02 February 2021: Review Article
Intravenous Infusion of Lidocaine Can Accelerate Postoperative Early Recovery in Patients Undergoing Surgery for Obstructive Sleep Apnea
Chenglan Xie1ABCDEFG*, Qiao Wang1BF, De Huai2ABCDEFGDOI: 10.12659/MSM.926990
Med Sci Monit 2021; 27:e926990
Abstract
ABSTRACT: Obstructive sleep apnea (OSA) is defined by intermittent and recurrent episodes of partial or complete obstruction of the upper airway during sleep. Intermittent and recurrent hypoxia/reoxygenation is the main pathophysiological mechanism of OSA. Its consequences include systemic inflammation, activation of the sympathetic nervous system, and release of oxygen free radicals. Infusion of intravenous (IV) lidocaine has anti-inflammatory, antihyperalgesic, and analgesic properties, supporting its use as an anesthetic adjuvant. Lidocaine can reduce nociception and/or cardiovascular responses to surgical stress, as well as postoperative pain and/or analgesic requirements. Because of the high prevalence of OSA in obese patients, the use of opioids to manage postoperative pain in that population is often accompanied by the development of adverse respiratory events, such as hypoventilation and hypoxemia. IV infusion of lidocaine has been shown to enhance the quality of early recovery after laparoscopic bariatric and upper airway surgery. However, limited evidence exists regarding its use in patients undergoing surgery for OSA. In addition, whether IV infusion of lidocaine can improve postoperative early recovery in patients undergoing surgery for OSA remains unknown. Therefore, we hypothesized that IV infusion of lidocaine can improve postoperative early recovery in patients undergoing surgery for OSA. Perioperative infusion also may be a promising analgesic adjunct to enhanced recovery after surgery (ERAS) protocols.
Keywords: Infusions, Intravenous, Lidocaine, Sleep apnea, obstructive, Analgesics, Analgesics, Opioid, Anesthesia Recovery Period, Anesthetics, Local, Pain Measurement, Pain, Postoperative, Polysomnography, Postoperative Complications, Postoperative Period
Background
The incidence of obstructive sleep apnea (OSA), defined as intermittent and recurrent episodes of partial or complete obstruction of the upper airway during sleep, has been rising throughout the world [1,2]. Untreated OSA is associated with decreased quality of life, increased risk of cardiovascular disease and all-cause mortality, and impaired cognitive function [3]. Continuous positive airway pressure (CPAP) is the first-line therapy for OSA, but it is not always tolerated. Surgeries indicated for OSA have varying degrees of efficacy. Uvulopalatopharyngoplasty (UPPP) is the most common surgical treatment for OSA. Adenotonsillectomy is the first-line surgical therapy for children with OSA. Aside from tracheostomy, maxillomandibular advancement is the most successful surgical intervention for OSA, with outcomes similar to those with CPAP [4].
Pain after surgical procedures for OSA is classified as moderate to severe. Postoperative pain is a noxious stimulus produced by tissue damage caused by surgery, which can result in problems such as sleep disturbance, respiratory adverse effects, cardiovascular adverse effects, impaired gastrointestinal motility, and poor wound healing. In addition to affecting patient recovery, postoperative pain can reduce patient satisfaction and quality of life after surgery. Appropriate control of pain can reduce morbidity, improve surgical results, and decrease hospital costs. Opioids are the mainstay analgesics for moderate as well as severe acute pain. However, use of these drugs can increase incidence of postoperative complications, including vomiting, respiratory depression, excessive sedation, pruritus, and slowing of gastrointestinal function, as well as urinary retention. Recent data also suggest that extensive use of opioids is associated with hyperalgesia [5]. These opioid-related adverse events (AEs) prevent smooth postoperative early recovery and prolong hospital stays.
