentobarbital anesthesia immediately before the blast injury procedure at a dose of 40 mg/kg. The animals were humanely sacrificed by cervical dislocation 72 h after blast injury.
The systemic blast injury (SBI) model was designed with a BST type-1 biological shock tube (Research Institute of Field Surgery, Army Medical University, Beijing, China) with a driving pressure of 6.0 MPa. Overpressure peak value and positive pressure action time of the vertical plane was measured by a baroceptor. Data were analyzed with DASYlab 5.0 software (Data Acquisition System Laboratory).
Mice were kept at room temperature (22±2°C), with ad libitum access to food and water. The Institutional Animal Care and Use Committee of the Army Medical University approved all animal care and experimentation (SYXK-PLA-2007035). Efforts were made to minimize the suffering of the mice. Due to the experimental intervention, no mice died without euthanasia.
OBSERVATION INDICES:
Mortality rate was measured at 1, 6, 24, and 72 h after injury (60 BALB/C mice and 59 C57BL/6 mice). Pulmonary tissue sections were extracted from euthanized animals and prepared according to standard procedures. Pathomorphological examination of the sections by light microscopy, using routine hematoxylin and eosin staining, revealed the hemorrhagic and edemic state of pulmonary tissues.
COMPARISON OF MICROVASCULAR PERMEABILITY:
Microvascular permeability was measured by the fluorescence tracer method at 0.5, 1, and 4 h after injury in 20 BALB/C mice and 20 C57BL/6 mice. Prior to trauma, 5 BALB/C mice and 5 C57BL/6 mice were injected with 0.2 mL of 0.5% fluorescein sodium (FINa) (with saline water as a blank control) through the caudal vein for 10 min, and 0.2 g of pulmonary tissue was collected, homogenized, and centrifuged (4000 rpm, 20 min). The supernatant was measured by fluorescence spectrophotometer (437-nm excitation, 518 nm emitted, and 5-mm slit width). Microvascular permeability was indirectly measured by the quantity of FINa per gram of tissue using a fluorescence intensity standard curve. The standard curve was delineated according to a standard intensity. The regression equation was
RNA EXTRACTION AND FLUORESCENCE QUANTITATIVE POLYMERASE CHAIN REACTION:
Primer sequences were synthesized from Shanghai Shenyou Company as follows:
A previously described procedure was used for RNA extraction [6], and RNA was stored at 4°C for future use. An extracted RNA sample volume of 20 μL was made up by 3 μL of template, 10×2 μL buffer, 4 μL of MgCl2 (25 mmol/L), 2 μL of dNTP (10 mmol/L), 0.5 μL of enzyme inhibitor (40 U/μL), 1 μL of AWV reverse transcriptase (5 U/μL), 1 μL of Oligo dT-Adaptor primer (2.5 pmol/μL), and 6.5 μL of enzyme-free water. Water was added to the template for denaturation at 72°C, and then the rest of the sample volume was added for the reaction. The reaction conditions were 72°C for 2.5 min, 42°C for 40 min, and 95°C for 5 min, respectively. The sample volume for fluorescence quantitative polymerase chain reaction (PCR) included 1 μL of template, 10×5 μL of buffer, 5 μL of MgCl2 (25 mmol/L), and 1 μL of dNTP (10 mmol/L). The following were then added to the reaction system: 0.8 μL of sense chain (20 pmol/μL), 0.8 μL of antisense strand (20 pmol/μL), 0.4 μL of probe (20 pmol/μL), 0.5 μL of Taq polymerase (5 U/μL), and 50 μL of H2O. The efficiency of the PCR was evaluated for each of the amplification conditions. Reaction conditions were as follows: 94°C for 2 min, 94°C for 10 s, 53°C for 30 s, and 72°C for 40 s, repeating for 45 cycles, followed by 72°C for 10 min.
NORTHERN BLOT ANALYSES:
Tissue samples from the heart, brain, spleen, lungs, liver, skeletal muscles, kidneys, and testicles were used for northern blot analyses. The expression of
An established protocol [6] was used for construction of the random primers and labeled cDNA probes and for purification.
IN SITU HYBRIDIZATION:
Paraffin sections were prepared from pulmonary tissue of mice. The sections were placed on RNAse-free glass slides coated with adhesive. The slides were air-dried for 2 h, washed in diethyl pyrocarbonate (DEPC) plus phosphate-buffered saline (PBS), and then washed 3 times in DEPC-water, before being stored at −20°C.
IMMUNOSTAINING FOR DIGOXIGENIN:
Pretreatment of the sections and washing were done according to a previously reported method [7]. Slides were incubated with pre-absorbed antidigoxigenin antibody (coupled to alkaline phosphatase) diluted to a concentration of 1: 2000 in 20% sheep serum in PBS with Tween (PBT) overnight at 4°C. Slides were then washed 3 times for 30 min each in PBT at room temperature. Next, they were washed in alkaline phosphatase buffer without levamisole for 5 min, followed by levamisole to block endogenous alkaline phosphatase at room temperature for 5 min. Subsequently, 4.5 μL of nitroblue tetrazolium (NBT) and 3.5 μL of 5-bromo-4-chloro-3-indolyl phosphate (BCIP) were added to each milliliter of alkaline phosphatase buffer, and the slides were immersed in this solution. Slides were then developed in the dark for up to 16 h, depending on the abundance of RNA. The slides were washed with PBS after the process was completed. Alkaline phosphatase buffer was used to wash out the NBT/BCIP; because of the stability of alkaline phosphatase, the reaction could be repeated at a later time. Slides were fixed MEMFA [composed of 0.1 M 3-morpholinopropanesulfoinc acid, 2 mM ethylenebis(oxyethylenenitrilo)tetraacetic acid,1 mM magnesium sulfate, and 3.7% formaldehyde] at room temperature for at least 15 min. Mounting of slides, hybridization, processing, and color development were based on previous methods [7].
