29 May 2021: Lab/In Vitro Research
Silencing Ribosomal Protein L22 Promotes Proliferation and Migration, and Inhibits Apoptosis of Gastric Cancer Cells by Regulating the Murine Double Minute 2-Protein 53 (MDM2-p53) Signaling Pathway
Zhenqing Sun1ABCE, Zhigang Qiu1ABCE, Zhengkun Wang1DE, Honghui Chi2DE, Peipei Shan3CE*DOI: 10.12659/MSM.928375
Med Sci Monit 2021; 27:e928375
Abstract
BACKGROUND: The aim of this study was to investigate the effect of ribosomal protein L22 (RPL22) on gastric cancer (GC) cell proliferation, migration, and apoptosis, and its correlation with the murine double minute 2-protein 53 (MDM2-p53) signaling pathway.
MATERIAL AND METHODS: The RPL22 expression in GC tissues and cells was detected by quantitative reverse transcription-polymerase chain reaction and western blotting. RPL22 was overexpressed in the MKN-45 cells by the transfection of a vector, pcDNA3.1 (pcDNA)-RPL22, whereas it was silenced in the MGC-803 cells by the transfection of short interfering (si) RNA (si-RPL22). Flow cytometric analysis, cell viability assays, wound healing assays, and transwell assays were utilized to explore the influences of RPL22 on the apoptosis, proliferation, migration, and invasion. Nutlin-3 (an MDM2-p53 inhibitor) was used to inhibit MDM2-p53 signaling.
RESULTS: The RPL22 expression was downregulated in GC tissues and cells. It was significantly lower in the advanced GC tissues than in the early GC tissues, and was significantly lower in the lymphatic metastatic tissues than in the non-lymphatic metastatic tissues. The transfection of si-RPL22 accelerated the ability of GC cells to proliferate and metastasize, whereas apoptosis was dampened. The transfection of pcDNA-RPL22 exerted the opposite effect on the GC cells; MDM2 expression was upregulated in RPL22-silenced GC cells, while the expression of p53 was downregulated. In vitro, treatment with nutlin-3 reversed the promoting effects of si-RPL22 on GC progression.
CONCLUSIONS: In vitro, the silencing of RPL22 aggravates GC by regulating the MDM2-p53 signaling pathway.
Keywords: Apoptosis Inducing Factor, Apoptosis Regulatory Proteins, carcinogenesis, Gene Silencing, Piperazines, Proto-Oncogene Proteins c-mdm2, RNA, Small Interfering, RNA-Binding Proteins, Ribosomal Proteins, Tumor Cells, Cultured, Tumor Suppressor Protein p53
Background
Gastric cancer (GC), a gastrointestinal malignant tumor, has serious consequences on a patient’s life and health [1]. GC is asymptomatic in the early stages, and diagnosed at an advanced stage in the majority of patients [2]. It has high mortality and successful therapeutic strategies are limited [3]. At present, surgery is considered the most effective and radical treatment for GC [4], and despite improvements in the surgical techniques and other treatments, the prognosis for advanced GC remains poor [5]. Therefore, exploring the mechanisms underlying GC tumorigenesis can make a valuable contribution in the clinical treatment of GC.
Ribosomal proteins (RPs) are a major component of ribosomes and play fundamental roles in ribosome biogenesis [6]. Increasing evidence has shown RPs to be closely associated with cell growth and tumorigenesis in various cancers, including GC [7]. The expression of ribosomal protein L34 (RPL34) is upregulated in GC cells, whereas a knockdown of RPL34 significantly suppresses proliferation and facilitates apoptosis in GC cells [8]. The expression of ribosomal protein L15 (RPL15) is upregulated in GC cells, and RPL15 silencing can restrain the growth of GC cells [9]. Ribosomal protein S13 (RPS13) and ribosomal protein L23 (RPL23) are upregulated in GC cells, and promote multidrug resistance in GC cells by suppressing apoptosis [10]. Ribosomal protein L22 (RPL22) is a part of the 60S large ribosomal subunit [11]. Yang et al [12] showed that there was minimal expression of RPL22 in non-small cell lung cancer (NSCLC). Ferreira et al reported that RPL22 played a crucial role in microsatellite instability in the carcinogenesis of colorectal and endometrial tumors [13]. Rao et al showed that the loss of RPL22 promoted lymphoma progression through accelerating angiogenesis [14]. However, the possible role and function of RPL22 in GC tumorigenesis is still unclear.
Protein 53 (p53) is acknowledged as a tumor suppressor, which modulates cellular processes, including the cell cycle, senescence, and apoptosis [15]. The ubiquitination and degradation of the p53 proteasome are controlled by human murine double minute 2 (MDM2), an oncogenic E3 ligase [16]. MDM2 exerts its tumor-suppressing function by inhibiting p53 activity and stability [17]. Increasing evidence suggests that p53 and MDM2 play vital roles in many cancers, including breast cancer [18], lung cancer [19], hepatocellular carcinoma [20], and GC [21]. Previous research has shown that several RPs play a critical role in MDM2-p53 signaling. For example, RPL5 and RPL11 bind to MDM2 and activate p53 [22,23]. RPL23 [24] and RPS7 [25] are MDM2-binding partners. A previous study proved that RPL22, a p53 activator, inhibited the colony formation of osteosarcoma cells through the regulation of MDM2-p53 signaling [26]. However, the regulatory mechanisms between RPL22 and MDM2-p53 signaling on GC progression are relatively unknown.
We investigated the expression of RPL22 in GC cells and tissues, and the possible functions of RPL22 in GC tumorigenesis, in vitro. The interactions between RPL22 and MDM2-p53 signaling in the GC cells were evaluated. These findings have potential applications for the clinical treatment of GC.
Material and Methods
TISSUE COLLECTION:
GC tissues and normal tissues were collected for the detection of RPL22 expression. From September 2016 to November 2018, 40 paired GC tissues and normal tissues were obtained from 40 patients with GC (22 men and 18 women, aged 35 years to 55 years). The inclusion criteria were patients with a first-time diagnosis, no history of radiotherapy, chemotherapy, or other adjuvant therapy. The exclusion criteria were the presence of other malignant tumors, and patients who had received GC treatment before admission.
CELL CULTURE:
The human gastric epithelial cell line (GES-1) and GC lines (MKN-45 and MGC-803) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). Subsequently, the cells were cultured in a Roswell Park Memorial Institute (RPMI)-1640 medium (Gibco, Carlsbad, CA, USA) containing 10% fetal bovine serum and 1% penicillin/streptomycin at 37°C with 5% CO2.
CELL TRANSFECTION:
RPL22 was silenced and overexpressed in the MKN-45 and MGC-803 cells by the transfection of RPL22 short interfering (si) RNA, (si-RPL22) and a vector (pcDNA) pcDNA3.1-RPL22 (pcDNA-RPL22), respectively (Table 1). pcDNA-RPL22, pcDNA negative control (pcDNA-NC), si-RPL22, and siRNA negative control (si-NC) were purchased from Han Biotech (Shanghai, China). Lipofectamine 3000 (Invitrogen, Carlsbad, CA, USA) was used to perform the relevant transfection trails according to the instructions. The cells without transfection were used as the blank group. The duration for transfection was approximately 48 h, followed by the collection of transfected cells for the experiments. In addition, nutlin-3 (a MDM2-p53 inhibitor, ab120646, Abcam, Cambridge, UK) was used to treat the transfected cells for 60 min.
QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (QRT-PCR):
A qRT-PCR was used to detect the expression of RPL22 in the GC tissues and cells. Total RNA was initially extracted from the cells and tissues using TRIzol Reagent (Promega, Madison, WI, USA), followed by synthesizing complementary DNA (cDNA) using the PrimeScript RT reagent kit (Takara, Kyoto, Japan), and performing a qRT-PCR with synergy brands green (SYBR Green) FAST Mastermix (Qiagen, Dusseldorf, Germany). The relative expression level was assessed by the 2−ΔΔCT method (27). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used to normalize the expression of RPL22. The primer sequences are listed in Table 1.
WESTERN BLOTTING:
The protein levels of RPL22, MDM2, p53, and GAPDH were determined by a western blot. The MGC-803 and MKN-45 cells were primarily lysed with a radioimmunoprecipitation assay (RIPA) buffer to collect the total protein. This was followed by separation of the protein product with a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, which was then transferred onto a polyvinylidene fluoride membrane (Millipore, Billerica, MA, USA). The membrane was blocked with 5% bovine serum albumin for 1 h, followed by an overnight incubation at 4°C with the diluted primary antibodies anti-RPL22 (1: 1000, ab77720; Abcam, Cambridge, UK), anti-MDM2 (1: 1000, #86934; Cell Signaling Technology, Danvers, MA, USA), anti-p53(1: 1000, #2527; Cell Signaling Technology, Danvers, MA, USA), and anti-GAPDH antibodies (1: 1000, #5174; Cell Signaling Technology, Danvers, MA, USA). After washing 3 times, the horseradish peroxidase (HRP)-conjugated secondary antibody (anti-rabbit; #14708; Cell Signaling Technology, Danvers, MA, USA) was added. Following incubation for 1 h at room temperature, the immunoblotting was visualized using an electrochemiluminescence exposure solution under a gel imaging system (Ultra-Violet Products, Upland, CA, USA).
CELL VIABILITY ASSAY:
A CCK-8 assay was used to measure the viability of the GC cells. The MGC-803 and MKN-45 cells were planted in 96-well plates (2×104 cells/well), and cultured for 24, 48, 72, and 96 h, respectively. Next, approximately 10 μL of CCK-8 solution (BD Biosciences, San Jose, CA, USA) was added and the incubation continued for another 2 h at 37°C. Cell viability was determined using a microplate reader (BioTek Instruments Inc., Winooski, VT, USA) at an optical density of 450 nm (OD450).
FLOW CYTOMETRIC ANALYSIS:
The cell apoptosis was determined using flow cytometric analysis. The cells (1×106 cells/mL) were cultured for 24 h in 96-well plates, followed by staining with fluorescein isothiocyanate (FITC) (5 μL) and propidium iodide (PI) (10 μL), using the Annexin V-FITC apoptosis detection kit (Thermo Fisher Scientific, Waltham, MA, USA) at 25°C for 20 min in the dark. The apoptotic cells (FITC+, PI±) were measured using a flow cytometer (BD Biosciences, San Jose, CA, USA). The Annexin V measurements were used as the horizontal axis and those of PI were used as the vertical axis. The cells represented in the lower left quadrant were viable cells, the cells in the upper left quadrant were necrotic cells, the cells in the lower right quadrant were early apoptotic cells, and those in the upper right quadrant were late apoptotic cells. The total apoptotic cells were calculated as the cells in the upper right quadrant plus cells in the lower right quadrant.
WOUND HEALING ASSAY:
The migration of the GC cells was determined by a wound healing assay. After transfection, the MGC-803 and MKN-45 cells were plated on 6-well plates (5×105 cells/well), and a vertical scratch was created at the center of the well using a 1 mL aseptic liquid remover. The migration of the GC cells was assessed using Image-Pro Plus Analysis software (Media Cybemetics, Silver Spring, MD, USA). The relative migration rate was calculated as (1–scratch area at 48 h/scratch area at 0 h)×100%.
TRANSWELL ASSAY:
A transwell assay was used to assess the invasion abilities of the GC cells. The cells (2×105 cells/well) cultured in a serum-free medium were seeded into the Matrigel-precoated (BD Biosciences, San Jose, CA, USA) upper chambers. A medium (600 μL) containing 100 ng/mL of chemokine stromal cell-derived factor-1 (Sino Biological, Beijing, China) was added to the lower chambers. After 24 h of culturing, the invasion cells were fixed in methanol and stained with 0.1% crystal violet for 30 min. Cells from 4 random fields were photographed and counted.
STATISTICAL ANALYSIS:
Each assay was performed at least 3 times. Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS version 22.0) (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 7.0 (Graphpad Software, San Diego, CA, USA). Data were presented as mean±standard deviation. Comparisons between the 2 groups were performed using the t test, and comparisons between >2 groups were carried out by one-way analysis of variance, followed by Fisher’s Least Significant Difference test.
Each participant gave written informed consent. All the procedures followed in this study were approved by the Ethics Committee of our hospital (approval ID: QYFY WZLL25915).
Results
DECREASED EXPRESSION OF RPL22 OBSERVED IN GASTRIC CANCER TISSUES AND CELL LINES:
The expression of RPL22 was initially detected in the tissues using a qRT-PCR analysis. In comparison with normal tissues, a declined expression of RPL22 was observed in the GC tissues (P<0.001) (Figure 1A). We determined the expression of RPL22 in the GC cell lines (AGS, MKN-45, and MGC-803). The results of the qRT-PCR and western blotting revealed that RPL22 expression was dramatically reduced in the GC cell lines in contrast to the GES-1 cells (P<0.05) (Figure 1B, 1C). As presented in Table 2, RPL22 expression had no correlation with gender, age, tumor location, and degree of histological type of the tissue; however, it was strongly correlated with the early stage of GC (P=0.0372) and non-lymphatic (P=0.0212) metastasis.
RPL22 AFFECTS THE PROLIFERATION AND APOPTOSIS OF GASTRIC CANCER CELLS:
To identify the role of RPL22 on GC carcinogenicity, first, we detected the transfection efficiency after transfection of si-RPL22-1/-2/pcDNA-RPL22 into the GC cells. The results of the qRT-PCR and western blotting revealed that RPL22 expression was distinctly elevated by the transfection with pcDNA-RPL22 in the MKN-45 cells, while it was dampened by the transfection with si-RPL22 in the MGC803 cells (P<0.01) (Figure 2A, 2B). The si-RPL22-1 was selected for subsequent transfection experiments due to its higher silencing efficiency. The CCK-8 assay results showed that the si-RPL22-1 transfection significantly increased the OD450 values of the MGC803 cells at 48, 72, and 96 h post culture. However, the overexpression of RPL22 obtained the opposite result (P<0.01) (Figure 2C). The apoptosis rate of the si-RPL22-1-transfected MGC803 cells was remarkably decreased (P<0.01), while that of the pcDNA-RPL22-transfected MKN-45 cells increased substantially when compared with the blank group (P<0.01) (Figure 2D).
RPL22 AFFECTS THE METASTASIS OF GASTRIC CANCER CELLS:
The abilities of migration and invasion were visibly elevated by the transfection of si-RPL22-1 in MGC803 cells compared to the blank cells (Figure 3A, 3B); however, these abilities were significantly restrained by the transfection of pcDNA-RPL22 in the MKN-45 cells (P<0.01).