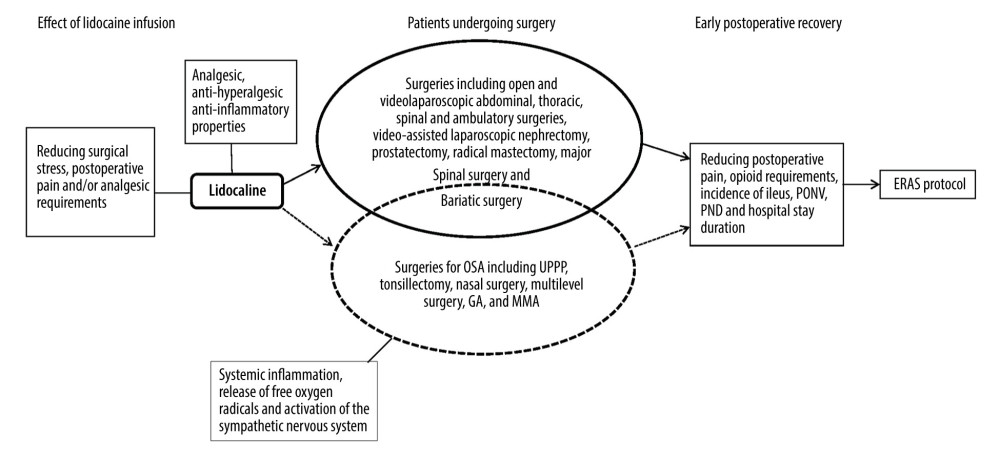
Patients with OSA are very sensitive to the respiratory depression caused by opioids [6]. An alternative to opioids is intravenous (IV) infusion of lidocaine, which has antihyperalgesic, anti-inflammatory, and analgesic characteristics that reduce nociception and/or cardiovascular responses to surgical stress, as well as postoperative pain and/or analgesic requirements (Figure 1). Perioperative infusion of lidocaine may be a useful adjunct to enhanced recovery after surgery (ERAS) protocols [7–10]. Although perioperative infusion of the drug also has a significant impact on reducing intraoperative hemodynamic instability and anesthetic requirements, our major focus here is perioperative recovery, including reduction in postoperative pain, opioid-related AEs, and postoperative neurocognitive dysfunction (PND), and shortening of hospital stays. Few studies have examined the effect of IV infusion of lidocaine for OSA surgery. There are only 3 studies available on the topic [11–13]. The first study showed no benefit in adults undergoing tonsillectomy. Another study, despite clearly being underpowered, showed that IV infusion of lidocaine can improve the quality of postoperative early recovery after upper airway surgery, including UPPP. The third study showed that IV infusion of lidocaine decreased postoperative vomiting in children undergoing tonsillectomy. However, it remains unknown whether IV infusion of lidocaine also can enhance postoperative early recovery in patients undergoing surgery for OSA.
Hypothesis
Clinical applications of perioperative infusion of lidocaine have been studied in many trials in patients undergoing abdominal procedures, ambulatory procedures, and other types of surgery [12–41]. In these surgical procedures, lidocaine infusion has been shown to reduce postoperative pain, opioid requirements, ileus duration, and incidence of postoperative nausea and vomiting (PONV) and PND, and shorten hospital stays. IV infusion of lidocaine has been shown to enhance the quality of early recovery after laparoscopic bariatric and upper airway surgery [13,17,42,43]. However, a single study showed that IV lidocaine significantly decreased postoperative opioid consumption, but when used after bariatric surgery, it was clinically irrelevant [44]. Continuous ketamine and remifentanil infusion plus nefopam, paracetamol, and parecoxib before wound closure may partly explain those results. Because of the high incidence of OSA in patients who are obese, the use of opioids in this population is often accompanied by the development of adverse respiratory events such as hypoventilation and hypoxemia. Perioperative infusion of lidocaine may result in a postoperative benefit to patients with OSA, which may be useful for ERAS protocols. Based on the studies described above, we hypothesized that IV infusion of lidocaine can improve postoperative early recovery in patients undergoing surgery for OSA (Figure 1).
Hypothesis Evaluation
CLINICAL APPLICATIONS:
Meta-analyses and systematic reviews have shown that perioperative infusion of lidocaine is effective in reducing postoperative pain, opioid requirements, and duration of ileus, and shortening hospital stays [45–56]. However, an updated review by the Cochrane Collaboration, based on new analysis techniques, resulted in a substantial downgrade in conclusions about perioperative use of IV lidocaine [57]. The quality of evidence on which to base a conclusion about the benefit of the drug’s use in this setting was limited, given the heterogeneity of the studies included in the analysis [58].
