26 March 2021: Clinical Research
Developmental Patterns of Fecal Bile Acids in Healthy Neonates and Children
Jing-Jing Xiong1ABCE, Hong-Wei Hu1BCF, Chuan-Zhi Xu2C, Jian-Wen Yin3BC, Mei Liu1B, Li-Zhi Zhang1B, Yong Duan4B, Yong-Kun Huang1ABCE*DOI: 10.12659/MSM.928214
Med Sci Monit 2021; 27:e928214
Abstract
BACKGROUND: Normal profiles of FBAs in healthy neonates and children in Kunming city and surrounding areas in China have not been previously determined. The objective of this study was to determine a developmental pattern of fecal bile acids (FBAs) in healthy neonates and children.
MATERIAL AND METHODS: A cross-sectional study was performed on 238 healthy neonates and children recruited in the First Affiliated Hospital of Kunming Medical University, China from October 2015 to September 2016. Secreted primary and secondary FBAs in fresh feces were quantitated by liquid chromatography mass spectrometry (LC-MS). Amounts of FBAs in feces were compared among various age groups.
RESULTS: Trace amounts of cholic acid and chenodiol acid of primary FBAs were detectable at day 3 after birth, with a significant increase from day 3 to day 7. The primary FBAs gradually decreased from day 25 to the age of 6 years old. In contrast, a significant amount of glycochenodeoxycholic acid was detected on day 3 but decreased to a trace amount by day 7 and onwards. Primary FBAs appeared to maintain a high level, accounting for 98% of total FBAs, with no significant changes from day 7 to day 25 after birth. They gradually decreased from 90% to 10% from age 6 months to 6 years old. While the secondary FBAs were barely detected in neonates, only accounting for 2% of total FBAs, they were gradually elevated to 90% of total FBAs from age 6 months to 6 years old.
CONCLUSIONS: The liver can effectively synthesize primary bile acids 7 days after birth, and fecal primary bile acids tend to be stable after the neonate stage. Secondary bile acids continuously increase along with the maturation of intestinal flora, which reaches a relatively stable level at around 3 years old.
Keywords: Bile Acids and Salts, Child, Cross-Sectional Studies, Liver, Chromatography, Liquid, Feces, Infant, Newborn, Mass Spectrometry
Background
Bile acids (BAs) are primarily synthesized from cholesterol in the hepatocytes of the liver through a series of steps of sterol ring hydroxylation and side chain oxidation [1]. In humans, the primary BAs synthesized in the liver are cholic acid (CA) and chenodeoxycholic acid (CDCA). After being secreted into the intestine, the primary bile acids are metabolized into secondary bile acids, deoxycholic acid (DCA), ursocholic acid (UCA), ursodeoxycholic acid (UDCA), and lithocholic acid (LCA)) by the gut microbiota [2]. Both primary BAs and secondary BAs are conjugated with glycine and taurine and then released in the small intestine to facilitate lipid emulsification, digestion, and absorption. In physiological conditions, the conjugated BAs are decomposed in the upper ileum and colon by colonized bacteria and most of intestinal bile acids are reabsorbed at the terminal ileum for enterohepatic circulation [3,4].
Bile acids solubilize dietary fat and activate pancreatic colipase to lipase for the absorption of dietary lipids, excrete cholesterol, and have an antimicrobial effect [5]. In addition, BAs are also ligands for the farnesoid X receptor (FXR) and the G protein-coupled bile acid receptor 1 (GPBAR1) [6]. A previous study found that the amount of total BAs in feces decreased between birth and 7 days of age and thereafter increased in the neonatal period. The amount of total BAs at 7 days of age was significantly lower than at 6 to 12 months or at 2 to 4 years [7]. The study indicated that the hepatic excretion and transport of BAs were immature in infants who were younger than 3 months of age. Another study found that the level of primary BAs in children’s feces increased at 3 months of age and reached a peak at 6 months of age before decreasing at 12 months of age. The level of secondary BAs started increasing at 6 months of age and dramatically elevated between 6–12 months of age before reaching a peak at 24 months of age. A decrease in level of primary BAs and an increase in secondary BAs coincided with weaning [8]. The study suggested that the change in gut microbiota from infants to adults after weaning and the diet might contribute to the profile of BAs in children.