SILENCING RPL22 ACTIVATES THE MDM2-P53 SIGNALING PATHWAY:
We determined the protein expression of MDM2 and p53 after the transfection of si-RPL22-1/pcDNA-RPL22. The results of western blotting demonstrated that expression of MDM2 increased, while p53 expression decreased in the MGC803 cells in the si-RPL22-1 group compared to the cells in the blank group (P<0.01) (Figure 4A). In contrast, the transfection with pcDNA-RPL22 downregulated the expression of MDM2, and upregulated the expression of p53 in the MKN-45 cells (P<0.01) (Figure 4B).
SI-RPL22-1 PROMOTES THE PROLIFERATION, MIGRATION, AND INVASION OF MKN-45 CELLS BY MODULATING MDM2-P53 SIGNALING:
To confirm the influence of RPL22 on MDM2-p53 signaling in the GC cells, MDM2-p53 signaling was blocked by nutlin-3 treatment. The treatment with 50 μM nutlin-3 dramatically elevated the p53 expression, as well as decreased the protein expression of MDM2 in the MKN-45 cells (P<0.01) (Figure 5A). Nutlin-3 not only reversed the promoting effects of si-RPL22 on proliferation, migration and invasion, but also the inhibiting effects on the apoptosis of MKN-45 cells (P<0.01) (Figure 5B–5E).
Discussion
RPs are abundant RNA-binding proteins, and the expression of many RPs is altered during tumorigenesis [28]. Studies have shown the upregulation and downregulation of RPs in different types of cancers, including the upregulation of RPL7a [29], RPL19 [30], RPL15 [9], and RPL34 [28], in human colorectal carcinoma, breast cancer, and GC, respectively; and the downregulation of RPL22 in NSCLC [12]. Evidence suggests that the abnormal expression of RPs was closely associated with tumorigenesis. In the present study, the RPL22 expression was significantly reduced in GC cells (
RPs are considered to be regulators in the tumorigenesis of different cancers. The knockdown of RPS15A can suppress cell growth and colony formation, as well as induce cell apoptosis in human glioblastomas [34]. In vitro, the knockdown of RPL34 significantly inhibited cell proliferation in esophageal cancer [35]. The knockdown of RPL13 was shown to attenuate cell growth and induce cell cycle arrest in gastrointestinal cancer [33]. We found that RPL22 silencing efficiently increased the OD450 values and decreased the apoptosis rate. Our findings suggest that RPL22 could be a participant in the progression and development of GC by modulating cell proliferation and apoptosis. Therefore, we inferred that RPL22 can inhibit the development of GC, and be a potential therapeutic strategy for GC patients. We further explored the mechanisms underlying the RPL22-mediated GC metastasis. Metastasis, which involves the transfer of malignant tumors from an organ to a distant organ, is a frequent cause of death in patients with cancer [36]. In our research, we found that RPL22 silencing efficiently elevated the number of migrating and invading cells. Our findings were similar to those of previous studies. For example, the knockdown of RPS3 was shown to decrease the invasion and migration of osteosarcoma cells [37]. RPS24 silencing efficiently blocked the cell migration of colon cancer cells [38]. The silencing of RPS7 was shown to reduce cell invasion and migration, decrease the expression of β-catenin, MMP2 and MMP13 (genes), and increase the E-cadherin expression in RPS7-silenced cells [39]. Our results demonstrated that the silencing of RPL22 promoted the metastasis of GC cells. Therefore, our findings suggest that RPL22 could be involved in the progression of GC by the regulation of cell migration and invasion. Under clinical conditions, RPL22 can inhibit the metastasis of GC, thereby contributing to an improvement in the poor prognosis of GC.
p53 is a tumor suppressor, and its function is significantly inhibited by binding to the MDM2 oncoprotein [40]. Hence, blocking the MDM2-p53 interaction to reactivate the p53 function can be a potential therapeutic cancer strategy. In our study, p53 expression was significantly decreased in the si-RPL22-transfected GC cells (
Conclusions
A low expression of RPL22 was found in the GC tissues and cells. RPL22 silencing expedited the GC cells’ proliferation, migration, and invasion, as well as inhibited cell apoptosis by activating the MDM2-p53 signaling. Hence, we infer that RPL22 could be an underlying target for GC treatment. However, this study did not confirm the detailed mechanisms between RPL22 and MDM2-p53 signaling in vivo, which are a limitation of the present study. In the future, further experiments are needed to elucidate these issues.
Figures
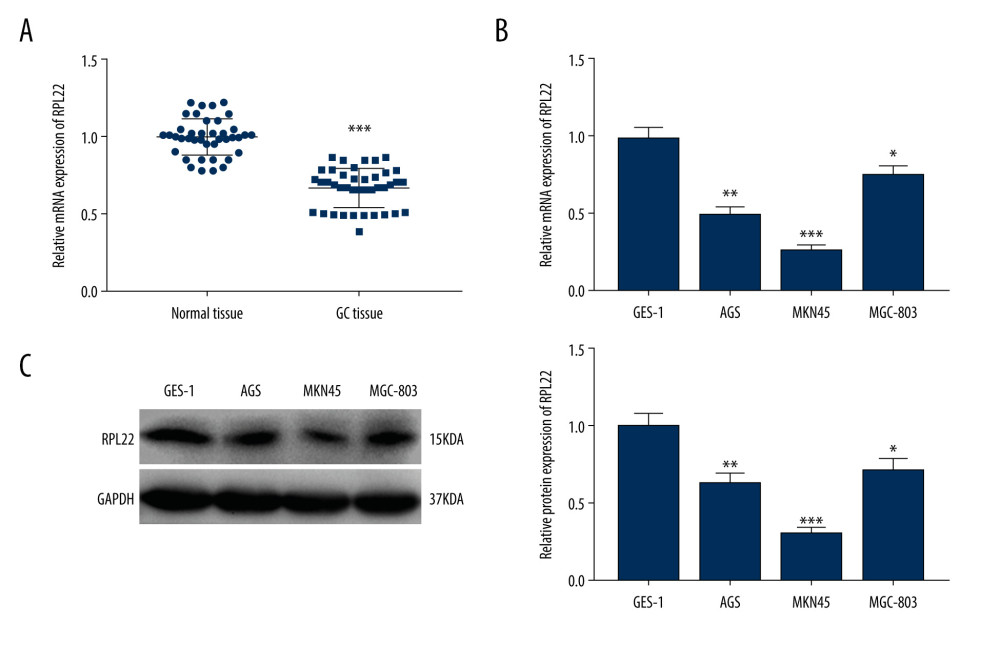 Figure 1. Ribosomal protein L22 (RPL22) is downregulated in gastric cancer tissues and cell lines. (A) The mRNA expression of RPL22 in gastric cancer tissues and normal tissues was detected by a quantitative real-time-polymerase chain reaction. (B) The mRNA expression of RPL22 in gastric cancer cell lines (AGS, MKN-45, and MGC-803) and the normal human gastric epithelial cell line (GES-1) was detected by a quantitative real-time-polymerase chain reaction. (C) The protein expression of RPL22 in gastric cancer cell lines and the normal human gastric epithelial cell line (GES-1) was detected by a western blot. (A) *** P<0.001 vs normal tissues; (B, C) * P<0.05, ** P<0.01, *** P<0.001 vs GES-1.