Infusion of lidocaine has been shown to be of benefit in open and video-assisted laparoscopic abdominal, thoracic, spinal, and ambulatory surgeries [14–21,33,36,38–42,59–64]. Therefore, it can be used in postsurgical recovery protocols, specifically ERAS protocols. Other studies have found evidence of benefit for infusion of lidocaine for conditions involving acute and chronic pain, including radical prostatectomy, open nephrectomy, major spinal surgery, and radical mastectomy [22,30,31,37,52,59,65–67]. In bariatric surgery and upper airway surgery, infusion of lidocaine reduced opioid consumption, which was associated with improved recovery scores [13,17,42]. Furthermore, OSA is more prevalent in individuals who are morbidly obese than in the general population. Perioperative infusion of lidocaine could be beneficial as an alternative for patients with obesity and OSA because they are fairly sensitive to the respiratory-depressant effects of opioids.
Besides improving analgesia, perioperative infusion of lidocaine decreases the incidence of PONV [12,28,35,39,46,48,50]. This effect has been attributed to an increase in intestinal motility and/or a reduction in postoperative pain and opioid use. Other studies suggest that the reduction is not related to an opioid-sparing effect [12]. Moreover, intraoperative administration of lidocaine decreases the occurrence of early PND in patients undergoing coronary artery bypass and spinal surgery, perhaps because of its neuroprotective effects [21,25,68].
Adenotonsillectomy is the first-line surgical therapy for children with OSA who do not have craniofacial anomalies, and tonsillectomy is one of the surgical therapies for adults with OSA. However, IV infusion of lidocaine had no beneficial effect on postoperative pain in adults undergoing tonsillectomy. The lack of a statistically significant analgesic effect in the abovementioned study might have been due to its small sample size [11]. The study of children undergoing tonsillectomy with and without adenoidectomy showed no difference in terms of opioid consumption during the intraoperative and postoperative periods. The absence of a difference, however, could be explained by the inadequate power of the study to find differences in terms of pain outcomes, because the sample size calculation was based on postoperative vomiting [12]. Our study showed that IV infusion of lidocaine can improve the quality of early postoperative recovery from upper airway surgery, including UPPP [13]. Even if the sample size was calculated based on postoperative recovery quality, the number of patients with OSA was clearly underpowered to show statistical differences.
SAFETY AND PROTOCOL FOR ADMINISTRATION:
Toxicity from perioperative lidocaine infusion is extremely uncommon. Perioral paresthesia, metallic taste, slurred speech, diplopia, light-headedness, tinnitus, confusion, agitation, muscular spasms, and seizures have been reported with plasma concentrations of lidocaine higher than 5 to 8 μg/mL [7,8,69]. A 55-year-old man with well-controlled HIV who was on highly active antiretroviral therapy was given 1 mg/kg/h of IV lidocaine for postoperative pain. He experienced tachycardia, hypertension, and oxygen desaturation on postoperative Day 2 because of a significant pharmacokinetic interaction between the lidocaine and his HIV medications [69]. Carabalona et al measured serum concentrations of lidocaine in patients undergoing bariatric surgery. Lidocaine dosage was based on adjusted body weight (ideal body weight+0.4×[current bodyweight–ideal body weight]). Patients received a 1.5 mg/kg IV bolus of lidocaine over 10 minutes (with a maximal dose of 100 mg) after anesthesia induction, followed by continuous infusion of 2 mg/kg/h throughout the operation. The rate of lidocaine infusion was reduced to 1 mg/kg/h until discharge from the recovery room. The study showed that serum concentrations of lidocaine never exceeded 5 μg/mL and the median serum concentration was 1.45 μg/mL (range, 0.98–1.88 μg/mL) during bariatric surgery [70]. In addition, no ideal protocol has been established for administration of systemic lidocaine. When lidocaine is administered as a 1 mg/kg bolus following continuous infusion of 2 mg/kg/h, plasma concentrations are well below the toxic level (5 μg/mL) even after 1 day [9]. Thus, recommended lidocaine doses in the perioperative period are 1 to 2 mg/kg as an initial bolus, followed by continuous infusion of 1 to 2 mg/kg/h and infusion termination at the end of the post-anesthesia care unit (PACU) stay because of the lack of clear benefit of prolonged infusion beyond that [7,9,46,71]. Monitoring of lidocaine levels also can be considered in patients who are at increased risk of lidocaine toxicity, have abnormal kidney or liver functions, or who cannot communicate about symptoms of lidocaine toxicity. Treatment of systemic lidocaine toxicity has been described and involves combining measures for symptom control, such as oxygen and benzodiazepines, with lipid emulsion injection [72].