Neonates and children undergo significant diet changes during development. In addition, the maturation of intestinal flora is an extensive process for many years in early life. It has been suggested that the maturation of liver function and the evolution of intestinal flora during early life stages can significantly affect the excretion of FBAs [9–11]. A significant change in the secretion of fecal BAs in children may indicate the existence of liver and inflammatory bowel diseases, chronic diarrhea, and allergic purpura [12–19]. Therefore, it is critical to understand the profile and changes of FBAs in neonates and children. In this study, we performed a cross-sectional analysis and determined the normal profiling of FBAs in healthy neonates and children in Kunming city, China.
Material and Methods
STUDY DESIGN:
A cross-sectional study was designed to quantify the amount of FBAs in the developmental stages of neonates and children enrolled at the First Affiliated Hospital of Kunming Medical University, Kunming, China from October 2015 to September 2016. Healthy neonates who were born in the Department of Obstetrics and healthy children who underwent regular physical examination in the Department of Pediatrics were randomly sampled. The study was approved by the Ethics Committee of the First Affiliated Hospital of Kunming Medical University. Written consent forms were signed by the parents of neonates and children enrolled before the study began.
STUDY POPULATION:
The study included 2 populations. Neonates were enrolled based on the following inclusion criteria: 1) Born in the Department of Obstetrics of the First Affiliated Hospital of Kunming Medical University between October 2015 and September 2016; 2) Full-term natural birth; 3) Subjected to exclusive breastfeeding. Neonates whose mothers had abnormal results of physical and laboratory examinations or who were administered antibiotics before delivery were excluded. Children were enrolled if they met the following inclusion criteria: 1) age 6 months to 6 years old; 2) Body mass index (BMI) in normal range (18–23); 3) no allergic, gastrointestinal, or immune system diseases, and no family history of genetic and metabolic diseases. Children subjected to antibiotics or microecological agents within 4 weeks before screening were excluded.
DETECTION AND QUANTIFICATION OF FBAS:
Fresh fecal samples were collected and immediately stored at −80°C in a freezer. Before testing, samples were thawed at room temperature and pretreated as previously reported [20]. Briefly, BAs were extracted with 2 mL methanol per gram fecal sample. After vertexing, the mixture was centrifuged for 5 min at 4000 rpm and the supernatant was filtered through a 0.22-micron filter. An aliquot (10 ul) of the extracted sample was loaded onto ZORBAX RRHD 2.1×50×18 micron chromatographic column (Agilent Company, USA) on the 1290 ultra-high performance liquid chromatography (Agilent Company, USA) equipped with the API4000 triple quadrupole mass spectrometer (AB Company, USA). A mixture of 0.1% formic acid and acetonitrile was used as the eluent, and the separation was carried out by step gradient elution at a flow rate of 0.3 ml/min. The mobile phase composition was gradually changed as follows: formic acid-acetonitrile (95: 5, v/v) for 0–5 min, formic acid-acetonitrile (4: 6, v/v) for 5–15 min, formic acid-acetonitrile (5: 95, v/v) for 15–17 min, and formic acid-acetonitrile, 95: 5, v/v for 2 min. For the MS detection, the voltages were set as follows: 1) port voltage was −10 V, 2) collision voltages were set as −77, −30, −30, −30, −30 and −40 V, 3) outlet voltages of the collision chamber were −11, −20, −20, −20, −20 and −20 V.
Standards (Canadian TRC, Toronto Research Chemicals, Toronto, Canada) were used to estimate the retention time and mass charge ratio of FBAs. The retention times of glycochenodeoxycholic acid (GCDCA), ursodeoxycholic acid (UDCA), chenodiol acid (CDCA), deoxycholic acid (DCA, cholic acid (CA), and lithocholic acid (LCA), were 13.41, 11.71, 13.04, 13.30, 12.00, and 14.57 min, respectively, and the m/z GCDCA, UDCA, CDCA, DCAm CA, and LCA were 448.3, 391.2, 391.2, 391.2, 407.2, and 375.3, respectively.