Figure 1. Ribosomal protein L22 (RPL22) is downregulated in gastric cancer tissues and cell lines. (A) The mRNA expression of RPL22 in gastric cancer tissues and normal tissues was detected by a quantitative real-time-polymerase chain reaction. (B) The mRNA expression of RPL22 in gastric cancer cell lines (AGS, MKN-45, and MGC-803) and the normal human gastric epithelial cell line (GES-1) was detected by a quantitative real-time-polymerase chain reaction. (C) The protein expression of RPL22 in gastric cancer cell lines and the normal human gastric epithelial cell line (GES-1) was detected by a western blot. (A) *** P<0.001 vs normal tissues; (B, C) * P<0.05, ** P<0.01, *** P<0.001 vs GES-1. 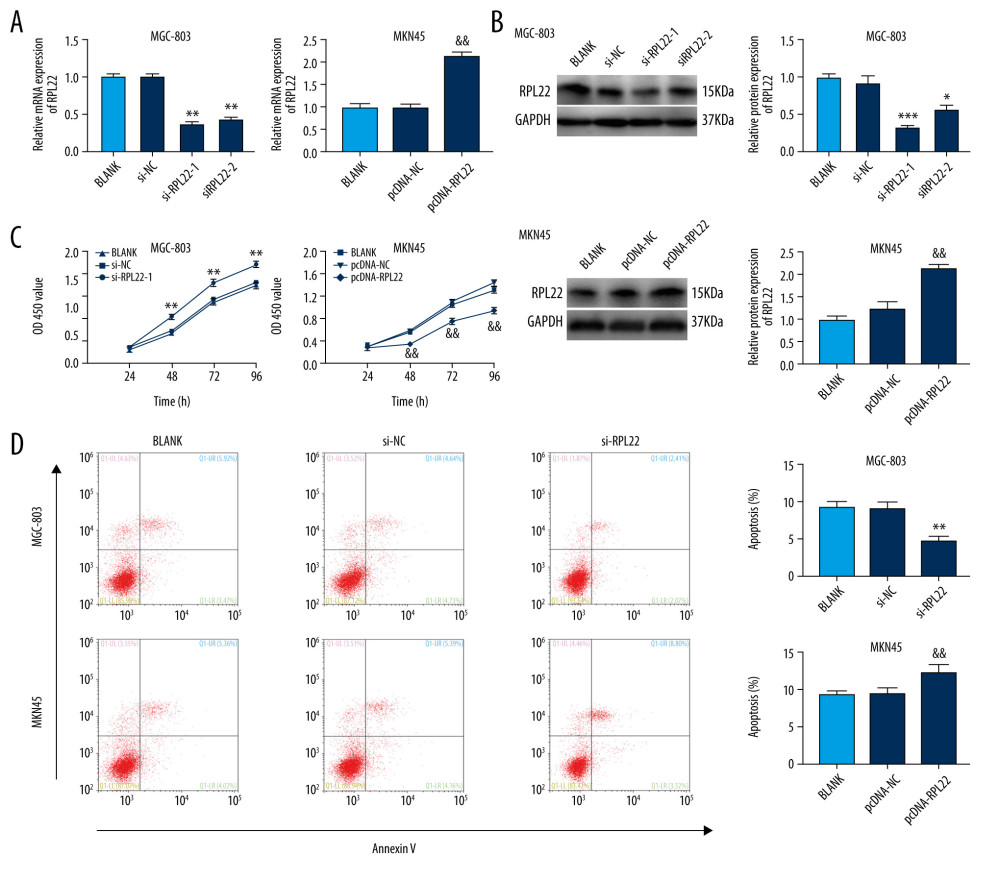 Figure 2. Ribosomal protein L22 (RPL22) influences the proliferation and apoptosis of gastric cancer cells.(A) The mRNA expression of RPL22 in MKN-45 and MGC-803 cells was detected by a quantitative real-time polymerase chain reaction. (B) The protein expression of RPL22 in MKN-45 and MGC-803 cells was detected by a western blot. (C) The optical density value of the MKN-45 and MGC-803 cells was detected by a CCK-8 assay. (D) The apoptosis rate of the MKN-45 and MGC-803 cells was detected by a flow cytometry assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-2 – cells transfected with siRNA-RPL22-2. (A–D) * P<0.05, ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK.
Figure 2. Ribosomal protein L22 (RPL22) influences the proliferation and apoptosis of gastric cancer cells.(A) The mRNA expression of RPL22 in MKN-45 and MGC-803 cells was detected by a quantitative real-time polymerase chain reaction. (B) The protein expression of RPL22 in MKN-45 and MGC-803 cells was detected by a western blot. (C) The optical density value of the MKN-45 and MGC-803 cells was detected by a CCK-8 assay. (D) The apoptosis rate of the MKN-45 and MGC-803 cells was detected by a flow cytometry assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-2 – cells transfected with siRNA-RPL22-2. (A–D) * P<0.05, ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK. 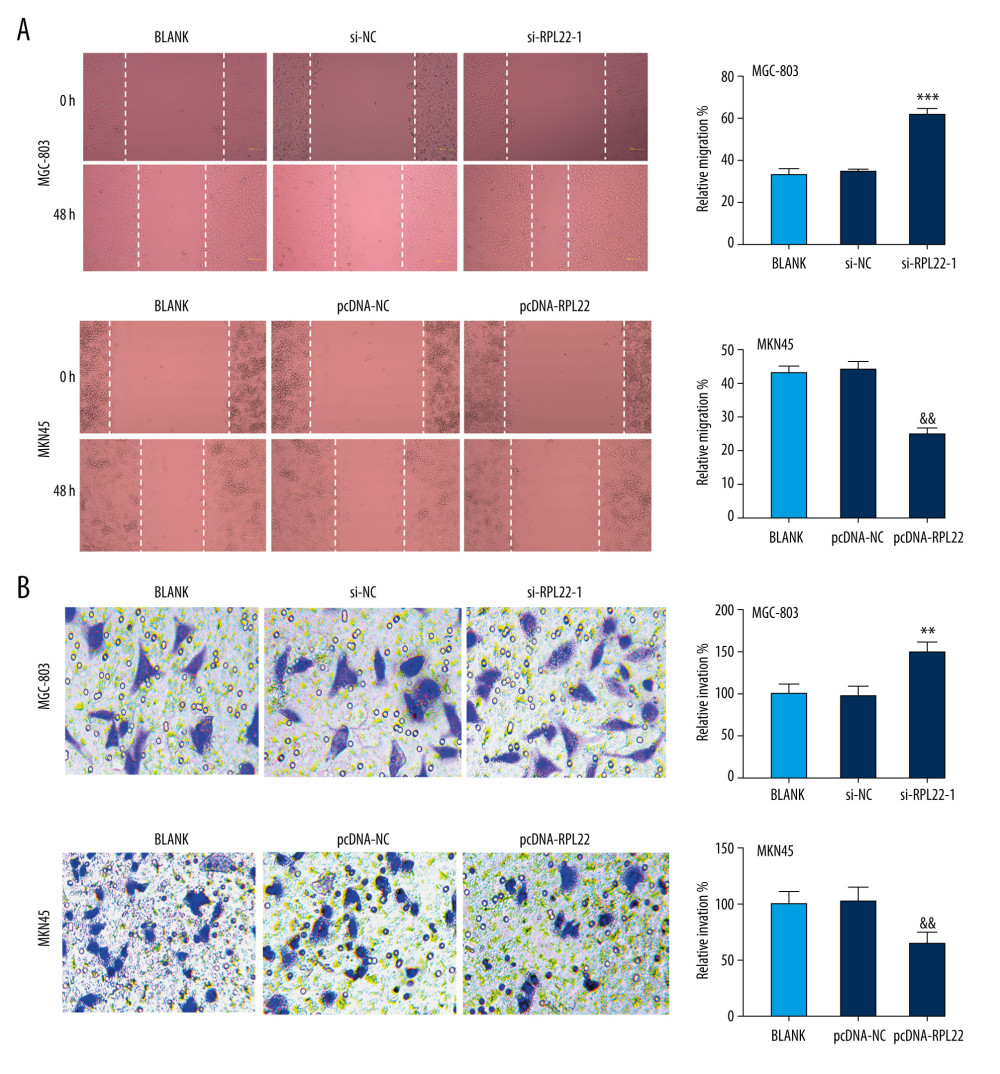 Figure 3. Ribosomal protein L22 (RPL22) influences the migration and invasion of gastric cancer cells.(A) The relative migration of MKN-45 and MGC-803 cells was detected by a wound healing assay. (B) The relative invasion of MKN-45 and MGC-803 cells was detected by a transwell assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK.