MECHANISMS OF ACTION:
Lidocaine is an amide-based local anesthetic with antihyperalgesic, analgesic, and anti-inflammatory characteristics. The drug’s analgesic effects are considered to be mediated via suppressing spontaneous impulses generated from injured nerve fibers as well as the proximal dorsal root ganglion. This happens through inhibiting sodium channels and G-protein-coupled and N-methyl-D-aspartate receptors [7,8,73,74]. Many studies have demonstrated that perioperative lidocaine infusion is very efficient while underscoring that its clinical efficiency may differ, based on the surgical process. Nevertheless, no apparent mechanistic rationale exists for why lidocaine efficiency would vary from what has been observed in, for example, gastrointestinal surgery. Blood levels of inflammatory mediators have been shown to be higher after gastrointestinal surgery than after less invasive procedures. Therefore, perioperative infusion of lidocaine would be less efficient for processes, including abdominal hysterectomy, total hip arthroplasty, and laparoscopic renal surgery, which are probably less invasive because they are associated with a relatively low degree of inflammation [9,75–77]. Therefore, the difference in effectiveness of lidocaine may be due, in part, to the drug’s anti-inflammatory properties. Systemic lidocaine significantly attenuates plasma levels of complement and proinflammatory cytokines such as interleukin (IL)-6, IL-8, and IL-1 receptor antagonist (IL-1RA) at the end of surgery and up to 72 hours after the procedure [62,78]. The anti-inflammatory effects of lidocaine are attributable to neural transmission blockade with tissue injury, which leads to neurogenic inflammation attenuation and activation of the intrinsic anti-inflammatory pathway. Lidocaine inhibits migration and release of lysosomal enzymes and granulocytes, which leads to reduced release of anti-inflammatory and proinflammatory cytokines, thereby promoting peripheral and central sensitization suppression and resulting in the hypothetical antihyperalgesic effect [45].
Besides its anti-inflammatory properties, lidocaine is a scavenger of reactive oxygen species and can inhibit the sympathetic response to surgery [73,79,80]. The pathophysiological consequences of intermittent hypoxia in OSA are systemic inflammation, release of oxygen free radicals, and activation of the sympathetic nervous system [6]. Therefore, perioperative infusion of lidocaine may be effective in improving postoperative early recovery in patients with OSA.
Conclusions
Perioperative infusion of lidocaine can improve postoperative early recovery by reducing postoperative pain and complications, shortening hospital stays, and enhancing patient satisfaction without compromising safety. Thus, it can be considered in ERAS protocols for patients undergoing OSA surgery. More studies are necessary to establish the optimum dose, timing, and duration of lidocaine infusion in this setting.