STATISTICAL ANALYSIS:
SPSS 19 (IBM, Armonk, NY, USA) was used for statistical analyses. The normally distributed data were expressed as mean and standard deviation (x±s). Comparisons among multiple groups were performed using one-way ANOVA with SNK-q for post hoc tests. The skewedly distributed data were expressed as median [1st quartile, 3rd quartile]. Kruskal-Wallis analysis was used to test the difference among groups. Mann-Whitney test was used to determine the difference between 2 groups. The correlation between age and BA was analyzed by Spearman rank correlation test. P<0.05 was considered as statistically significant.
Results
FBA concentrations were quantified in neonates at day 3, 7, 14, and 25 after birth. As shown in Table 1, the levels of primary BA, CA, and CDCA were barely detected at day 3. However, they were dramatically increased by more than 100 times at day 7 and reached 103 and 16 pg/g, respectively. While the amount of CDCA was continually increased at day 14 and day 25, the level of CA was decreased at day 14 and day 25. In contrast, the amount of GCDCA reached approximately 4 pg/g at day 3 and then decreased at days 7, 14, and 25. We found that the changes in FBAs were significantly different at day 14 as compared to day 7. Our Spearman analyses indicated that there were significant positive correlations of CA and CDCA with age (r=0.224 and r=0.426, respectively, both
There was a significant increase in secondary FBAs in children aged 6 months to 6 years (Table 4). The amount of DCA was gradually elevated by approximately 4000-fold from age 6 months to 6 years old. The levels of LCA were also increased by 3884-fold from age 6 months to 3–6 years old, while the amount of UDCA was elevated by approximately 6-fold from age 6 months old to 3–6 years old. Spearman analysis showed positive correlations of all 3 FBAs – DCA, LCA, and UDCA – with children’s age (r=0.757 for DCA, r=0.724 for LCA, and r=0.357 for UDCA; all
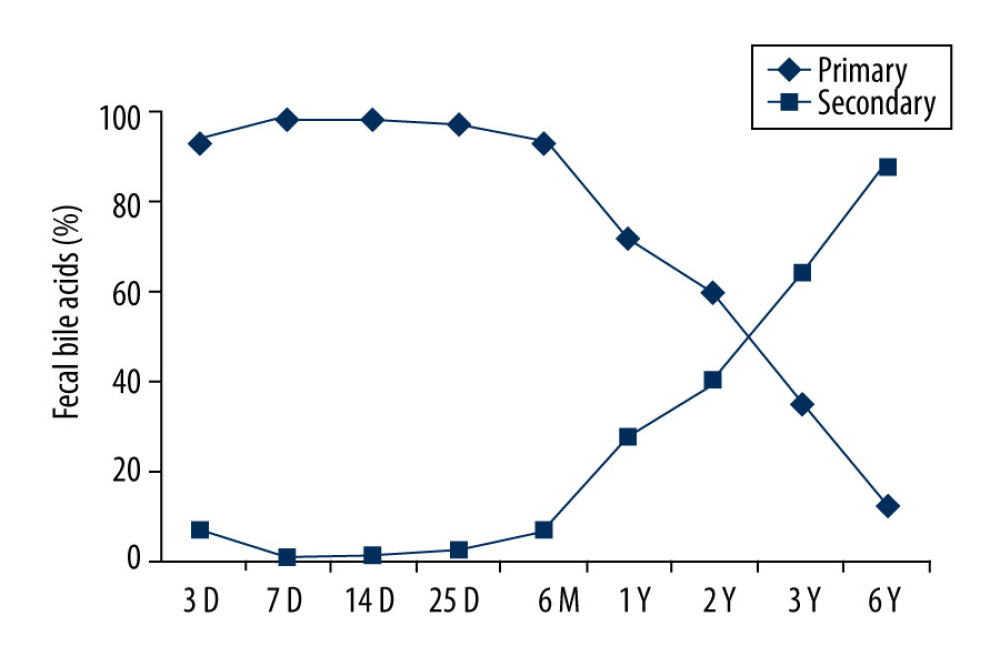
The mean percentage of primary FBAs was increased from 93% of total FBAs to 99% from birth to 7 days old, then gradually decreased to 10% at age 6 years old. In contrast, the percentage of secondary FBAs at 7 days of age decreased from 7% of total FBAs to 1% from birth to 7 days old and then gradually increased to 90% of total FBAs at age 6 years old (Figure 1).