Figure 3. Ribosomal protein L22 (RPL22) influences the migration and invasion of gastric cancer cells.(A) The relative migration of MKN-45 and MGC-803 cells was detected by a wound healing assay. (B) The relative invasion of MKN-45 and MGC-803 cells was detected by a transwell assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK. 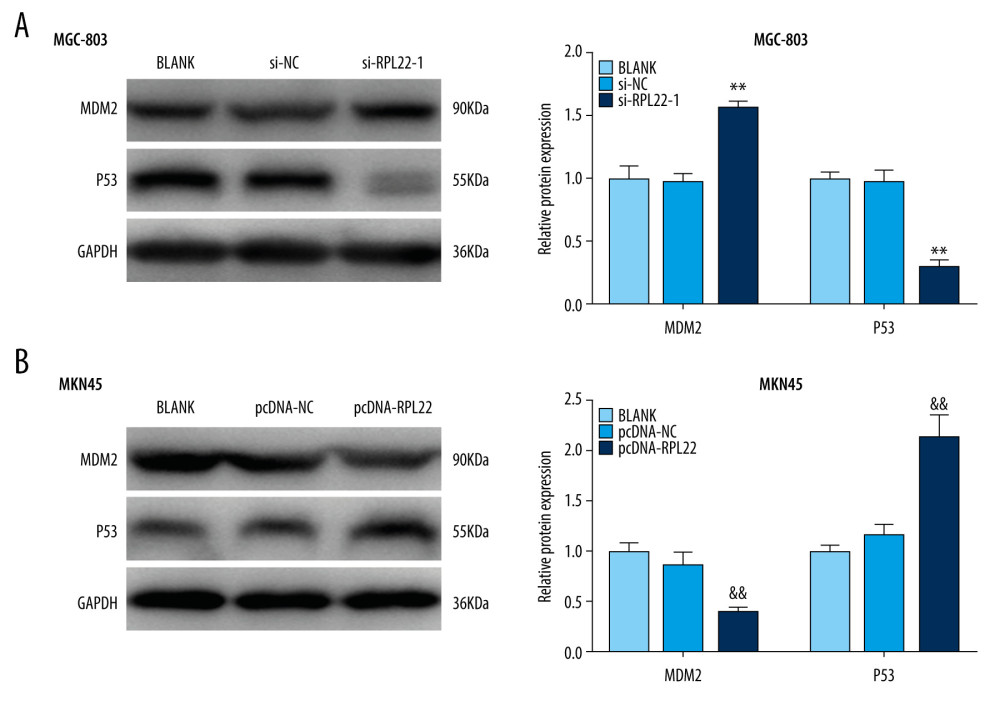 Figure 4. Silencing the ribosomal protein L22 (RPL22) activates MDM2-p53 signaling. (A) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MGC-803 cells was detected by a western blot. (B) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MKN45 cells was detected by a western blot. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22 plasmid; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01 vs BLANK; ## P<0.01 vs BLANK.
Figure 4. Silencing the ribosomal protein L22 (RPL22) activates MDM2-p53 signaling. (A) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MGC-803 cells was detected by a western blot. (B) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MKN45 cells was detected by a western blot. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22 plasmid; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01 vs BLANK; ## P<0.01 vs BLANK. 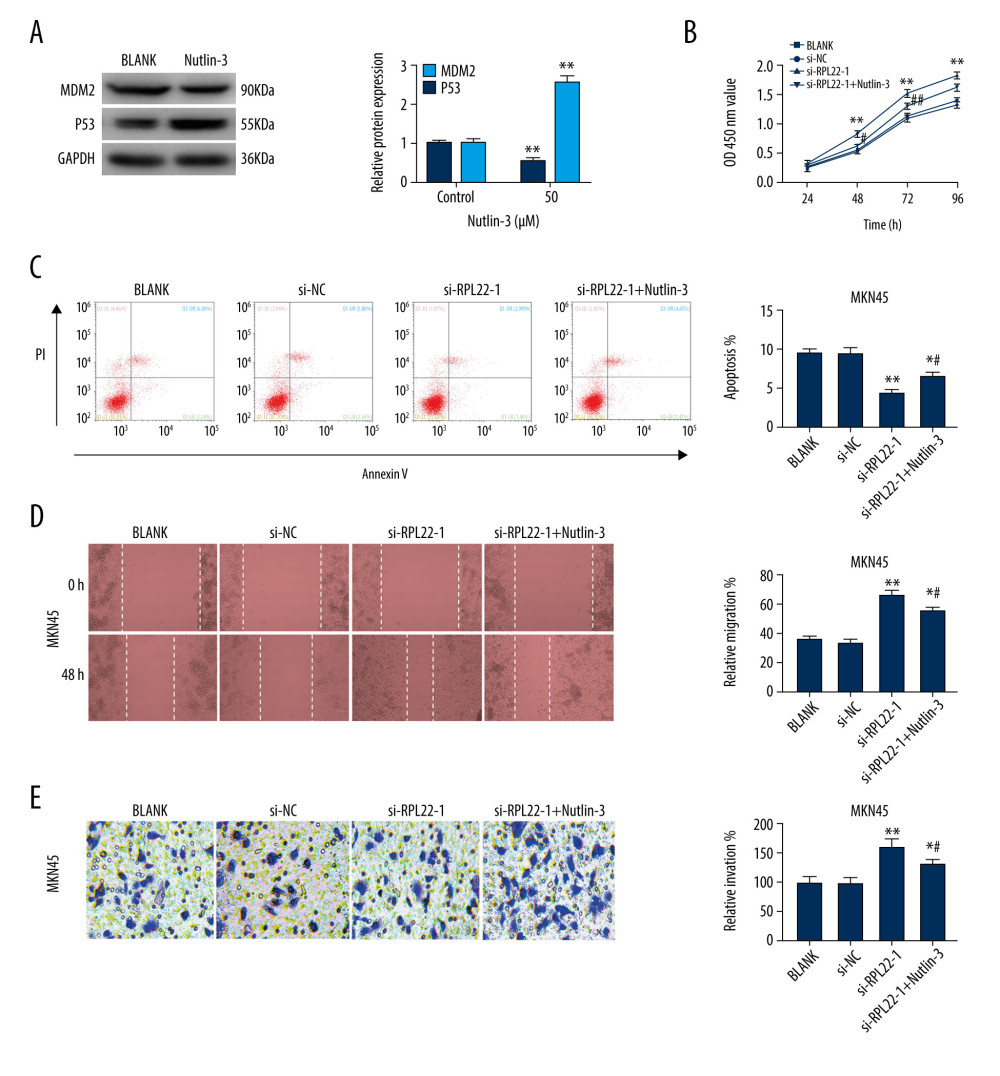 Figure 5. Ribosomal protein L22 (RPL22) inhibits the proliferation, migration, and invasion of MKN-45 cells through regulation of MDM2-p53 signaling. (A) The protein expression of MDM2 and p53 in MKN-45 cells was detected by a western blot.(B) The optic density (OD450) value of the MKN-45 cells was detected by a CCK-8 assay. (C) The apoptosis rate of the MKN-45 cells was detected by a flow cytometry assay. (D) The relative migration of the MKN-45 cells was detected by a wound healing assay. (E) The relative invasion of the MKN-45 cells was detected by a transwell assay. BLANK – cells without transfection; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-1+nutlin-3 – cells transfected with siRNA-RPL22-1 and nutlin-3. (A) ** P<0.01 vs control; (B–E) ** P<0.01 vs BLANK; # P<0.05, ## P<0.01 vs si-RPL22-1.