References
1. Bandla P, Brooks LJ, Trimarchi T, Obstructive sleep apnea syndrome in children: Anesthesiol Clin North Am, 2005; 23(3); 535-49
2. Payen JF, Jaber S, Levy P, Obstructive sleep-apnoea syndrome in adult and its perioperative management: Ann Fr D Anesth Et De Reanim, 2010; 29(11); 787-92
3. Vasu TS, Grewal R, Doghramji K, Obstructive sleep apnea syndrome and perioperative complications: A systematic review of the literature: J Clin Sleep Med, 2012; 8(2); 199-207
4. Dicus Brookes CC, Boyd SB, Controversies in obstructive sleep apnea surgery: Oral Maxillofac Surg Clin North Am, 2017; 29(4); 503-13
5. Yi P, Pryzbylkowski P, Opioid iInduced hyperalgesia: Pain Med, 2015; 16(Suppl 1); S32-36
6. Gross JB, Apfelbaum JL, Caplan RA, An updated report by the American Society of Anesthesiologists Task Force on Perioperative Management of patients with obstructive sleep apnea: Anesthesiology, 2014; 120(2); 268-86
7. Estebe JP, Intravenous lidocaine: Best Pract Res Clin Anaesthesiol, 2017; 31(4); 513-21
8. Soto G, Naranjo Gonzalez M, Calero F, Intravenous lidocaine infusion: Rev Esp Anestesiol Reanim, 2018; 65(5); 269-74
9. Dunn LK, Durieux ME, Perioperative use of intravenous lidocaine: Anesthesiology, 2017; 126(4); 729-37
10. Beaussier M, Delbos A, Maurice-Szamburski A, Perioperative use of intravenous lidocaine: Drugs, 2018; 78(12); 1229-46
11. Striebel HW, Klettke U, Is intravenous lidocaine infusion suitable for postoperative pain management?: Schmerz, 1992; 6(4); 245-50
12. Echevarría GC, Altermatt FR, Paredes S, A randomised controlled trial: Eur J Anaesthesiol, 2018; 35(5); 343-48
13. Wang Q, Ding X, Huai D, Effect of intravenous lidocaine infusion on postoperative early recovery quality in upper airway surgery: Laryngoscope, 2020 [Online ahead of print]
14. Baral BK, Bhattarai BK, Rahman TR, Perioperative intravenous lidocaine infusion on postoperative pain relief in patients undergoing upper abdominal surgery: Nepal Med Coll J, 2010; 12(4); 215-20
15. Ahn E, Kang H, Choi GJ, A prospective, randomized, double-blind, placebo-controlled study: Int Surg, 2015; 100(3); 394-401
16. Kim TH, Kang H, Choi YS, Pre- and intraoperative lidocaine injection for preemptive analgesics in laparoscopic gastrectomy: A prospective, randomized, double-blind, placebo-controlled study: J Laparoendosc Adv Surg Tech A, 2013; 23(8); 663-68
17. De Oliveira GS, Duncan K, Fitzgerald P, A randomized double-blinded placebo-controlled trial: Obes Surg, 2014; 24(2); 212-18
18. De Oliveira GS, Fitzgerald P, Streicher LF, Systemic lidocaine to improve postoperative quality of recovery after ambulatory laparoscopic surgery: Anesth Analg, 2012; 115(2); 262-67
19. Kaba A, Laurent SR, Detroz BJ, Intravenous lidocaine infusion facilitates acute rehabilitation after laparoscopic colectomy: Anesthesiology, 2007; 106(1); 11-18
20. Cui W, Li Y, Li S, Wang R, Systemic administration of lidocaine reduces morphine requirements and postoperative pain of patients undergoing thoracic surgery after propofol-remifentanil-based anaesthesia: Eur J Anaesthesiol, 2010; 27(1); 41-46
21. Chen K, Wei P, Zheng Q, Neuroprotective effects of intravenous lidocaine on early postoperative cognitive dysfunction in elderly patients following spine surgery: Med Sci Monit, 2015; 21; 1402-7
22. Groudine SB, Fisher HA, Kaufman RP, Intravenous lidocaine speeds the return of bowel function, decreases postoperative pain, and shortens hospital stay in patients undergoing radical retropubic prostatectomy: Anesth Analg, 1998; 86(2); 235-39
23. Choi GJ, Kang H, Ahn EJ, A prospective, randomized, double-blind, placebo-controlled trial: World J Surg, 2016; 40(12); 2941-47