Discussion
In this study, we demonstrated that primary FBAs, CD, and CDCA started to be detectable and were significantly increased from day 3 to day 7 after birth in neonates. The amounts of total BAs in feces increased by 26-fold from day 3 to day 7. The primary FBAs gradually decreased from day 25 to age 6 years old. In contrast, a significant amount of GCDCA was detected at day 3 but decreased to a trace amount at day 7 and afterwards. Primary FBAs appeared to maintain a high level, accounting for 98% of total FBAs, with no significant changes from day 7 to day 25 after birth. They were gradually decreased from 90% to 10% from 6 months to 6 years old. While the secondary FBAs were barely detected in neonates, only accounting for 2% of total FBAs, children’s secondary FBAs were gradually elevated to 90% of total FBAs from 6 months to 6 years old. Our study results agree with previous findings, suggesting the liver maturation at this stage supports initiation of effective primary BA synthesis [21]. At the same time period, GCDCA, the product of conjugation of primary BA and glycine in the liver, remained low in feces of neonates and significantly declined after 7 days of birth, which may be caused by the digestion of GCDCA due to initiated colonization of intestine flora. It has been pointed out that the abundance and variety of fecal flora, including the abundance of Bacteroides, increases with time after birth [22–25], and that Bacteroides in feces can catalyze decomposition of bile acids [26–28]. Our data on total FBAs in neonates disagree with Kimura’s report, in which the amounts of total BAs in feces decreased between birth and 7 days of age [7]. This difference could be explained by the following different quantification approaches: 1) In this study, we only included CA CDCA and GCDCA of primary BAs and DCA, LCA, and UDCA of secondary BAs. We did not quantify the amount of sulfated BA, which accounts for 54% of total FBAs after birth [7]. 2) We did not completely recover the excreted BAs after solvolysis, hydrolysis, and derivatization. 3) We only took the samples on day 3 and 7. We did not take neonate’s samples at day 0 and had no data of the level of total FBAs at day 0. In children aged 6 months to 6 year, their primary FBAs were at similar level as those in neonates after 7 days and no significant difference was detected among different age groups of children. Our results indicate that after the maturation of liver function and very early colocalization of microbiota, the primary FBAs tend to stay stable.
The secondary BAs in feces were mainly DCA and LCA. We found that although trace amounts of secondary FBAs could be detected and were statistically significant in neonates, the general levels of secondary FBAs were very low. Our results suggested that the intestine biota was the major source of fecal secondary BAs. It has been reported that Clostridia can catalyze the conversion of primary BA 7α-hydroxydeoxygenation to secondary Bas [26–28]. Since the abundance and diversity of intestinal flora were both low at the early stage of the neonates and the
Although we performed a cross-sectional study in 238 healthy neonates and children in the First Affiliated Hospital of Kunming Medical University, China and provided a developmental pattern of fecal bile acids that could be used to determine the abnormal levels of FBAs associated with metabolic diseases, there were certain limitations in this study. First, the sample size was not big enough to estimate a normal range of FBAs in neonates and children, but we could detect relative trends of changes. Second, the time points assessed were not enough to illustrate the detailed variations of FBAs, although a general trend of change could be observed. More time points are needed in future studies. Third, we only evaluated that levels of FBAs and did not consider numerous factors such as the liver function and composition intestinal flora that could affect the levels of FBAs in children. Fourth, this study only provides information about several qualitative changes with age in neonates and children and lacked data on the quantitative fecal excretion of bile acids that reflects BA synthesis. In future research, GC-MS analysis should be used to obtain a complete recovery of BA excretion after solvolysis, hydrolysis, and derivatization. It is also critical to evaluate the level of sulfated BA in FBAs.