Figure 5. Ribosomal protein L22 (RPL22) inhibits the proliferation, migration, and invasion of MKN-45 cells through regulation of MDM2-p53 signaling. (A) The protein expression of MDM2 and p53 in MKN-45 cells was detected by a western blot.(B) The optic density (OD450) value of the MKN-45 cells was detected by a CCK-8 assay. (C) The apoptosis rate of the MKN-45 cells was detected by a flow cytometry assay. (D) The relative migration of the MKN-45 cells was detected by a wound healing assay. (E) The relative invasion of the MKN-45 cells was detected by a transwell assay. BLANK – cells without transfection; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-1+nutlin-3 – cells transfected with siRNA-RPL22-1 and nutlin-3. (A) ** P<0.01 vs control; (B–E) ** P<0.01 vs BLANK; # P<0.05, ## P<0.01 vs si-RPL22-1. References
1. Venerito M, Link A, Rokkas T, Review: Gastric cancer – clinical aspects: Helicobacter, 2019; 24; e12643
2. Song H, Zhu J, Lu D, Molecular-targeted first-line therapy for advanced gastric cancer: Cochrane Database Syst Rev, 2016; 7(7); CD011461
3. Duraes C, Almeida GM, Seruca R, Biomarkers for gastric cancer: Prognostic, predictive or targets of therapy?: Virchows Arch, 2014; 464(3); 367-78
4. Fugazzola P, Ansaloni L, Sartelli M, Advanced gastric cancer: The value of surgery: Acta Biomed, 2018; 89(Suppl8); 110-16
5. Feng W, Zhu X, Efficacy prediction of targeted therapy for gastric cancer: The current status (review): Mol Med Rep, 2018; 18(2); 1238-46
6. Penzo M, Montanaro L, Treré D, The ribosome biogenesis – cancer connection: Cells, 2019; 8(1); 55
7. Shi D, Liu J, RPS15a silencing suppresses cell proliferation and migration of gastric cancer: Yonsei Med J, 2018; 59(10); 1166-73
8. Liu H, Liang S, Yang XI, RNAi-mediated RPL34 knockdown suppresses the growth of human gastric cancer cells: Oncol Rep, 2015; 34(5); 2267-72
9. Wang H, Zhao LN, Li KZ, Overexpression of ribosomal protein L15 is associated with cell proliferation in gastric cancer: BMC Cancer, 2006; 6(1); 91
10. Shi Y, Zhai H, Wang X, Ribosomal proteins S13 and L23 promote multidrug resistance in gastric cancer cells by suppressing drug-induced apoptosis: Experimental Cell Research, 2004; 296(2); 337-46
11. Feng Z, Zhang L, Wu Y: Planta, 2019; 250(6); 1897-910
12. Yang M, Sun H, Wang H, Down-regulation of ribosomal protein L22 in non-small cell lung cancer: Medical Oncology, 2013; 30(3); 646
13. Ferreira AM, Tuominen I, van Dijk-Bos K, High frequency of RPL22 mutations in microsatellite-unstable colorectal and endometrial tumors: Hum Mutat, 2014; 35(12); 1442-45
14. Rao S, Stadanlick JE, Cai KQ, Abstract 5215: Loss of Rpl22 promotes tumor progression through regulation of angiogenesis: Cancer Res, 2015; 75(15 Suppl); 5215
15. Baker SJ, Vogelstein B, p53: A tumor suppressor hiding in plain sight: J Mol Cell Biol, 2019; 11(7); 536-38
16. Ranjan A, Bera K, Iwakuma T, Murine double minute 2, a potential p53-independent regulator of liver cancer metastasis: Hepatoma Res, 2016; 2; 114-21
17. Nag S, Zhang X, Srivenugopal KS, Targeting MDM2-p53 interaction for cancer therapy: are we there yet?: Curr Med Chem, 2014; 21(5); 553-74
18. Qin JJ, Wang W, Zhang R, Experimental therapy of advanced breast cancer: Targeting NFAT1-MDM2-p53 pathway: Prog Mol Biol Transl Sci, 2017; 151; 195-216
19. Xing Y, Liu Y, Liu T, TNFAIP8 promotes the proliferation and cisplatin chemoresistance of non-small cell lung cancer through MDM2/p53 pathway: Cell Commun Signal, 2018; 16(1); 43
20. Cao H, Chen X, Wang Z, The role of MDM2-p53 axis dysfunction in the hepatocellular carcinoma transformation: Cell Death Discov, 2020; 6(1); 53
21. Yi H, Yan X, Luo Q, A novel small molecule inhibitor of MDM2-p53 (APG-115) enhances radiosensitivity of gastric adenocarcinoma: J Exp Clin Cancer Res, 2018; 37(1); 1-10
22. Qi Z, Hui X, Chai SC, Hydrophilic residues are crucial for ribosomal protein L11 (RPL11) interaction with zinc finger domain of MDM2 and p53 protein activation: J Biol Chem, 2011; 286(44); 38264-74
23. Turi Z, Senkyrikova M, Mistrik M, Perturbation of RNA Polymerase I transcription machinery by ablation of HEATR1 triggers the RPL5/RPL11-MDM2-p53 ribosome biogenesis stress checkpoint pathway in human cells: Cell Cycle, 2018; 17(1); 92-101
24. Zhang Y, Shi Y, Li X, Inhibition of the p53-MDM2 interaction by adenovirus delivery of ribosomal protein L23 stabilizes p53 and induces cell cycle arrest and apoptosis in gastric cancer: J Gene Med, 2010; 12(2); 147-56
25. Yan Z, Poyurovsky MV, Li Y, Ribosomal protein S7 is both a regulator and a substrate of MDM2: Mol Cell, 2009; 35(3); 316-26
26. Bo C, Fang Z, Liao P, Cancer-mutated ribosome protein L22 (RPL22/eL22) suppresses cancer cell survival by blocking p53-MDM2 circuit: Oncotarget, 2017; 8; 90651-61
27. Livak KJ, Schmittgen TD, Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method: Methods, 2001; 25(4); 402-8
28. Xu X, Xiong X, Sun Y, The role of ribosomal proteins in the regulation of cell proliferation, tumorigenesis, and genomic integrity: Sci China Life Sci, 2016; 59(7); 656-72
29. Wang Y, Cheong D, Chan S, Ribosomal protein L7a gene is up-regulated but not fused to the tyrosine kinase receptor as chimeric trk oncogene in human colorectal carcinoma: Int J Oncol, 2000; 16(4); 757-62
30. Hong M, Kim HR, Kim I, Ribosomal protein L19 overexpression activates the unfolded protein response and sensitizes MCF7 breast cancer cells to endoplasmic reticulum stress-induced cell death: Biochem Biophys Res Commun, 2014; 450(1); 673-78
31. Pan Y, Jun Q, Zhou LRibosomal protein 16 overexpresses in prostate cancer and promotes tumor progression: Journal of Shanghai Jiaotong University (Medical Science), 2018; 38(4); 394-99 [in Chinese]
32. Huang CJ, Chien CC, Yang SH, Faecal ribosomal protein L19 is a genetic prognostic factor for survival in colorectal cancer: J Cell Mol Med, 2008; 12(5b); 1936-43
33. Kobayashi T, Sasaki Y, Oshima Y, Activation of the ribosomal protein L13 gene in human gastrointestinal cancer: Int J Mol Med, 2006; 18(1); 161-70
34. Zhang C, Fu J, Xue F, Knockdown of ribosomal protein S15A induces human glioblastoma cell apoptosis: World J Surg Oncol, 2016; 4(1); 129
35. Fan H, Li J, Jia Y, Silencing of ribosomal protein L34 (RPL34) inhibits the proliferation and invasion of esophageal cancer cells: Oncol Res, 2017; 25; 1061-68
