30 January 2021: Clinical Research
Evaluation of Reduced-Dose Decitabine and Azacitidine for Treating Myelodysplastic Syndromes: A Retrospective Study
Naibo Hu12DE, Tiejun Qin1B, Xiaoyan Du3C, Bingyi Wang3DE, Xiaoyun Wang3E, Zefeng Xu1DF, Lijuan Pan1F, Shiqiang Qu1DF, Zhijian Xiao1A*DOI: 10.12659/MSM.928454
Med Sci Monit 2021; 27:e928454
Abstract
BACKGROUND: Hypomethylating agents (HMA) are considered the first-line therapy for high-risk myelodysplastic syndromes (MDS). However, as the efficacy and safety of rational dosing regimens are lacking, we evaluated the effectiveness and safety of reduced-dose azacitidine (AZA) vs. decitabine (DAC) in adult MDS patients.
MATERIAL AND METHODS: This retrospective study was conducted at the Institute of Hematology & Blood Diseases Hospital, for hospitalized MDS patients diagnosed (WHO 2008 classification criteria) from May 2006 to February 2020. These AZA- and DCA-naive patients treated with AZA 100 mg/(m²·day) for 5 days to 7 days or DAC 20 mg/(m²·day) for 3 days to 4 days, or 20 mg/(m²·day) 1 day/week for 3 weeks/month were assessed for treatment responses and adverse events.
RESULTS: Of the 158 enrolled MDS patients, 120 and 38 patients were administered reduced-dose DAC and AZA, respectively. All the patients received a median of 2 treatment cycles. The overall response rates (ORR) were 50.0% and 73.3% in the AZA and DAC groups, respectively (P=0.007). The percentage of platelet transfusion dependence in the AZA group was lower than the DAC group (P=0.026). The multivariate analysis demonstrated that the DAC treatment was a significant factor for improved responses (OR 2.928; 95% CI 1.267–6.896; P=0.012), and the absolute neutrophil count (ANC) was a predictor of the ORR (OR 0.725; 95% CI 0.558–0.898; P=0.008). Neutropenia (P=0.016) and infection (P=0.032) incidences were higher in the DAC group.
CONCLUSIONS: The reduced-dose DAC group demonstrated a better response than the AZA group in MDS patients with different prognostic risks. The patients’ pre-treatment ANC was a significant factor associated with the ORR.
Keywords: Azacitidine, Dose-Response Relationship, Drug, Myelodysplastic syndromes, decitabine, Drug Tapering
Background
Myelodysplastic syndromes (MDS) are hematologic diseases characterized by ineffective and dysplastic hematopoiesis, which causes cytopenia and can trigger acute leukemia [1]. MDS is divided into lower- (low and intermediate-1) and higher-risk (intermediate-2 and higher) MDS according to the International Prognostic Scoring System (IPSS), and the clinical decisions about treatment are in accordance with these risk classifications [2]. The treatment of lower-risk MDS is aimed at improving cytopenia and providing optimal supportive care [2]. The patients with higher-risk MDS have an increased risk of conversion to acute myeloid leukemia (AML). The primary goal of the higher-risk MDS treatment is to use hypomethylating agents (HMA), chemotherapy, or transplantation to alter the course of the disease [3]. The deoxyribonucleic acid hypomethylating agents (HMA), azacitidine (AZA), and decitabine (DAC) are first-line therapies in the treatment of higher-risk MDS.
Several multicenter phase II or III clinical trials have compared either AZA or DAC with conventional treatments, including the best supportive care, and both drugs showed a significant overall survival benefit in patients with MDS [4–6]. Several retrospective studies compared the effectiveness and safety of AZA or DAC. However, all those reports focused on the patients on normal doses of AZA or DAC treatment [7–9]. Because the normal-dose HMA result in adverse effects, many MDS patients receive reduced-dose HMA treatment. Moreover, only a proportion of patients who can tolerate the standard-dose demethylation therapy can achieve favorable outcomes, and several cycles are needed before the efficacy of this therapy can be observed. Therefore, it is essential to discover the parameters that can determine whether a patient will respond to the HMA, avoid adverse effects, and not require alternative treatments. Currently, there is a lack of data on the effectiveness and safety of reduced-dose AZA
Therefore, the present study retrospectively assessed the effectiveness and safety of reduced-dose AZA and reduced-dose DAC in treating MDS patients, and investigated the factors influencing patients’ responses to the HMA.
Material and Methods
PATIENTS:
The MDS patients at the Institute of Hematology & Blood Diseases Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College (Tianjin, China) who received HMA (DAC or AZA) for the first time between May 2006 and February 2020 were retrospectively reviewed. All the patients diagnosed with MDS (aged ≥18 years) who were unable to tolerate the standard dose of DAC of 20 mg/(m2·day) (body surface area-based dosing) for 5 days every cycle (4 weeks=1 cycle) or AZA 75 mg/(m2·day) for 7 days every cycle due to age, physical condition, or complications were included in this study. These patients are typically >70 years old, have severe long-term comorbidities or uncontrolled infections before treatment, indicating intolerance and the refusal of standard-dose chemotherapy. The exclusion criteria were patients who had already developed AML, or MDS accompanied by lymphoma, myeloma, or other blood diseases; received any previous chemotherapy (including aclariamycin, cytarabine, daunorubicin, idarubicin, fludarabine, topotecan), and hematopoietic stem-cell transplantation; previously been treated with HMA, including patients treated with other dosages of HMA, or treated with a combination of HMA (DAC and AZA); used any drugs, which could improve the blood indicator results including androgen, erythropoietin, or thalidomide.