STATISTICAL ANALYSES:
Data were analyzed using SPSS18.0 software (SPSS, Chicago, IL, USA). The mortality rate between the 2 mouse lines and between male and female mice in the same group were compared by χ2 test. Microvascular permeability was compared by independent sample
Results
DIFFERENCES IN MORTALITY BETWEEN BALB/C AND C57BL/6 MICE AFTER BLAST INJURY:
The mortality rates at 1, 6, 24, and 72 h after injury were measured (Table 1). Our data show a significant difference in total mortality between BALB/C and C57BL/6 mice after blast injury (P<0.01). C57BL/6 mice showed greater resistance to blast injury and a higher survival rate compared with BALB/C mice. Most of the animal deaths due to blast injury occurred within 1 h. No significant difference in mortality was found between male and female mice of the same strain (Table 2).
COMPARISON OF PATHOLOGICAL CHANGES BEFORE AND AFTER LUNG INJURY:
We evaluated the histology of the mouse lungs after injury, using tissue sections stained with hematoxylin and eosin. We noted pathological manifestations of acute lung injury within 72 h after blast-wave injury. Our findings showed a large number of red blood cells, increased alveolar edema fluid, a widened alveolar gap, inflammatory cell infiltration, shedding of bronchial epithelial cells, and disruption of normal lung tissue structure (Figure 1). Overall, the pathological changes in C57BL/6 mice were significantly milder compared with BALB/C mice.
CHANGES IN PULMONARY PERMEABILITY:
We calculated FINa levels as a measure of microvascular permeability (Table 3). At 1 and 4 h after injury, FINa levels in BALB/C mice were significantly higher than in C57BL/6 mice (P<0.05). A significant change in the level of FINa occurred earlier in BALB/C mice compared with C57BL/6 mice (0.5 vs 1 h, respectively; P<0.05). The pulmonary microvascular permeability in BALB/C mice was significantly higher than in C57BL/6 mice at 1 and 4 h after injury. Interestingly, the microvascular permeability of both strains continued to increase up to 4 h after injury.
:
Cxcl15 gene expression was measured by fluorescence quantitative PCR. Under physiological conditions, the expression of Cxcl15 in C57BL/6 mice was significantly lower than in BALB/C mice (P<0. 01; Table 4).
Furthermore, we found Cxcl15 gene expression in BALB/C mice was significantly higher than in C57BL/6 mice after injury (P<0.05). Cxcl15 gene expression continuously increased until 6 h after injury in BALB/C mice, but it descended in C57BL/6 mice at 1 h after injury. Peak expression level of Cxcl15 was observed at 6 h after injury and return to baseline after 24 h (Table 5, Figures 2, 3A, 3B).
:
The Cxcl15 gene was hybridized in 8 major tissue sources of mice on Poly A+RNA blots by the northern blot method (Figure 3C). Our results show Cxcl15 gene expression, specifically in lung. Our data also suggest that the Cxcl15 gene had 2 subtypes, one that was 1 kb and another that was 2.4 kb.
:
We investigated the distribution of the Cxcl15 gene in lung tissues of mice with in situ hybridization. Anti-digoxigenin antibody labeled with alkaline phosphatase was applied, and a positive signal was the development of a purple color following addition of BCIP/NBT after labeling with a digoxin random primer. The results suggest that expression of the Cxcl15 gene was located mainly in the cytoplasm of the bronchial epithelial cells and may also have been present in the nucleus, which needs to be further characterized (Figure 3D, 3E).
Discussion
A heterogeneous traumatic response has been observed among individuals, with different genetic backgrounds presumably explaining varying degrees of resistance to the same level of trauma. The majority of published studies offer preliminary data, but they lack sufficient evidence to form a useful clinical explanation. Therefore, in this study we sought to address some key discrepancies regarding which animal models are most appropriate and what experimental measures best represent relevant measurements to support human diagnostic and therapeutic advances.
The most relevant experimental animal model should satisfy several key conditions. First, it should offer a heterogeneous traumatic response. Second, its genetic background should be well defined. Therefore, we used BALB/C and C57BL/6 inbred mice as experimental animals in this study. They have similar body weights, and the inbreeding coefficient of homologous series mice reaches 98.6%, with hybrid genes representing 1.4% [8]. Some studies have indicated that BALB/C and C57BL/6 mice have different responses to some pathological factors, mainly due to differences in their genetic backgrounds [9–11]. Therefore, we believe that inbred animals are most suitable for studying heterogeneity of traumatic responses.
Regarding the choice of injury model, we believe that the most relevant experimental system should satisfy the following conditions: have a single-wound agent, the most appropriate being a physical one; follow a nonanesthetized animal protocol; be capable of processing a high number of animals quickly; and demonstrate reproducibility. Previous studies indicate that the shockwave generated by a biological shock tube offers a straightforward method to control injury pressure with consistent, systemic trauma to animals. Thus, this injury method has become the standard research protocol. The BST type-1 biological shock tube manufactured by the Research Institute of Field Surgery of the Third Military Medical University generates a stable vertical plane wave producing uniform injury to animals in a fixed posture and position. Our previous work [12] showed that the SBI model with BST type-1 is reliable, reproducible, and comparable among test subjects.