24. Tauzin-Fin P, Bernard O, Sesay M, Benefits of intravenous lidocaine on post-operative pain and acute rehabilitation after laparoscopic nephrectomy: J Anaesthesiol Clin Pharmacol, 2014; 30(3); 366-72
25. Wang D, Wu X, Li J, The effect of lidocaine on early postoperative cognitive dysfunction after coronary artery bypass surgery: Anesth Analg, 2002; 95(5); 1134-41
26. Zhang H, Yang L, Zhu X, Association between intraoperative intravenous lidocaine infusion and survival in patients undergoing pancreatectomy for pancreatic cancer: A retrospective study: Br J Anaesth, 2020; 125(2); 141-48
27. Kim DH, Park JY, Yu J, A randomized, double-blind, controlled trial: Anesth Analg, 2020; 131(1); 220-27
28. Ghimire A, Subedi A, Bhattarai B, The effect of intraoperative lidocaine infusion on opioid consumption and pain after totally extraperitoneal laparoscopic inguinal hernioplasty: A randomized controlled trial: BMC Anesthesiol, 2020; 20(1); 137
29. Chen M, Lu Y, Liu H, A randomized, double-blinded, controlled study: BMC Anesthesiol, 2020; 20(1); 132
30. Batko I, Kościelniak-Merak B, Tomasik PJ, A prospective, randomized, double-blind study: Pharmacol Rep, 2020; 72(3); 744-55
31. Batko I, Kościelniak-Merak B, Tomasik PJ, Lidocaine reduces sevoflurane consumption and improves recovery profile in children undergoing major spine surgery: Med Sci Monit, 2020; 26; e919971
32. Moeen SM, Moeen AM, Usage of intravenous lidocaine infusion with enhanced recovery pathway in patients scheduled for open radical cystectomy: A randomized trial: Pain Physician, 2019; 22(2); E71-80
33. Ibrahim A, Aly M, Farrag W, Effect of intravenous lidocaine infusion on long-term postoperative pain after spinal fusion surgery: Medicine (Baltimore), 2018; 97(13); e0229
34. Ates İ, Aydin ME, Ahiskalioglu A, A prospective, randomized, double-blind study: Eur Arch Otorhinolaryngol, 2020; 277(4); 1095-100
35. Wang T, Liu H, Sun JH, Efficacy of intravenous lidocaine in improving post-operative nausea, vomiting and early recovery after laparoscopic gynaecological surgery: Exp Ther Med, 2019; 17(6); 4723-29
36. Koshyari HS, Asthana V, Agrawal S, Evaluation of lignocaine infusion on recovery profile, quality of recovery, and postoperative analgesia in patients undergoing total abdominal hysterectomy: J Anaesthesiol Clin Pharmacol, 2019; 35(4); 528-32
37. Nakhli MS, Kahloul M, Guizani T, Intravenous lidocaine as adjuvant to general anesthesia in renal surgery: Libyan J Med, 2018; 13(1); 1433418
38. Murthy TK, Kumar PV, Effect of perioperative intravenous lignocaine infusion on haemodynamic responses and postoperative analgesia in laparoscopic cholecystectomy surgeries: Anesth Pain Med, 2018; 8(2); e63490
39. Liu ZJ, Zhang LY, Zheng XGEffects of continuous intravenous intraoperative lidocaine infusion on opioids consumption and postoperative recovery in patients undergoing video-assisted thoracoscopic lobectomy: Zhongguo Yi Xue Ke Xue Yuan Xue Bao, 2018; 40(2); 163-69 [in Chinese]
40. Ho MLJ, Kerr SJ, Stevens J, Intravenous lidocaine infusions for 48 hours in open colorectal surgery: A prospective, randomized, double-blinded, placebo-controlled trial: Korean J Anesthesiol, 2018; 71(1); 57-65
41. Dewinter G, Coppens S, Van de Velde M, A prospective, randomized, double-blind controlled clinical trial: Ann Surg, 2018; 268(5); 769-75
42. Sakata RK, de Lima RC, Valadão JA, Randomized, double-blind study of the effect of intraoperative Intravenous lidocaine on the opioid consumption and criteria for hospital discharge after bariatric surgery: Obes Surg, 2020; 30(4); 1189-93
43. Gupta C, Valecha UK, Singh SP, A comparative randomised study in bariatric surgical patients: Indian J Anaesth, 2020; 64(1); 31-36