The electronic health records of the eligible patients were screened and extracted from a cooperative medical intelligence platform (Yidu Cloud Technology Co., Ltd., Beijing, China). This study was approved by the Institute of Hematology & Blood Diseases Hospital Ethics Committee, and exempted from obtaining informed consent by the patients as this was a retrospective study. The patients were evaluated using the IPSS and divided into 2 categories, the lower-risk (low and intermediate-1) and higher-risk (intermediate-2 and higher) groups, and analyzed using the Revised IPSS (IPSS-R) at the beginning of the hypomethylating therapy. The mutational gene targets were examined using high-throughput sequencing (Illumina, San Diego, CA). The potential gene mutations were determined according to the Catalogue of Somatic Mutations in Cancer.
TREATMENT:
The enrolled MDS patients who could not tolerate the standard-dose HMA were treated with reduced-dose DAC before AZA was available in China. Although AZA is recommended for these patients, AZA was registered and marketed in China only in 2018, and has a less-potent myelosuppressive effect. The exceptions were patients who could not tolerate subcutaneous AZA injections, explicitly refused AZA treatment because of the distrust of a newly marketed drug, and still used DAC for sequential therapy.
The DAC was administered intravenously, 20 mg/(m2·day) for 3 days to 4 days or 20 mg/(m2·day) for 1 day/week for 3 weeks/month including a 1-week break. The AZA was administered subcutaneously at a fixed dose of 100 mg daily for 5 days to 7 days, and the treatment schedule was repeated every 4 weeks to 6 weeks. It was recommended that patients be treated with a reduced-dose HMA continuously until the patient developed intolerance and was unable to continue this therapy. None of the patients had more than 4 cycles owing to their physical condition, comorbidities, financial reasons, and personal attitudes or beliefs. Four cycles of DAC and AZA treatment are generally recommended in clinical practice. Therefore, the treatments were terminated or continued based on the individual’s physical conditions. The first treatment evaluation was performed at the end of the HMA treatment cycle 1 or 2. Transfusion dependence was defined as the need for more than 4 units of red blood cells (RBCs) or 16 units of platelets (PLT) within 8 weeks before treatment. The study population consisted of 2 groups, 1 group accepted the reduced-dose DAC treatment (DAC group) and the other group accepted the reduced-dose AZA treatment (AZA group).
ASSESSMENT OF EFFICACY AND SAFETY:
The response to the treatment was evaluated in patients who received at least 1 cycle of HMA according to the revised International Working Group (IWG) 2006 criteria [10]. The overall response rate (ORR) includes complete remission (CR), partial remission (PR), marrow CR (mCR), and hematologic improvement (HI). Among them, HI was defined as satisfying at least 1 of the following responses: erythroid response (HI-E), PLT response (HI-P), or neutrophil response (HI-N). The hematological response time or improvement was assessed according to the IWG 2000 criteria for MDS [11]. Adverse events were evaluated according to the Common Terminology Criteria for Adverse Events (4.0) [12].
STATISTICAL ANALYSIS:
All the statistical analysis was conducted using the R Project for statistical computing (R version 3.6.3, Holding the Windsock, CRAN). The continuous variables were presented as mean±standard deviation (SD) or median (interquartile range [(IQR]) depending on whether the data conformed to normal distribution. The categorical variables were shown as counts or percentages. The Mann-Whitney U test was used to compare the intergroup continuous variables; and Fisher’s exact test or the Chi-square test was used to compare the categorical variables between the 2 groups.
The data to support the findings of this study are available on request from the corresponding author.