Regarding the choice of index to quantify the sensitivity to trauma of different animals, we believe that mortality rate is the preferred index. Considering that we chose a whole-body shock injury model and that the ultimate effect of trauma is a direct impact on the organism’s survival, we believe that mortality rate is a reliable and easily quantifiable index. In addition to the mortality rate, we chose the pathological changes in pulmonary tissue to quantify the sensitivity to trauma. Previous studies in our laboratory have observed the following phenomena [4]. Compared with other organs, the pathological response in lungs is the earliest and most serious after injury, and it is primarily characterized by congestion and edema. Pulmonary edema is a common characteristic of pulmonary injury. It leads to changes in microvascular permeability, which is associated with vascular structure alteration. Important pathological characteristics of the response to injury include morphological and functional changes in blood vessels, typically their microvascular permeability. The endothelial cytoskeleton plays a critical role in maintaining vascular permeability [13]. We therefore chose microvascular permeability as an indirect index for anatomical structure correlated with the heterogeneity of traumatic responses.
We chose to examine
Our current results revealed significant differences in the mortality and the severity of lung injury between C57BL/6 and BALB/C inbred mice within 72 h after blast injury. The C57BL/6 mice showed greater resistance to the blast injury and had milder lung injuries than the BALB/C mice. Furthermore, most of mouse deaths occurred within 1 h after blast injury. As previously mentioned, we aimed to reduce the impact of nonexperimental factors, and this heterogeneity in the same injury condition was very likely the result of differences in the genetic background between BALB/C and C57BL/6 mice. Furthermore, our results showed no significant differences in mortality and lung injury severity between the male and the female mice of the same strain, which is consistent with previous reports [9,11,21] showing that the differences in acute lung injury susceptibility between different strains of mice is an autosomal genetic characteristic.