36. Chen X, Liu B, Yang R, Integrated analysis of long non-coding RNAs in human colorectal cancer: Oncotarget, 2016; 7(17); 23897-908
37. Nagao-Kitamoto H, Setoguchi T, Kitamoto S, Ribosomal protein S3 regulates GLI2-mediated osteosarcoma invasion: Cancer Lett, 2015; 356(2); 855-61
38. Wang Y, Sui J, Li X, RPS24 knockdown inhibits colorectal cancer cell migration and proliferation in vitro: Gene, 2015; 571(2); 286-91
39. Zhen W, Lu L, Sun J, Abstract 4303: The small ribosomal protein subunit S7 functions as a tumor suppressor to regulate ovarian tumorigenesis through PI3K/AKT and MAPK pathways: Cancer Res, 2013; 73; 4303
40. Åberg E, Saccoccia F, Grabherr M, Evolution of the p53-MDM2 pathway: BMC Evol Biol, 2017; 17(1); 177
41. Chen D, Zhang Z, Li M, Ribosomal protein S7 as a novel modulator of p53-MDM2 interaction: Binding to MDM2, stabilization of p53 protein, and activation of p53 function: Oncogene, 2007; 26(35); 5029-37
Figures
 Figure 1. Ribosomal protein L22 (RPL22) is downregulated in gastric cancer tissues and cell lines. (A) The mRNA expression of RPL22 in gastric cancer tissues and normal tissues was detected by a quantitative real-time-polymerase chain reaction. (B) The mRNA expression of RPL22 in gastric cancer cell lines (AGS, MKN-45, and MGC-803) and the normal human gastric epithelial cell line (GES-1) was detected by a quantitative real-time-polymerase chain reaction. (C) The protein expression of RPL22 in gastric cancer cell lines and the normal human gastric epithelial cell line (GES-1) was detected by a western blot. (A) *** P<0.001 vs normal tissues; (B, C) * P<0.05, ** P<0.01, *** P<0.001 vs GES-1.
Figure 1. Ribosomal protein L22 (RPL22) is downregulated in gastric cancer tissues and cell lines. (A) The mRNA expression of RPL22 in gastric cancer tissues and normal tissues was detected by a quantitative real-time-polymerase chain reaction. (B) The mRNA expression of RPL22 in gastric cancer cell lines (AGS, MKN-45, and MGC-803) and the normal human gastric epithelial cell line (GES-1) was detected by a quantitative real-time-polymerase chain reaction. (C) The protein expression of RPL22 in gastric cancer cell lines and the normal human gastric epithelial cell line (GES-1) was detected by a western blot. (A) *** P<0.001 vs normal tissues; (B, C) * P<0.05, ** P<0.01, *** P<0.001 vs GES-1. Figure 2. Ribosomal protein L22 (RPL22) influences the proliferation and apoptosis of gastric cancer cells.(A) The mRNA expression of RPL22 in MKN-45 and MGC-803 cells was detected by a quantitative real-time polymerase chain reaction. (B) The protein expression of RPL22 in MKN-45 and MGC-803 cells was detected by a western blot. (C) The optical density value of the MKN-45 and MGC-803 cells was detected by a CCK-8 assay. (D) The apoptosis rate of the MKN-45 and MGC-803 cells was detected by a flow cytometry assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-2 – cells transfected with siRNA-RPL22-2. (A–D) * P<0.05, ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK.
Figure 2. Ribosomal protein L22 (RPL22) influences the proliferation and apoptosis of gastric cancer cells.(A) The mRNA expression of RPL22 in MKN-45 and MGC-803 cells was detected by a quantitative real-time polymerase chain reaction. (B) The protein expression of RPL22 in MKN-45 and MGC-803 cells was detected by a western blot. (C) The optical density value of the MKN-45 and MGC-803 cells was detected by a CCK-8 assay. (D) The apoptosis rate of the MKN-45 and MGC-803 cells was detected by a flow cytometry assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-2 – cells transfected with siRNA-RPL22-2. (A–D) * P<0.05, ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK. Figure 3. Ribosomal protein L22 (RPL22) influences the migration and invasion of gastric cancer cells.(A) The relative migration of MKN-45 and MGC-803 cells was detected by a wound healing assay. (B) The relative invasion of MKN-45 and MGC-803 cells was detected by a transwell assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK.
Figure 3. Ribosomal protein L22 (RPL22) influences the migration and invasion of gastric cancer cells.(A) The relative migration of MKN-45 and MGC-803 cells was detected by a wound healing assay. (B) The relative invasion of MKN-45 and MGC-803 cells was detected by a transwell assay. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01, *** P<0.001 vs BLANK; && P<0.01 vs BLANK. Figure 4. Silencing the ribosomal protein L22 (RPL22) activates MDM2-p53 signaling. (A) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MGC-803 cells was detected by a western blot. (B) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MKN45 cells was detected by a western blot. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22 plasmid; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01 vs BLANK; ## P<0.01 vs BLANK.
Figure 4. Silencing the ribosomal protein L22 (RPL22) activates MDM2-p53 signaling. (A) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MGC-803 cells was detected by a western blot. (B) The expression of MDM2-p53 signaling-related proteins (MDM2 and p53) in MKN45 cells was detected by a western blot. BLANK – cells without transfection; pcDNA-NC – cells transfected with pcDNA negative control; pcDNA-RPL22 – cells transfected with pcDNA-RPL22 plasmid; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1. (A, B) ** P<0.01 vs BLANK; ## P<0.01 vs BLANK. Figure 5. Ribosomal protein L22 (RPL22) inhibits the proliferation, migration, and invasion of MKN-45 cells through regulation of MDM2-p53 signaling. (A) The protein expression of MDM2 and p53 in MKN-45 cells was detected by a western blot.(B) The optic density (OD450) value of the MKN-45 cells was detected by a CCK-8 assay. (C) The apoptosis rate of the MKN-45 cells was detected by a flow cytometry assay. (D) The relative migration of the MKN-45 cells was detected by a wound healing assay. (E) The relative invasion of the MKN-45 cells was detected by a transwell assay. BLANK – cells without transfection; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-1+nutlin-3 – cells transfected with siRNA-RPL22-1 and nutlin-3. (A) ** P<0.01 vs control; (B–E) ** P<0.01 vs BLANK; # P<0.05, ## P<0.01 vs si-RPL22-1.