Conclusions
In summary, we performed a cross-sectional study in healthy neonates and children in the First Affiliated Hospital of Kunming Medical University, China and found a developmental pattern of fecal bile acids that could be used determine abnormal levels of FBAs associated with metabolic diseases. We found that the liver could effectively synthesize primary BAs 7 days after birth, and fecal primary bile acids tend to be stable after the neonate stage. While the fecal secondary BAs continuously increased along with the maturation of intestinal flora, they increased until the age of 3 years old and then remained stable. There was an increase in DCA but a decrease in both LCA and UDCA.
References
1. Hundt M, Basit H, John S: Physiology, bile secretion, 2020, StatPearls, Treasure Island (FL)
2. Hofmann AF, Hagey LR, Key discoveries in bile acid chemistry and biology and their clinical applications: History of the last eight decades: J Lipid Res, 2014; 55; 1553-95
3. Epstein D, Mistry K, Whitelaw A, The effect of physiological concentrations of bile acids on in vitro growth of Mycobacterium tuberculosis: S Afr Med J, 2012; 102; 522-24
4. Li J, Li T, Bile acid receptors link nutrient sensing to metabolic regulation: Liver Res, 2017; 1; 17-25
5. Hofmann AF, Hagey LR, Krasowski MD, Bile salts of vertebrates: Structural variation and possible evolutionary significance: J Lipid Res, 2010; 51; 226-46
6. Fiorucci S, Mencarelli A, Palladino G, Cipriani S, Bile-acid-activated receptors: Targeting TGR5 and farnesoid-X-receptor in lipid and glucose disorders: Trends Pharmacol Sci, 2009; 30; 570-80
7. Kimura A, Yamakawa R, Ushijima K, Fetal bile acid metabolism during infancy. Analysis of 1 beta-hydroxylated bile acids in urine, meconium and feces: Hepatology, 1994; 20; 819-24
8. Khine WWT, Rahayu ES, See TY, Indonesian children fecal microbiome from birth until weaning was different from microbiomes of their mothers: Gut Microbes, 2020; 12; 1761240
9. Bäckhed F, Roswall J, Peng Y, Dynamics and stabilization of the human gut microbiome during the first year of life: Cell Host Microbe, 2015; 17; 852
10. Collado MC, Rautava S, Aakko J, Human gut colonisation may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid: Sci Rep, 2016; 6; 23129
11. Chong CYL, Bloomfield FH, O’Sullivan JM, Factors affecting gastrointestinal microbiome development in neonates: Nutrients, 2018; 10; 274
12. Asgharpour A, Kumar D, Sanyal A, Bile acids: Emerging role in management of liver diseases: Hepatol Int, 2015; 9; 527-33
13. Corte-Real J, Bohn T, Interaction of divalent minerals with liposoluble nutrients and phytochemicals during digestion and influences on their bioavailability – a review: Food Chem, 2018; 252; 285-93
14. Di Ciaula A, Garruti G, Lunardi Baccetto R, Bile acid physiology: Ann Hepatol, 2017; 16; s4-14