We also observed significant death in both strains at 1 h after injury. Thus, we chose this time point to compare microvascular permeability between the 2 strains. Our hematoxylin-eosin staining results showed that the cell structure of BALB/C mice was significantly more damaged compared with the C57BL/6 mice. There was also a significant difference in vascular permeability between the C57BL/6 and BALB/C mice. The increase of pulmonary microvascular permeability occurred earlier in BALB/C mice than in the C57BL/6 mice, and the degree of changes was more dramatic. Our findings were consistent with those of Barone et al [22], who found significant differences in the sensitivity to cerebral ischemia injury in BDF, CFW, and BALB/C mice. Their further studies on cerebral angiotomy revealed that the differences in cerebral ischemia injury were related to the heterogeneity of blood vessels. With regard to mechanisms, previous studies showed that the glycocalyx, caveolae, and pericytes have functional importance for vascular permeability [23]. In a separate study, the phenotypes between C57BL/6 and BALB/c mice were different in the asthma model with different distributions of proinflammatory cytokines and inflammatory cells [24].
Our study also showed that the levels of
Conclusions
In conclusion, our results indicate that a hereditary factor likely plays an important role in the response to trauma and the ultimate outcome. The
Figures
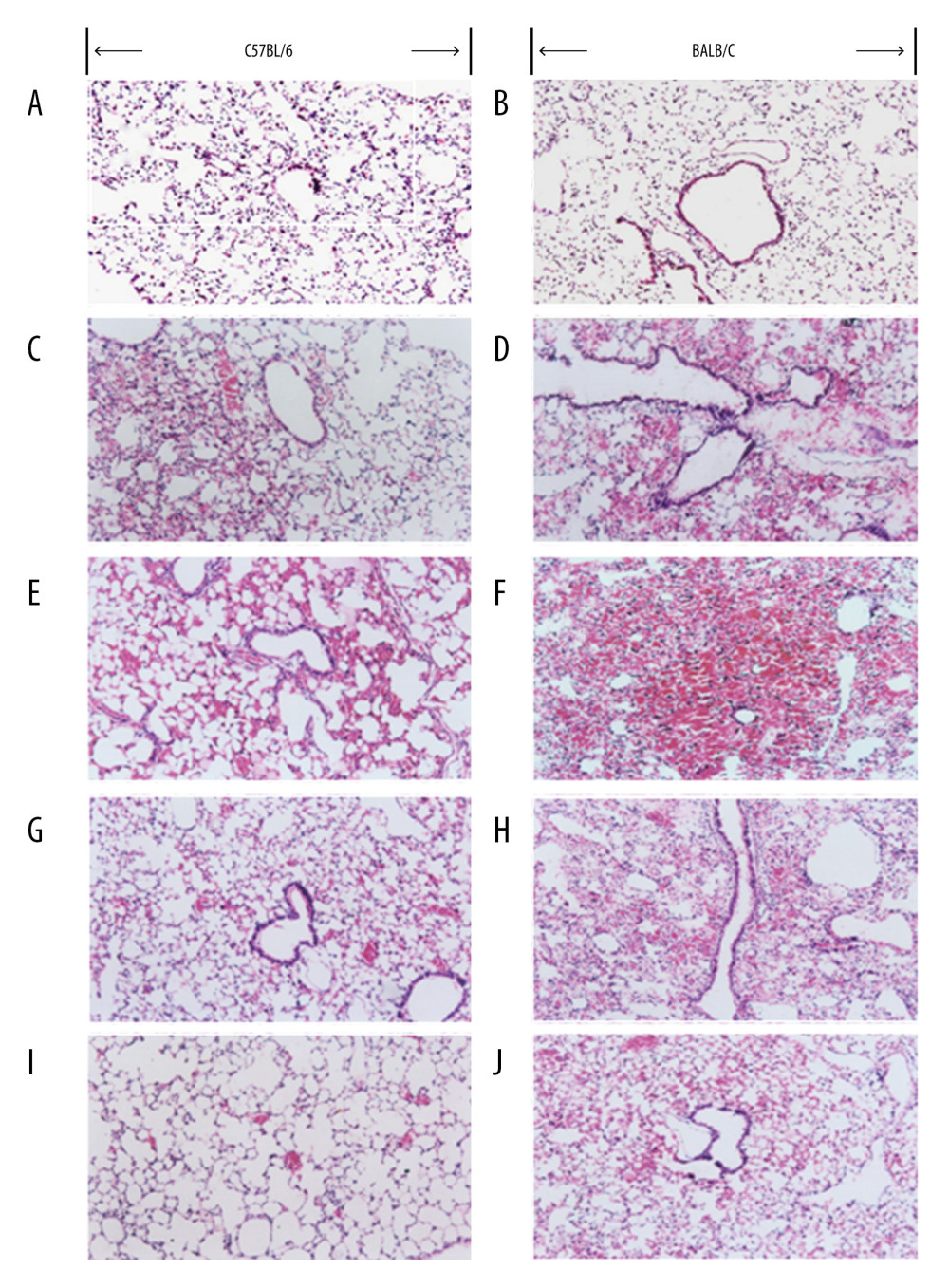 Figure 1. Histopathological changes in C57BL/6 and BALB/C mice before and after systemic blast injury (SBI). Left panels: C57BL/6; right panels: BALB/C. (A, B) before SBI; (C, E, G, I) 1, 6, 24, and 72 h post-SBI, respectively; (D, F, H, J) 1, 6, 24, and 72 h post-SBI respectively. All sections stained with hematoxylin and eosin; magnification, ×100.
Figure 1. Histopathological changes in C57BL/6 and BALB/C mice before and after systemic blast injury (SBI). Left panels: C57BL/6; right panels: BALB/C. (A, B) before SBI; (C, E, G, I) 1, 6, 24, and 72 h post-SBI, respectively; (D, F, H, J) 1, 6, 24, and 72 h post-SBI respectively. All sections stained with hematoxylin and eosin; magnification, ×100. 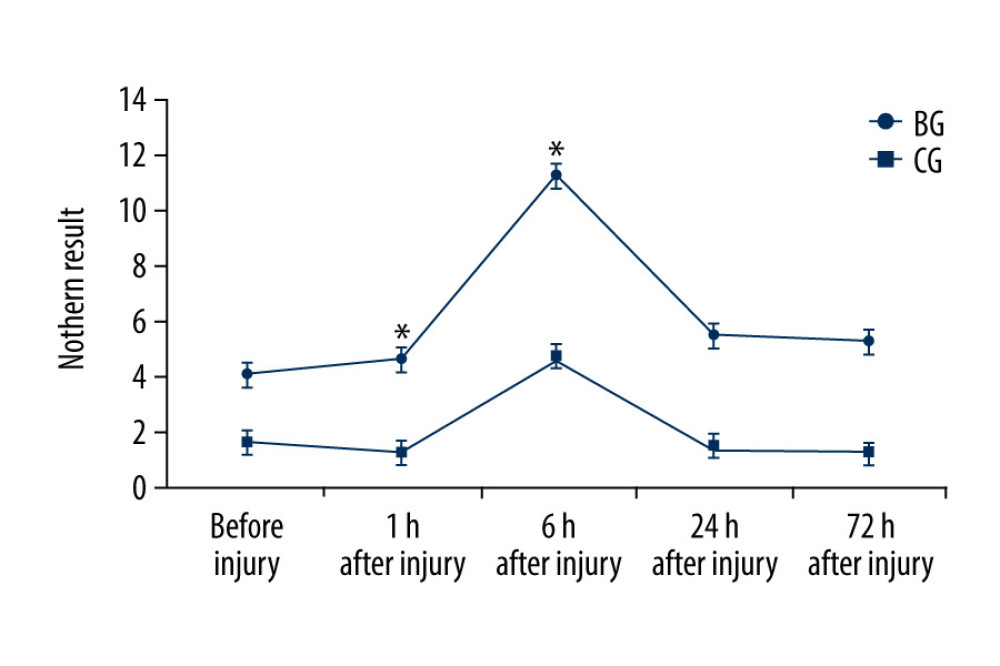 Figure 2. Results of Cxcl15 expression in C57BL/6 and BALB/C after injury (n=3, mean±SD). The abscissa is the time point of observation; the ordinate shows gene expression values for Cxcl15.