Figure 5. Ribosomal protein L22 (RPL22) inhibits the proliferation, migration, and invasion of MKN-45 cells through regulation of MDM2-p53 signaling. (A) The protein expression of MDM2 and p53 in MKN-45 cells was detected by a western blot.(B) The optic density (OD450) value of the MKN-45 cells was detected by a CCK-8 assay. (C) The apoptosis rate of the MKN-45 cells was detected by a flow cytometry assay. (D) The relative migration of the MKN-45 cells was detected by a wound healing assay. (E) The relative invasion of the MKN-45 cells was detected by a transwell assay. BLANK – cells without transfection; si-NC – cells transfected with siRNA negative control; si-RPL22-1 – cells transfected with siRNA-RPL22-1; si-RPL22-1+nutlin-3 – cells transfected with siRNA-RPL22-1 and nutlin-3. (A) ** P<0.01 vs control; (B–E) ** P<0.01 vs BLANK; # P<0.05, ## P<0.01 vs si-RPL22-1. Tables
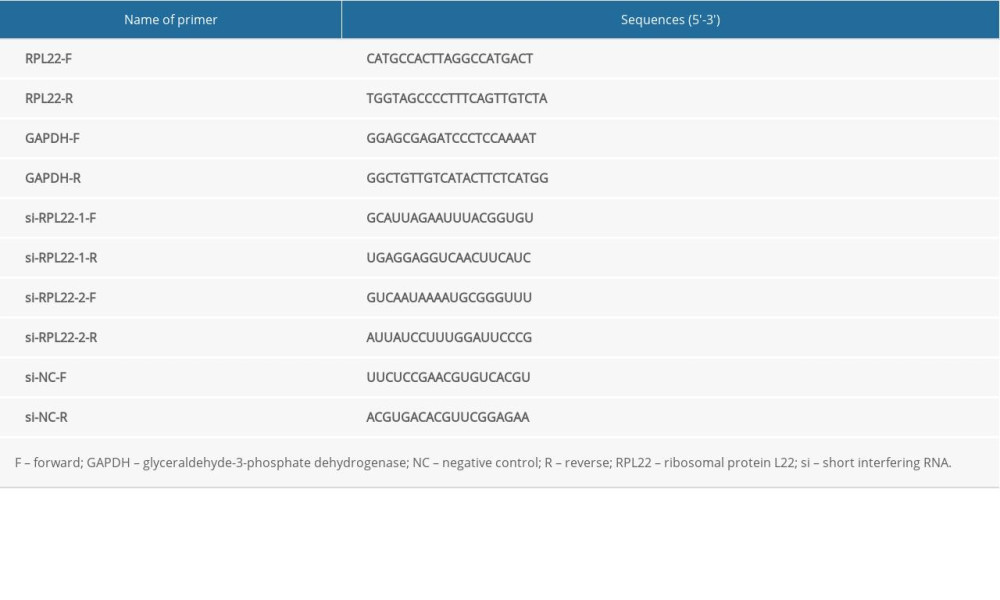 Table 1. Primer sequences used in the quantitative real-time polymerase chain reaction.
Table 1. Primer sequences used in the quantitative real-time polymerase chain reaction.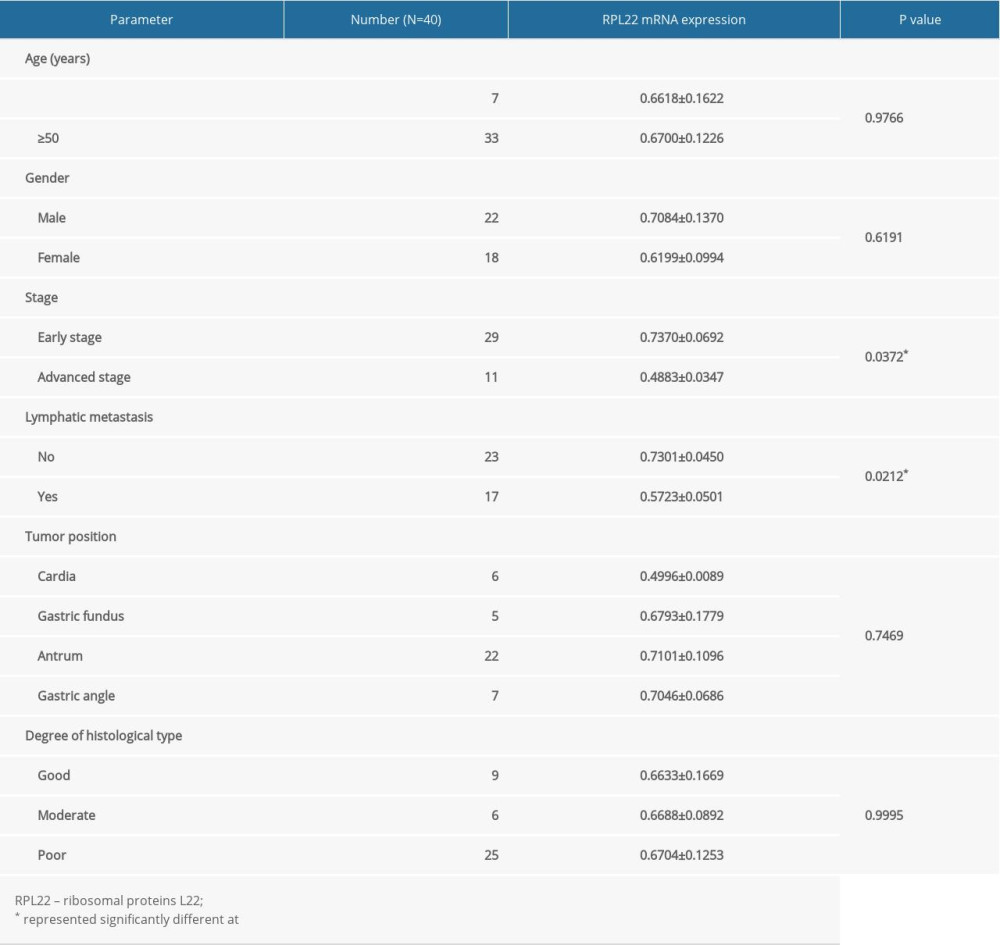 Table 2. Correlation between the expression of RPL22 and clinicopathological features of gastric cancer patients.
Table 2. Correlation between the expression of RPL22 and clinicopathological features of gastric cancer patients. Table 1. Primer sequences used in the quantitative real-time polymerase chain reaction.
Table 1. Primer sequences used in the quantitative real-time polymerase chain reaction. Table 2. Correlation between the expression of RPL22 and clinicopathological features of gastric cancer patients.
Table 2. Correlation between the expression of RPL22 and clinicopathological features of gastric cancer patients. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