15. Hamer HM, De Preter V, Windey K, Verbeke K, Functional analysis of colonic bacterial metabolism: Relevant to health?: Am J Physiol Gastrointest Liver Physiol, 2012; 302; G1-9
16. Hu HW, Duan J, Zhao BSignificance of detection of biomarker fecal bile acids in the diagnosis and treatment of childhood Henoch-Schönlein purpura: Zhongguo Dang Dai Er Ke Za Zhi, 2016; 18; 517-21 [in Chinese]
17. Li T, Apte U, Bile acid metabolism and signaling in cholestasis, inflammation, and cancer: Adv Pharmacol, 2015; 74; 263-302
18. Long SL, Gahan CGM, Joyce SA, Interactions between gut bacteria and bile in health and disease: Mol Aspects Med, 2017; 56; 54-65
19. van de Wiel SMW, de Waart DR, Oude Elferink RPJ, van de Graaf SFJ, Intestinal farnesoid X receptor activation by pharmacologic inhibition of the organic solute transporter α–β: Cell Mol Gastroenterol Hepatol, 2018; 5; 223-37
20. Kakiyama G, Muto A, Takei H, A simple and accurate HPLC method for fecal bile acid profile in healthy and cirrhotic subjects: Validation by GC-MS and LC-MS: J Lipid Res, 2014; 55; 978-90
21. Vaz FM, Ferdinandusse S, Bile acid analysis in human disorders of bile acid biosynthesis: Mol Aspects Med, 2017; 56; 10-24
22. Yassour M, Jason E, Hogstrom LJ, Strain-level analysis of mother-to-child bacterial transmission during the first few months of life: Cell Host Microbe, 2018; 24; 146-54.e4
23. Lee E, Kim BJ, Kang MJ, Dynamics of gut microbiota according to the delivery mode in healthy Korean infants: Allergy Asthma Immunol Res, 2016; 8; 471-77
24. Rutayisire E, Huang K, Liu Y, Tao F, The mode of delivery affects the diversity and colonization pattern of the gut microbiota during the first year of infants’ life: A systematic review: BMC Gastroenterol, 2016; 16; 86
25. Nagpal R, Tsuji H, Takahashi T, Ontogenesis of the gut microbiota composition in healthy, full-term, vaginally born and breast-fed infants over the first 3 years of life: A quantitative bird’s-eye view: Front Microbiol, 2017; 8; 1388
26. Eggert T, Bakonyi D, Hummel W, Enzymatic routes for the synthesis of ursodeoxycholic acid: J Biotechnol, 2014; 191; 11-21
27. Weingarden AR, Chen C, Bobr A: Am J Physiol Gastrointest Liver Physiol, 2014; 306; G310-19
28. Murakami M, Iwamoto J, Honda A, Detection of gut dysbiosis due to reduced clostridium subcluster XIVa using the fecal or serum bile acid profile: Inflamm Bowel Dis, 2018; 24; 1035-44
29. Cheng J, Ringel-Kulka T, Heikamp-de Jong I, Discordant temporal development of bacterial phyla and the emergence of core in the fecal microbiota of young children: ISME J, 2016; 10; 1002-14
30. Yatsunenko T, Rey FE, Manary MJ, Human gut microbiome viewed across age and geography: Nature, 2012; 486; 222-27
31. Ringel-Kulka T, Cheng J, Ringel Y, Intestinal microbiota in healthy U.S. young children and adults – a high throughput microarray analysis: PLoS One, 2013; 8; e64315
Tables
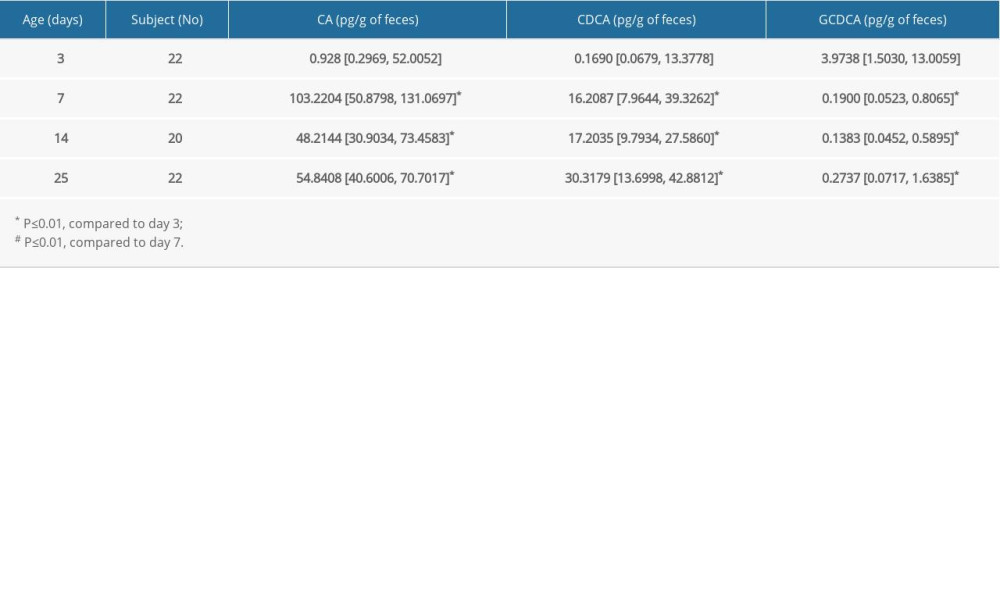 Table 1. Changes of primary FBAs in neonates at different days after birth.