Results
BASELINE PATIENT CHARACTERISTICS:
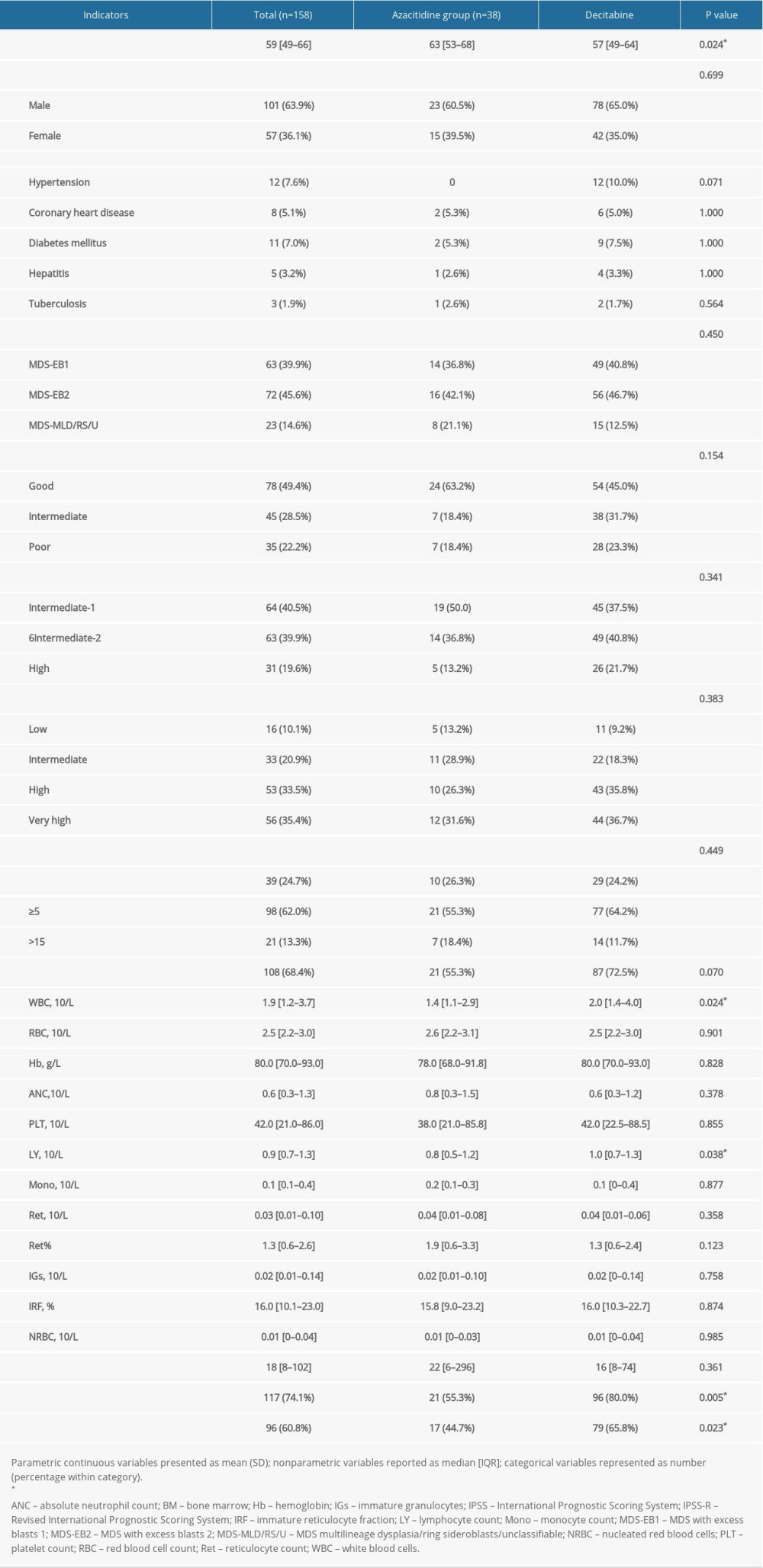
A total of 158 patients were screened after the DAC or AZA treatment during the study period. Thirty-eight (24.0%) patients received the reduced-dose AZA treatment, and 120 patients (75.9%) received the reduced-dose DAC treatment (Table 1). The median age of the AZA group (63 years, interquartile range [IQR] 53–68) was higher than the DAC group (57 years, IQR 49–64) (P=0.024). The proportion of patients with the MDS subtype, MDS with excess blasts 1 (MDS-EB1) (63/158, 39.9%) was higher than the proportion of patients with MDS subtype, MDS with excess blasts 2 (MDS-EB2) (72/158, 45.6%). Their cytogenetic results were divided into 3 groups: the good karyotype (78 patients, 49.4%), the intermediate karyotype (45 patients, 28.5%), and the poor karyotype (35 patients, 22.2%), based on the IPSS classification. These results were classified into low- (16 patients, 10.1%), intermediate- (33 patients, 20.9%), high- (53 patients, 33.5%), and very high-risk (56 patients, 35.4%) MDS patients, based on the IPSS-R classification. The number of patients from the 2 groups who received transfusion during the treatment were significantly different between the AZA and DAC groups (P=0.005). The baseline characteristics were well-balanced between the 2 groups (Table 1).