Figure 2. Results of Cxcl15 expression in C57BL/6 and BALB/C after injury (n=3, mean±SD). The abscissa is the time point of observation; the ordinate shows gene expression values for Cxcl15. 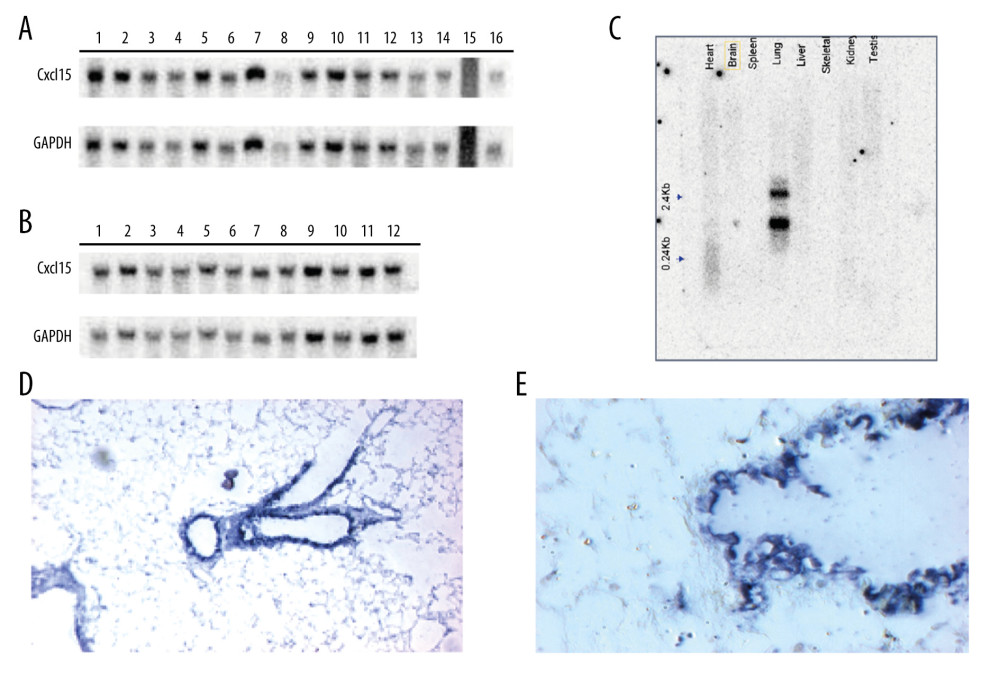 Figure 3. Distribution and expression of Cxcl15. (A) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice before injury, and 1 and 6 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples: 1–4 are samples from mice pre-injury; 5–10 are samples from mice 1 h after injury; and 11–16 are samples from mice 6 h after injury. (B) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice 24 and 72 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples; 1–6 are samples from mice 24 h after injury; 7–12 are samples from mice 72 h after injury. (C) Distribution of Cxcl15 in 8 main tissues in mice. (D) Expression of Cxcl15 in bronchial epithelial cells (magnification, ×100). (E) Expression of Cxcl15 in lung tissue of mice with bronchial epithelial cell cytoplasm (magnification, ×400).
Figure 3. Distribution and expression of Cxcl15. (A) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice before injury, and 1 and 6 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples: 1–4 are samples from mice pre-injury; 5–10 are samples from mice 1 h after injury; and 11–16 are samples from mice 6 h after injury. (B) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice 24 and 72 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples; 1–6 are samples from mice 24 h after injury; 7–12 are samples from mice 72 h after injury. (C) Distribution of Cxcl15 in 8 main tissues in mice. (D) Expression of Cxcl15 in bronchial epithelial cells (magnification, ×100). (E) Expression of Cxcl15 in lung tissue of mice with bronchial epithelial cell cytoplasm (magnification, ×400). Tables
Table 1. Mortality in BALB/C and C57BL/6 mice after blast injury.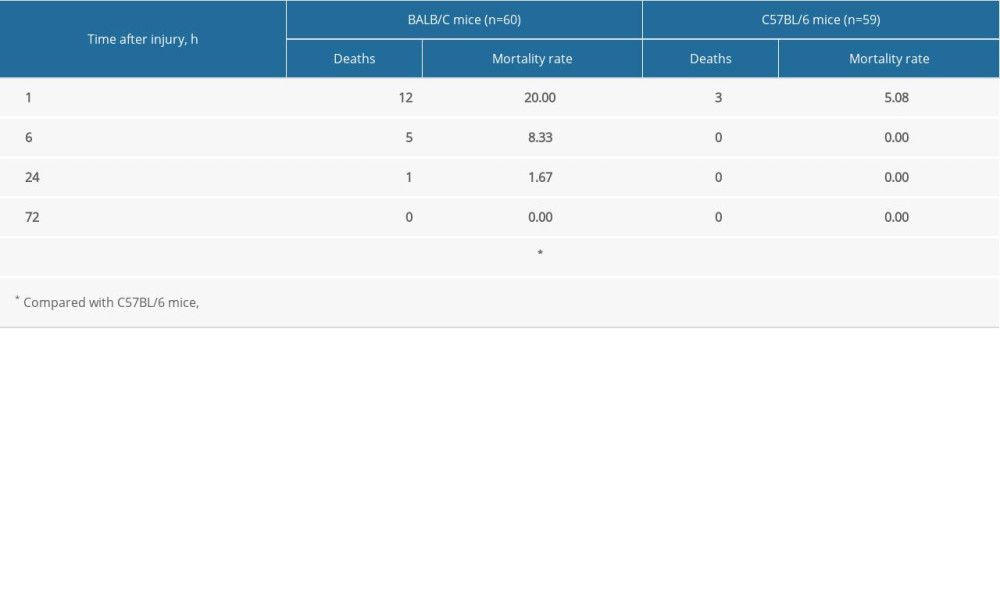 Table 2. Mortality among male and female mice.