Table 1. Changes of primary FBAs in neonates at different days after birth.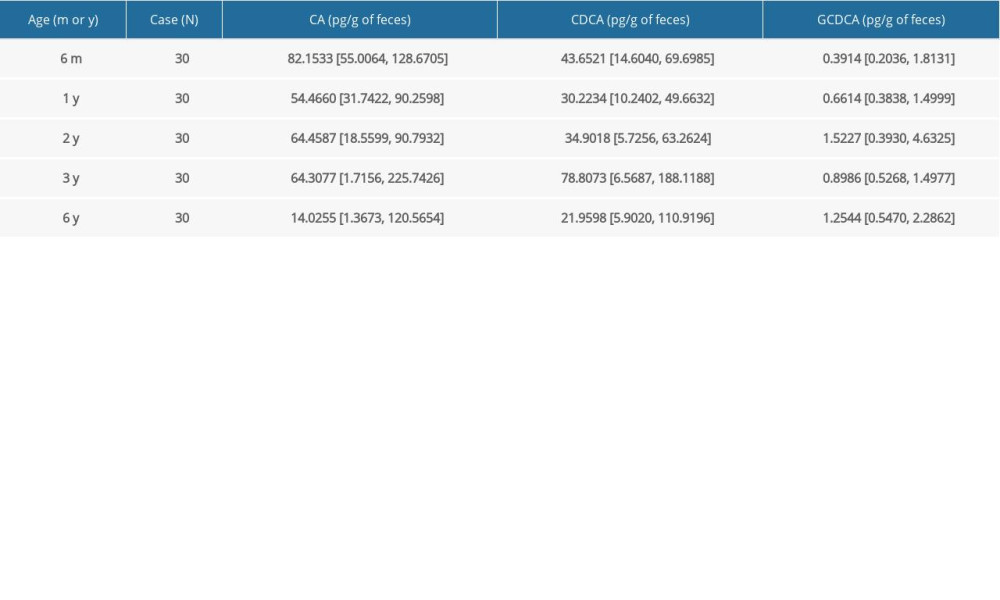 Table 2. Changes of primary FBAs in children at different ages.