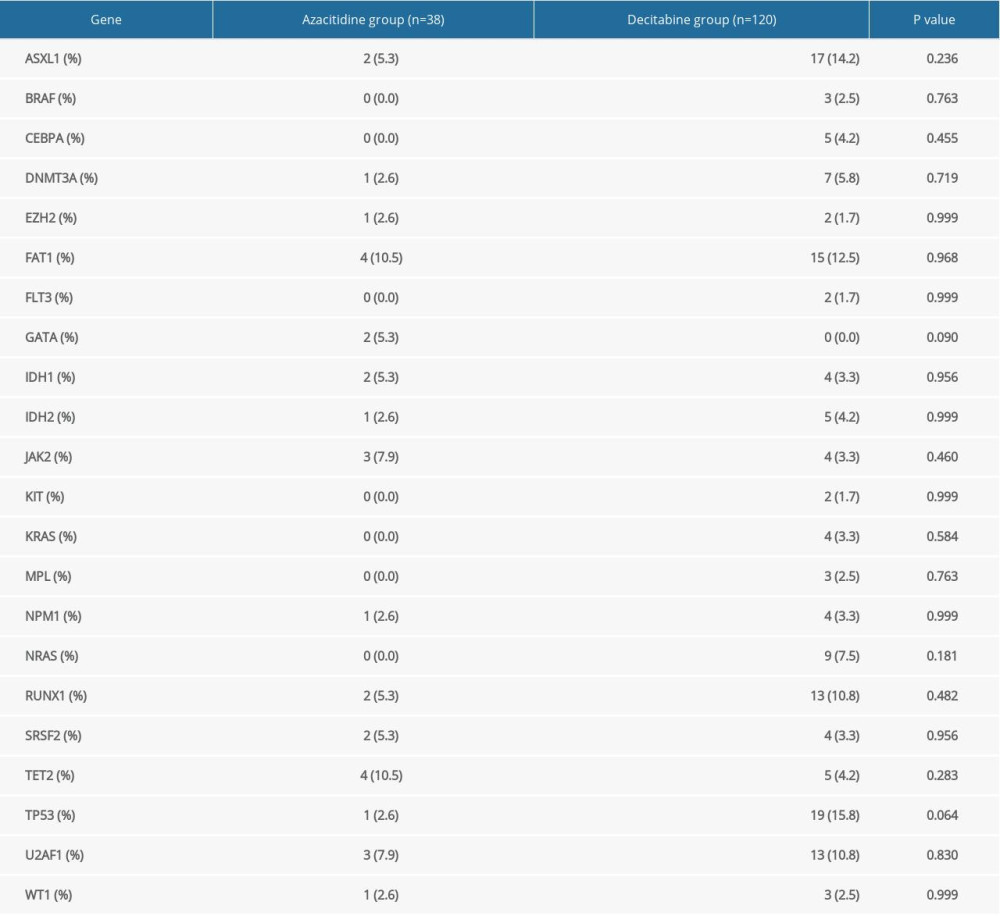
The molecular description of MDS will provide a better understanding, classification, and treatment of this disease as MDS are a group of clonal hematopoietic disorders marked by ineffective hematopoiesis and peripheral cytopenia. Any disruption of the hematopoietic progenitor proliferation and natural differentiation into mature myeloid elements has the potential to lead to ineffective hematopoiesis and an MDS phenotype. The observed gene mutations are detailed in Table 2. Eighty of the 158 patients (50.6%) presented with at least 1 mutation. The 5 most common mutations were TP53, ASXL1, FAT1, U2AF1, and RUNX1. Less commonly mutated genes included NRAS, TET2, DNMT3A, JAK2, IDH1, IDH2, SRSF2, CEBPA, NPM1, KRAS, WT1, BRAF, EZH2, MPL, FLT3, GATA, KIT. No significant differences were observed in the mutated genes between the 2 treatment arms.
TREATMENT RESPONSE:
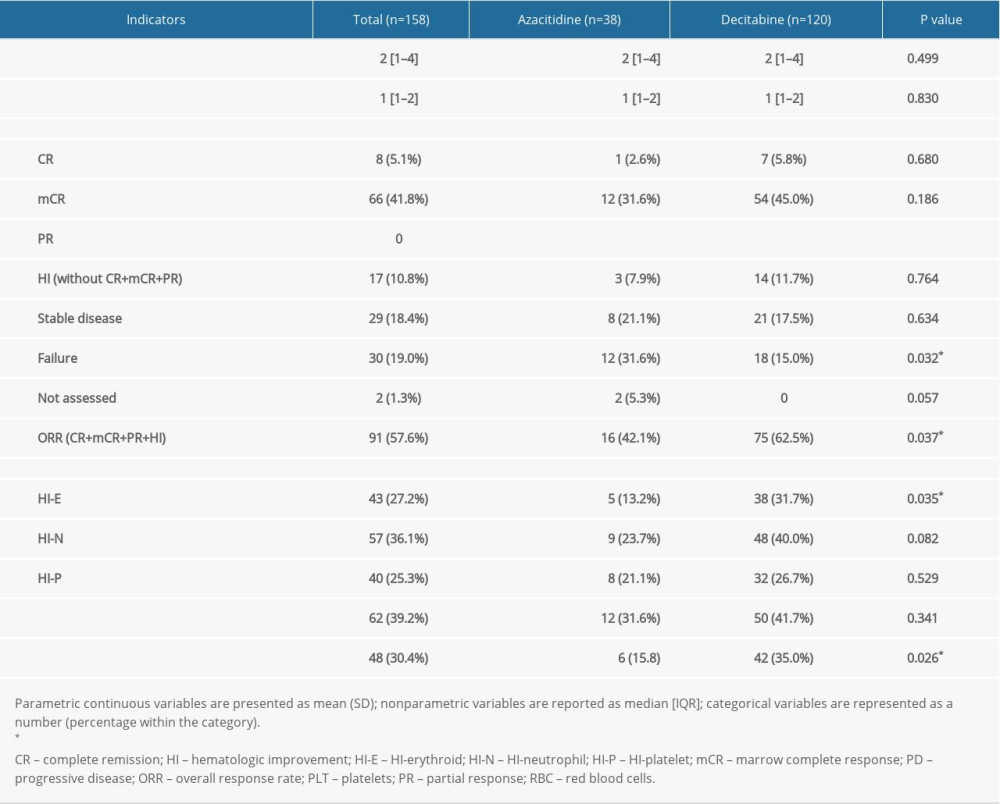
The patients in both groups received 2 (IQR 1–4) treatment cycles, with 1 cycle (IQR 1–2) as the median for response assessment (Table 3). With respect to the treatment responses to AZA and DAC, the best rate of response was CR in 8 (5.1%) patients, mCR in 66 (41.8%) patients, and stable disease (sd) in 20 (12.7%) patients. Twenty-nine patients (18.4%) received <2 cycles of HMA without any documented response or progression. The ORR in the DAC group was higher (75 patients, 62.5%) than the AZA group (16 patients, 42.1%) (P=0.007). Among the patients’ HI results, HI-E was observed in 43 (27.2%) patients, HI-N in 57 (36.1%) patients, and HI-P in 40 (25.3%) patients. The PLT transfusion independence was 15.8% (n=6) and 35.0% (n=42) in patients treated with AZA and DAC, respectively (P=0.026).