Table 2. Mortality among male and female mice.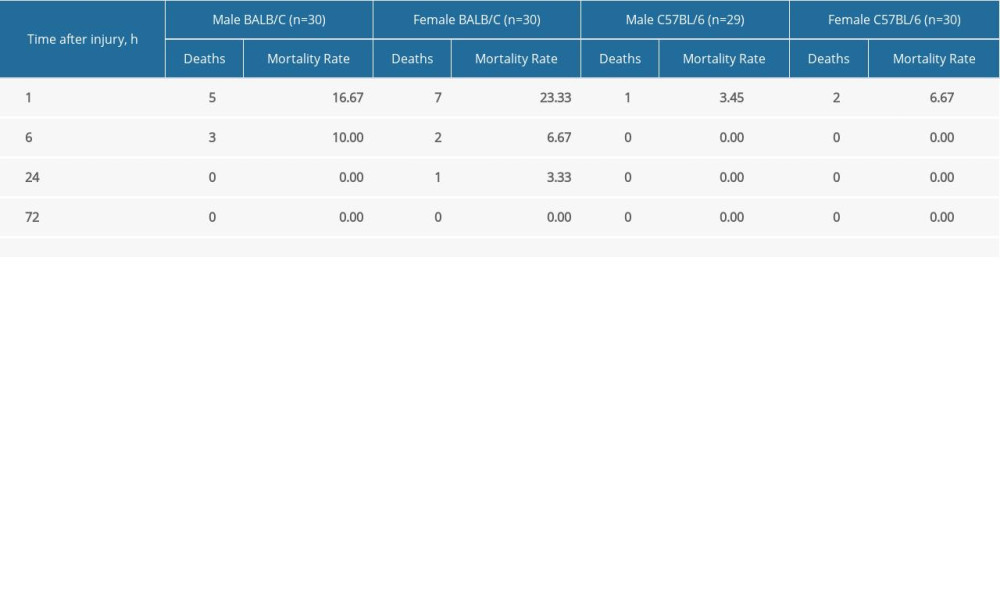 Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD).
Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD).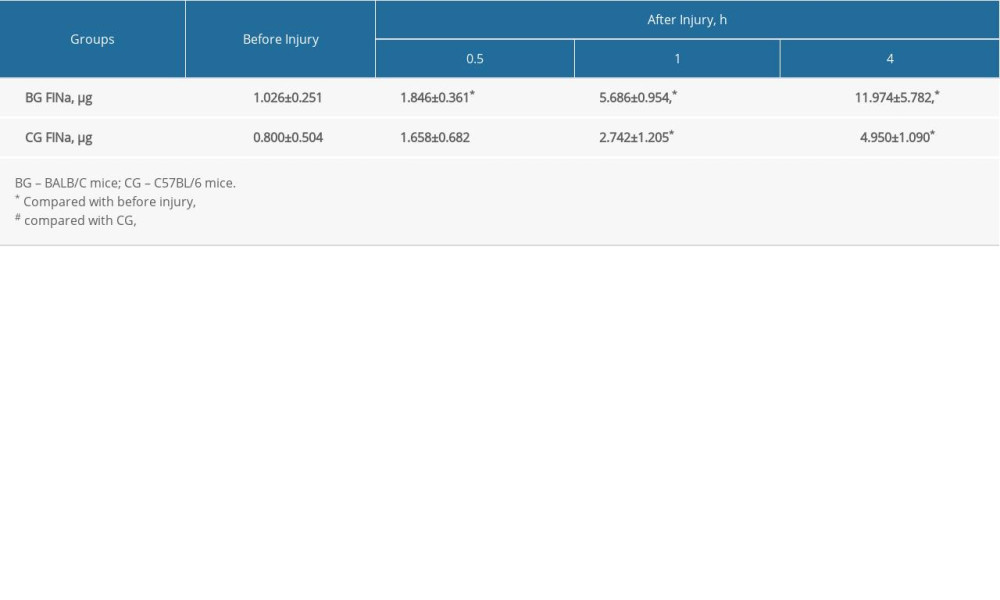 Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation).
Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation).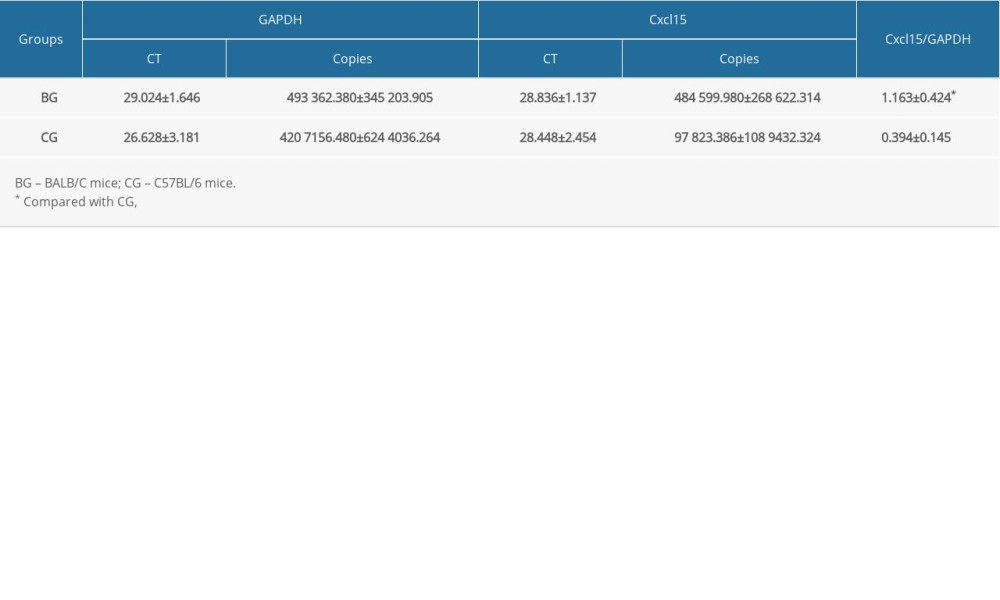 Table 5. Expression of Cxcl15 after trauma measured by northern blot.