Table 2. Changes of primary FBAs in children at different ages.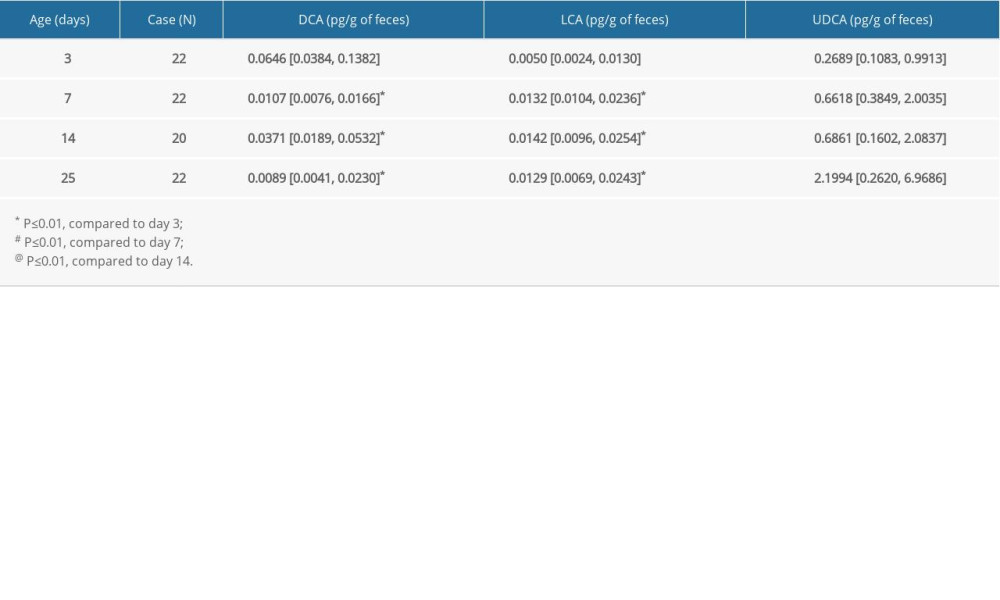 Table 3. Changes of secondary FBAs in neonates at different days after birth.
Table 3. Changes of secondary FBAs in neonates at different days after birth.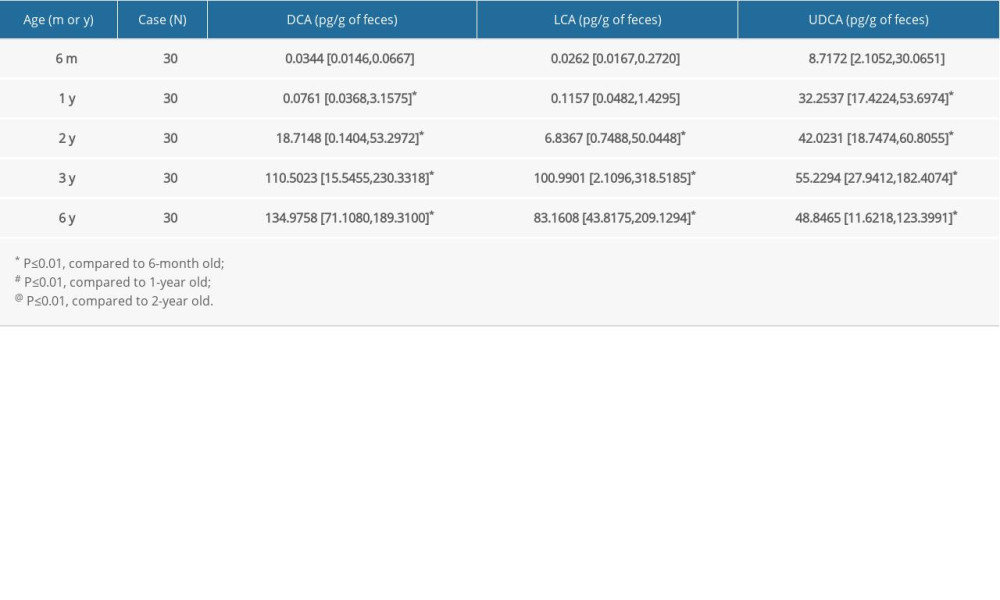 Table 4. Changes of secondary FBAs in children at different ages.
Table 4. Changes of secondary FBAs in children at different ages. Table 1. Changes of primary FBAs in neonates at different days after birth.
Table 1. Changes of primary FBAs in neonates at different days after birth. Table 2. Changes of primary FBAs in children at different ages.
Table 2. Changes of primary FBAs in children at different ages. Table 3. Changes of secondary FBAs in neonates at different days after birth.
Table 3. Changes of secondary FBAs in neonates at different days after birth. Table 4. Changes of secondary FBAs in children at different ages.
Table 4. Changes of secondary FBAs in children at different ages. In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952