SAFETY:
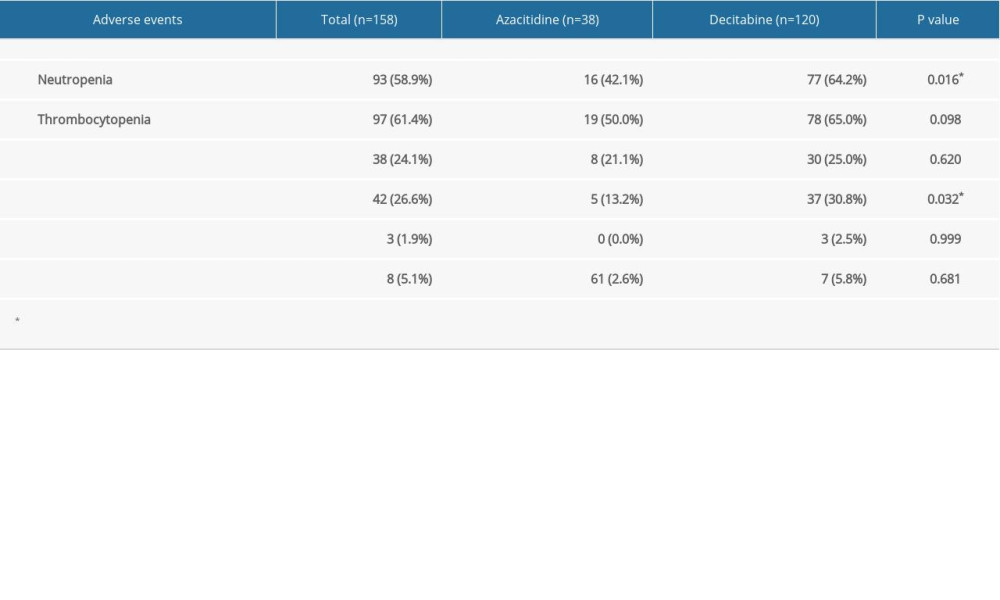
The incidence of adverse events after treatment with reduced-dose AZA and reduced-dose DAC regimens are shown in Table 4. The most common hematological adverse events (grade III–IV) were neutropenia (93 patients, 58.9%) and thrombocytopenia (97 patients, 61.4%). The treatment-related adverse events included infections (42 patients, 26.6%), hemorrhage (3 patients, 1.9%), and hepatic injury (8 patients, 5.1%). Statistically significant differences in the incidences of neutropenia (P=0.016) and infections (P=0.032) were observed between the 2 treatment groups.
PREDICTIVE FACTORS FOR THE OVERALL RESPONSE RATES IN REDUCED-DOSE AZA OR DAC TREATMENT:
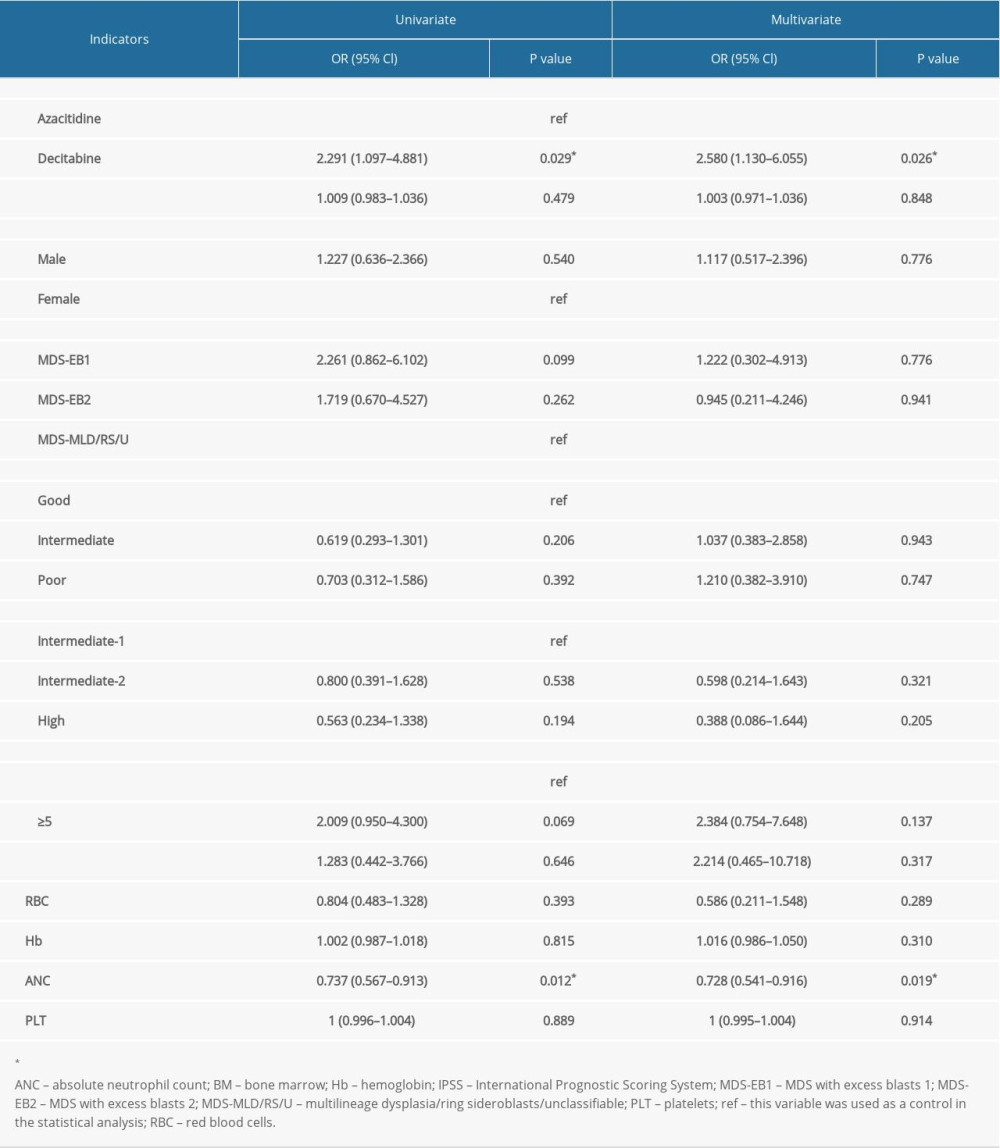
All the variables that could have an effect on the ORR to specific HMA were included in a univariate logistic regression analysis (Table 5). The DAC treatment (odds ratio [OR] 2.750; 95% Cl 1.294–5.885; P=0.008) showed a higher ORR. The multivariable regression analysis demonstrated the DAC treatment was a significant influential factor for a better response (OR 2.928; 95% Cl 1.267–6.896; P=0.012), whereas the ANC (OR 0.725; 95% Cl 0.558–0.898; P=0.008) was a significant predictor of the ORR.
Discussion
HMA are considered to be epigenetic drugs that can alter the natural course of the disease, and have been widely used in the treatment of MDS. However, attempts to explore the optimum therapeutic regimen to achieve better efficacy and maintenance time based on the drug dosage and treatment cycles remain the most concerning issues. Our results confirmed that adults with MDS treated with the reduced-dose DAC regimen (20 mg/[m2·day] for 3 days to 4 days or 20 mg/[m2·day] 1 day/week for 3 weeks/month including a 1-week break) had higher ORR and HI rates than those treated with the reduced-dose AZA regimen (100 mg for 5 days to 7 days). We identified that the DAC therapy and lower ANC were independent predictors for an improved ORR.