Table 5. Expression of Cxcl15 after trauma measured by northern blot.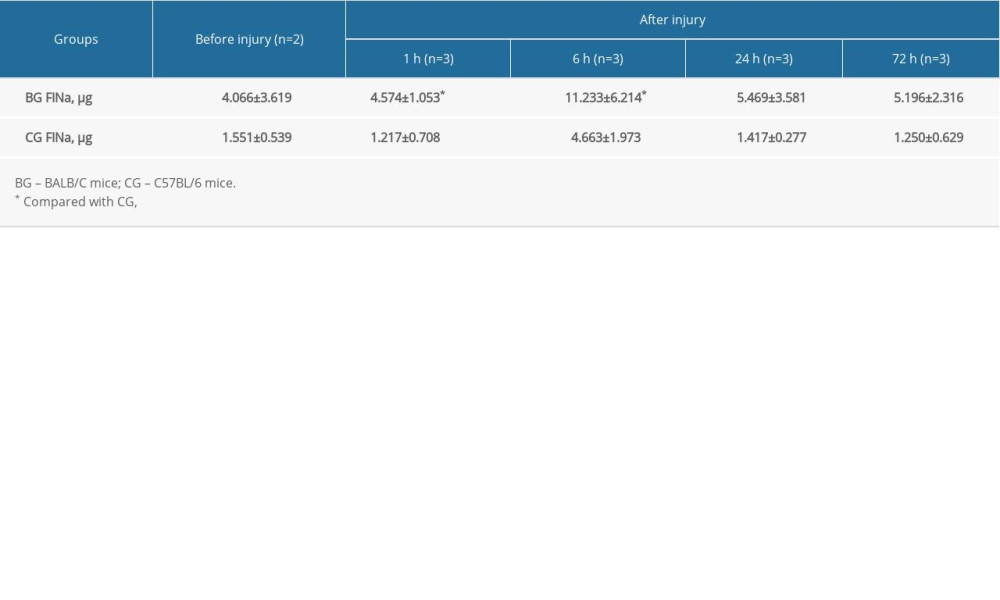
References
1. Radojicić C, Andrić B, Simović M, Genetic basis of resistance to trauma in inbred strains of mice: J Trauma, 1990; 30; 211-13
2. Bartnikowski M, Bartnikowski N, Woloszyk A, Genetic variation in mice affects closed femoral fracture pattern outcomes: Injury, 2019; 50(3); 639-47
3. Li L, Hua L, He Y, Bao Y, Differential effects of formaldehyde exposure on airway inflammation and bronchial hyperresponsiveness in BALB/c and C57BL/6 mice: PLoS One, 2017; 12(6); e0179231
4. Gang F, Li Y, Lixiong Z, Comparative study on the expression of early response genes of trauma of liver BABL/C and C57BL/6 spectra of mouse: Chin J Traumatol, 2002; 12; 13-15
5. Pfeifer R, Andruszkow JH, Busch D, Development of a standardized trauma-related lung injury model: J Surg Res, 2015; 196(2); 388-94
6. Jin D, Li MF, Hou Y: Molecular cloning, 1996; 343-66, Science Press
7. Yan Z, Wang H, Concise guide to molecular biology experiment: Science Press, 1999; 539-85
8. Shi X, Modern medical experimental zoology: The People’s Military Surgeon Press; 2000
9. Inman D, Guth L, Steward O, Genetic influences on secondary degeneration and wound healing following spinal cord injury in various strains of mice: J Comp Neurol, 2002; 451(3); 225-35
10. Wang X, Liao YP, Telesca D, The genetic heterogeneity among different mouse strains impacts the lung injury potential of multiwalled carbon nanotubes: Small, 2017; 13(33); 201700776
11. D’Abbondanza JA, Ai J, Lass E, Robust effects of genetic background on responses to subarachnoid hemorrhage in mice: J Cereb Blood Flow Metab, 2016; 36(11); 1942-54
12. Wang Z, Sun L, Yang Z, Development of serial bio-shock tubes and their application: Chin Med J (Engl), 1998; 111; 109-13
13. Rho SS, Ando K, Fukuhara S, Dynamic regulation of vascular permeability by vascular endothelial cadherin-mediated endothelial cell-cell junctions: J Nippon Med Sch, 2017; 84(4); 148-59
14. Abrams ST, Zhang N, Manson J, Circulating histones are mediators of trauma-associated lung injury: Am J Respir Crit Care Med, 2013; 187(2); 160-69
15. Xu J, Guardado J, Hoffman R, IL33-mediated ILC2 activation and neutrophil IL5 production in the lung response after severe trauma: A reverse translation study from a human cohort to a mouse trauma model: PLoS Med, 2017; 14(7); e1002365
16. Zhu JJ, Canter JA, Rodriguez LL, Arzt J, A novel bovine CXCL15 gene in the GRO chemokine gene cluster: Vet Immunol Immunopathol, 2020; 220; 109990
17. Schmitz JM, McCracken VJ, Dimmitt RA, Lorenz RG, Expression of CXCL15 (Lungkine) in murine gastrointestinal, urogenital, and endocrine organs: J Histochem Cytochem, 2007; 55; 515-24
18. Chen SC, Mehrad B, Deng JC, Impaired pulmonary host defense in mice lacking expression of the CXC chemokine lungkine: J Immunol, 2001; 166; 3362-68
19. Keeley EC, Mehrad B, Strieter RM, CXC chemokines in cancer angiogenesis and metastases: Adv Cancer Res, 2010; 106; 91-111
20. Rivera CG, Bader JS, Popel AS, Angiogenesis-associated crosstalk between collagens, CXC chemokines, and thrombospondin domain-containing proteins: Ann Biomed Eng, 2011; 39(8); 2213-22
21. Leary S, Das P, Ponnalagu D, Genetic strain and sex differences in a hyperoxia-induced mouse model of varying severity of bronchopulmonary dysplasia: Am J Pathol, 2019; 189(5); 999-1014
22. Barone FC, Knudsen DJ, Nelson AH, Mouse strain differences in susceptibility to cerebral ischemia are related to cerebral vascular anatomy: J Cereb Blood Flow Metab, 1993; 13; 683-92
23. Dvorak HF, Vascular permeability to plasma, plasma proteins, and cells: An update: Curr Opin Hematol, 2010; 17; 225-29
24. Chang YS, Kim YK, Jeon SG, Influence of the adjuvants and genetic background on the asthma model using recombinant Der f 2 in mice: Immune Netw, 2013; 13(6); 295-300
Figures