Currently, a grouped-therapy strategy is the international consensus for treating MDS. HMA administered to low-risk MDS patients improved their cytopenia. However, these patients do not have longer survival periods than the high-risk MDS patients [13,14]. Although the standard-dose regimen of HMA is efficient, it can cause myelosuppression, and cannot be prescribed when contraindicated in the elderly population due to its adverse effects [15,16]. Moreover, the genetic characteristics of patients affect the clinical phenotype, prognosis, and response to treatment. The mutations in TP53, ASXL1, TET2, RUNX1, and JAK2 are particularly common in treatment-related MDS [17]. Therefore, considering the real-world patients of varying ages and risk stratifications of the disease, patient characteristics, their varied treatment options and evaluation criterion, have inevitable heterogeneity. Neither the IPSS categories nor the mutations were statistically different between the 2 treatment arms.
The reduced-dose HMA treatment was effective in this study, with more than half the patients achieving an ORR (91 patients, 57.6%). DAC at reduced-doses can achieve epigenetic modification by targeting the inhibition of DNMT1, a protein-coding gene [18]. Some studies demonstrated that a reduced-dose DAC regimen has definite efficacy with reduced incidences of adverse reactions, including bone marrow suppression [19,20]. Our results suggest that the ORR of patients with MDS to the reduced-dose DAC treatment was approximately 62.5%, and the CR rate was 5.8%. Previous studies of reduced-dose DAC regimens have revealed similar ORR (range 44.0% to 68.0%), although those studies included smaller sample sizes [21]. A significant ORR advantage was not observed in the DAC group with a standard regimen (about 70%) compared to the reduced-dose DAC patients [22,23]. This could be related to the heterogeneity of patients.
As a commonly used HMA for MDS treatment, AZA is effective and safe for patients; the ORR to AZA ranged from 30% to 60% in previous studies. A study of 3 dosage regimens (all 28-day cycles) in patients with MDS suggested that the ORR to AZA was 39.4% (75 mg/[m2·day] for 5 days), 67.9% (75 m4m2·day] for 7 days including a 2-day break), and 51.3% (75 mg/[m2·day] for 7 days), respectively [24]. The ORR was 45.9% in patients who had lower-risk MDS treated with AZA4[25]. According to the Southwestern Oncology Group (SWOG) S1117, the ORR was 38% (CR rate: 24%; mCR rate: 12%, and HI rate: 14%) for the high-risk group receiving AZA alone [26]. Our results showed the ORR to the reduced-dose AZA treatment in adult patients with MDS in different risk stratifications was 42.1%. The rate of response was 2.6, 31.6, 7.9, and 21.1% for CR, mCR, sd with HI, and sd without HI, respectively. In our comparison of the reduced-dose strategies of DAC and AZA, the DAC group achieved higher treatment responses (CR: 5.8%
Myelosuppression and infections after the HMA treatments are significant safety concerns. The need for prophylactic use of antibiotics in HMA treatment-associated infections is currently inconclusive. Our results are consistent with Xu et al. [28], who reported that the incidence of hematological adverse events (grade III–IV) is higher in the DAC-treated patients. In our study, the HMA treatment-associated infections occurred in 13.2% and 30.8% of AZA- and DAC-treated patients, respectively. Results from a Korean study [29] suggest that ≥grade III neutropenia was more common in the DAC-treated patients, and the incidence of infections was significantly lower in the AZA group. Although the HMA used (DAC) was related to an increased risk of infection, there was no difference in the mortality induced by the infections. The reduced-dose HMA regimens were relatively safer in these patients.
Our results are consistent with another study [30], which found a significant ORR improvement in the DAC regimen over the AZA regimen, and analyzed the prognostic response factors. The ANC was the only identified prognostic factor associated with the ORR to HMA. The white blood cell count (WBC) and ANC generally decrease synchronously during myelosuppression. A significantly inhibited effect of the bone marrow’s hematopoietic function was seen in the ANC, and it is easy to acquire severe infections and other complications [31]. It suggests that early ANC detection may be an important predictor of the clinical response in patients with MDS. A study by Bao et al. showed that the complex karyotype, gene mutations, and doubling of the PLT count after 1 treatment cycle were the independent prognostic factors for predicting CR after the DAC treatment [32]. The different genetic variants lead to varied outcomes; however, the mechanism remains unknown.
Our study has several limitations. First, the study was based on a retrospective design although any evidence is vital for validation in future prospective cohort studies. Second, the imbalance in the number of MDS patients between the 2 treatment groups was due to the late launch of AZA in China. Third, the follow-up results of all the patients receiving DAC and AZA treatments were not accessible; therefore, we did not take the survival analysis into account. Currently, a prospective multicenter study organized by our team is ongoing.
Conclusions
In conclusion, our results show that DAC treatment with a reduced-dose regimen (20 mg/[m2·day] for 3 days to 4 days or 20 mg/[m2·day] 1 day/week for 3 weeks/month including a 1-week break) could have a better response than reduced-dose AZA therapy (100 mg for 5 days to 7 days) in patients with MDS with varied prognostic risks. The early ANC of patients was a significant factor associated with the ORR. Our study provides reliable clinical evidence for understanding the effectiveness of low-dosage HMA regimens in MDS patients. Although the number of patients in the 2 groups is unbalanced due to the different times of the availability of DAC and AZA in China, it reflects the Chinese reality of MDS treatment with reduced-dose HMA in the real world. Further prospective studies to explore the optimal therapeutic dose strategy with large-scale sample sizes are warranted.
References
1. Tefferi A, Vardiman JW, Myelodysplastic syndromes: N Engl J Med, 2009; 361; 1872-85
2. Fenaux P, Adès L, How we treat lower-risk myelodysplastic syndromes: Blood, 2013; 121; 4280-86
3. Bejar R, Tiu RV, Sekeres MA, Komrokji RS, Myelodysplastic syndromes: Recent advancements in risk stratification and unmet therapeutic challenges: Am Soc Clin Oncol Educ Book, 2013; 33; 256-70
4. Du X, Lai YY, Xiao ZJ, Efficacy, safety and pharmacokinetics of subcutaneous azacitidine in Chinese patients with higher risk myelodysplastic syndromes: Results from a multicenter, single-arm, open-label phase 2 study: Asia Pac J Clin Oncol, 2018; 14; 270-78
5. Fenaux P, Mufti GJ, Hellstrom-Lindberg E, Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: A randomised, open-label, phase III study: Lancet Oncol, 2009; 10; 223-32
6. Kantarjian H, Issa JP, Rosenfeld CS, Decitabine improves patient outcomes in myelodysplastic syndromes: Results of a phase III randomized study: Cancer, 2006; 106; 1794-803
7. Zeidan AM, Sekeres MA, Garcia-Manero G, Comparison of risk stratification tools in predicting outcomes of patients with higher-risk myelodysplastic syndromes treated with azanucleosides: Leukemia, 2016; 30; 649-57