 Figure 1. Histopathological changes in C57BL/6 and BALB/C mice before and after systemic blast injury (SBI). Left panels: C57BL/6; right panels: BALB/C. (A, B) before SBI; (C, E, G, I) 1, 6, 24, and 72 h post-SBI, respectively; (D, F, H, J) 1, 6, 24, and 72 h post-SBI respectively. All sections stained with hematoxylin and eosin; magnification, ×100.
Figure 1. Histopathological changes in C57BL/6 and BALB/C mice before and after systemic blast injury (SBI). Left panels: C57BL/6; right panels: BALB/C. (A, B) before SBI; (C, E, G, I) 1, 6, 24, and 72 h post-SBI, respectively; (D, F, H, J) 1, 6, 24, and 72 h post-SBI respectively. All sections stained with hematoxylin and eosin; magnification, ×100. Figure 2. Results of Cxcl15 expression in C57BL/6 and BALB/C after injury (n=3, mean±SD). The abscissa is the time point of observation; the ordinate shows gene expression values for Cxcl15.
Figure 2. Results of Cxcl15 expression in C57BL/6 and BALB/C after injury (n=3, mean±SD). The abscissa is the time point of observation; the ordinate shows gene expression values for Cxcl15. Figure 3. Distribution and expression of Cxcl15. (A) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice before injury, and 1 and 6 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples: 1–4 are samples from mice pre-injury; 5–10 are samples from mice 1 h after injury; and 11–16 are samples from mice 6 h after injury. (B) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice 24 and 72 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples; 1–6 are samples from mice 24 h after injury; 7–12 are samples from mice 72 h after injury. (C) Distribution of Cxcl15 in 8 main tissues in mice. (D) Expression of Cxcl15 in bronchial epithelial cells (magnification, ×100). (E) Expression of Cxcl15 in lung tissue of mice with bronchial epithelial cell cytoplasm (magnification, ×400).
Figure 3. Distribution and expression of Cxcl15. (A) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice before injury, and 1 and 6 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples: 1–4 are samples from mice pre-injury; 5–10 are samples from mice 1 h after injury; and 11–16 are samples from mice 6 h after injury. (B) Northern blot results of the Cxcl15 gene and GAPDH gene (internal reference) expression in C57BL/6 and BALB/C mice 24 and 72 h after injury. The odd numbers indicate C57BL/6 mouse samples, and the even numbers indicate BALB/C mouse samples; 1–6 are samples from mice 24 h after injury; 7–12 are samples from mice 72 h after injury. (C) Distribution of Cxcl15 in 8 main tissues in mice. (D) Expression of Cxcl15 in bronchial epithelial cells (magnification, ×100). (E) Expression of Cxcl15 in lung tissue of mice with bronchial epithelial cell cytoplasm (magnification, ×400). Tables
 Table 1. Mortality in BALB/C and C57BL/6 mice after blast injury.
Table 1. Mortality in BALB/C and C57BL/6 mice after blast injury. Table 2. Mortality among male and female mice.
Table 2. Mortality among male and female mice. Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD).
Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD). Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation).
Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation). Table 5. Expression of Cxcl15 after trauma measured by northern blot.
Table 5. Expression of Cxcl15 after trauma measured by northern blot. Table 1. Mortality in BALB/C and C57BL/6 mice after blast injury.
Table 1. Mortality in BALB/C and C57BL/6 mice after blast injury. Table 2. Mortality among male and female mice.
Table 2. Mortality among male and female mice. Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD).
Table 3. Fluorescein sodium (FINa) intensity before and after injury (n=5, mean±SD). Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation).
Table 4. Cxcl15 expression under physiological conditions measured by fluorescence quantitative polymerase chain reaction (mean±standard deviation). Table 5. Expression of Cxcl15 after trauma measured by northern blot.
Table 5. Expression of Cxcl15 after trauma measured by northern blot. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