8. Jabbour E, Garcia-Manero G, Ravandi F, Prognostic factors associated with disease progression and overall survival in patients with myelodysplastic syndromes treated with decitabine: Clin Lymphoma Myeloma Leuk, 2013; 13; 131-38
9. Ritchie EK, Safety and efficacy of azacitidine in the treatment of elderly patients with myelodysplastic syndrome: Clin Interv Aging, 2012; 7; 165-73
10. Cheson BD, Greenberg PL, Bennett JM, Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia: Blood, 2006; 108; 419-25
11. Cheson BD, Bennett JM, Kantarjian H, Report of an international working group to standardize response criteria for myelodysplastic syndromes: Blood, 2000; 96; 3671-74
12. National Cancer Institute (US), Common Terminology Criteria for Adverse Events (CTCAE v. 4.0): Cancer Therapy Evaluation Program, 2009 https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm
13. Garcia-Manero G, Myelodysplastic syndromes: 2015 Update on diagnosis, risk-stratification and management: Am J Hematol, 2015; 90; 831-41
14. Lee JH, Kim YJ, Sohn KS, Benefits of hypomethylating therapy in IPSS lower-risk myelodysplastic syndrome patients: A retrospective multicenter case series study: Leuk Res, 2017; 60; 135-44
15. Lee JH, Jang JH, Park J, A prospective multicenter observational study of decitabine treatment in Korean patients with myelodysplastic syndrome: Haematologica, 2011; 96; 1441-47
16. Wang R, Gross CP, Frick K, The impact of hypomethylating agents on the cost of care and survival of elderly patients with myelodysplastic syndromes: Leuk Res, 2012; 36; 1370-75
17. Shih AH, Levine RL, Molecular biology of myelodysplastic syndromes: Semin Oncol, 2011; 38; 613-20
18. Saunthararajah Y, Sekeres M, Advani A, Evaluation of noncytotoxic DNMT1-depleting therapy in patients with myelodysplastic syndromes: J Clin Invest, 2015; 125; 1043-55
19. Garcia-Manero G, Jabbour E, Borthakur G, Randomized open-label phase II study of decitabine in patients with low- or intermediate-risk myelodysplastic syndromes: J Clin Oncol, 2013; 31; 2548-53
20. Li X, Song Q, Chen Y, Decitabine of reduced dosage in Chinese patients with myelodysplastic syndrome: A retrospective analysis: PLoS One, 2014; 9; e95473
21. Baozhi F, Zhenzhen L, Guangsheng HLower risk myelodysplastic syndrome patients with transfusion dependent treated by dose-reduced decitabine: Zhonghua Yi Xue Za Zhi, 2013; 93; 3189-92 [in Chinese]
22. Jeong SH, Kim YJ, Lee JH, A prospective, multicenter, observational study of long-term decitabine treatment in patients with myelodysplastic syndrome: Oncotarget, 2015; 6; 44985-94
23. Lee JH, Jang JH, Park J, A prospective multicenter observational study of decitabine treatment in Korean patients with myelodysplastic syndrome: Haematologica, 2011; 96; 1441-47
24. Garcia-Delgado R, Miguel DD, Bailen A, Effectiveness and safety of different azacitidine dosage regimens in patients with myelodysplastic syndromes or acute myeloid leukemia: Leuk Res, 2014; 38; 744-50
25. Musto P, Maurillo L, Spagnoli A, Azacitidine for the treatment of lower risk myelodysplastic syndromes: A retrospective study of 74 patients enrolled in an Italian named patient program: Cancer, 2010; 116; 1485-94
26. Sekeres MA, Othus M, List AF, Randomized phase II study of azacitidine alone or in combination with lenalidomide or with vorinostat in higher-risk myelodysplastic syndromes and chronic myelomonocytic leukemia: North American Intergroup Study SWOG S1117: J Clin Oncol, 2017; 35; 2745-53
27. Jabbour E, Short NJ, Montalban-Bravo G: Blood, 2017; 130; 1514-22
28. Xu R, Chen X, Deng C, A retrospective study comparing azacitidine with decitabine in Chinese patients with refractory anemia with excess blast based on two clinical trials in a single center: Am J Transl Res, 2019; 11; 4533-41
29. Lee YG, Kim I, Yoon SS, Comparative analysis between azacitidine and decitabine for the treatment of myelodysplastic syndromes: Br J Haematol, 2013; 161; 339-47
30. Lee BH, Kang KW, Jeon MJ, Comparison between 5-day decitabine and 7-day azacitidine for lower-risk myelodysplastic syndromes with poor prognostic features: A retrospective multicentre cohort study: Sci Rep, 2020; 10; 39
31. Li B, Xu ZF, Cui RClinical features and survival analysis in primary myelodysplastic syndromes patients with immunological abnormalities: Zhonghua Xue Ye Xue Za Zhi, 2012; 33; 527-31 [in Chinese]
32. Bao ZH, Zhao HG, Yu HEClinical efficacy and prognostic factors of decitabine for treatment of myelodysplastic syndrome: Zhongguo Shi Yan Xue Ye Xue Za Zhi, 2018; 26; 1702-7 [in Chinese]
In Press
12 Mar 2024 : Clinical Research
Metabolomic Alterations in Methotrexate Treatment of Moderate-to-Severe PsoriasisMed Sci Monit In Press; DOI: 10.12659/MSM.943360
14 Mar 2024 : Clinical Research
Renal Dysfunction Increases Risk of Adverse Cardiovascular Events in 5-Year Follow-Up Study of Intermediate...Med Sci Monit In Press; DOI: 10.12659/MSM.943956
15 Mar 2024 : Clinical Research
Impact of One-Lung Ventilation on Oxygenation and Ventilation Time in Thoracoscopic Heart Surgery: A Compar...Med Sci Monit In Press; DOI: 10.12659/MSM.943089
14 Mar 2024 : Clinical Research
Differential DHA and EPA Levels in Women with Preterm and Term Births: A Tertiary Hospital Study in IndonesiaMed Sci Monit In Press; DOI: 10.12659/MSM.943895
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952