44. Plass F, Nicolle C, Zamparini M, A prospective, randomised, blinded, placebo-controlled study: Aneasthesia, 2020 [Online ahead of print]
45. McCarthy GC, Megalla SA, Habib AS, Impact of intravenous lidocaine infusion on postoperative analgesia and recovery from surgery: A systematic review of randomized controlled trials: Drugs, 2010; 70(9); 1149-63
46. Marret E, Rolin M, Beaussier M, Meta-analysis of intravenous lidocaine and postoperative recovery after abdominal surgery: Br J Surg, 2008; 95(11); 1331-38
47. Sun Y, Li T, Wang N, Meta-analysis of intravenous lidocaine and postoperative recovery after abdominal surgery: Dis Colon Rectum, 2012; 55(11); 1183-94
48. Vigneault L, Turgeon AF, Cote D, A meta-analysis of randomized controlled trials: Can J Anaesth, 2011; 58(1); 22-37
49. Rollins KE, Javanmard-Emamghissi H, Scott MJ, A meta-analysis of randomised controlled trials: Eur J Anaesthesiol, 2020; 37(8); 659-70
50. Nakajima D, Kawakami H, Mihara T, Effectiveness of intravenous lidocaine in preventing postoperative nausea and vomiting in pediatric patients: A systematic review and meta-analysis: PLoS One, 2020; 15(1); e0227904
51. Zhao JB, Li YL, Wang YM, A meta-analysis of randomized controlled trials: Medicine (Baltimore), 2018; 97(5); e9771
52. Chang YC, Liu CL, Liu TP, Effect of perioperative intravenous lidocaine infusion on acute and chronic pain after breast surgery: A meta-analysis of randomized controlled trials: Pain Pract, 2017; 17(3); 336-43
53. Kranke P, Jokinen J, Pace NL, Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery: Cochrane Database Syst Rev, 2015(7); CD009642
54. Weibel S, Jokinen J, Pace NL, Efficacy and safety of intravenous lidocaine for postoperative analgesia and recovery after surgery: A systematic review with trial sequential analysis: Br J Anaesth, 2016; 116(6); 770-83
55. Cooke C, Kennedy ED, Foo I, Meta-analysis of the effect of perioperative intravenous lidocaine on return of gastrointestinal function after colorectal surgery: Tech Coloproctol, 2019; 23(1); 15-24
56. Li J, Wang G, Xu W, A meta-analysis from randomized controlled trials: Int J Surg, 2018; 50; 137-45
57. Weibel S, Jelting Y, Pace NL, Continuous intravenous perioperative lidocaine infusion for postoperative pain and recovery in adults: Cochrane Database Syst Rev, 2018; 6(6); CD009642
58. Paterson HM, Continuous intravenous lidocaine infusion for postoperative pain and recovery in adults: Tech Coloproctol, 2019; 23(1); 69-71
59. Farag E, Ghobrial M, Sessler DI, Effect of perioperative intravenous lidocaine administration on pain, opioid consumption, and quality of life after complex spine surgery: Anesthesiology, 2013; 119(4); 932-40
60. Grady P, Clark N, Lenahan J, Effect of intraoperative intravenous lidocaine on postoperative pain and return of bowel function after laparoscopic abdominal gynecologic procedures: AANA J, 2012; 80(4); 282-88
61. McKay A, Gottschalk A, Ploppa A, Systemic lidocaine decreased the perioperative opioid analgesic requirements but failed to reduce discharge time after ambulatory surgery: Anesth Analg, 2009; 109(6); 1805-8
62. Herroeder S, Pecher S, Schonherr ME, A double-blinded, randomized, placebo-controlled trial: Ann Surg, 2007; 246(2); 192-200
63. Kang JG, Kim MH, Kim EH, Intraoperative intravenous lidocaine reduces hospital length of stay following open gastrectomy for stomach cancer in men: J Clin Anesth, 2012; 24(6); 465-70
64. Forster C, Vanhaudenhuyse A, Gast P, Arandomised placebo-controlled study: Br J Anaesth, 2018; 121(5); 1059-64
65. Jendoubi A, Naceur IB, Bouzouita A, A comparison between intravenous lidocaine and ketamine on acute and chronic pain after open nephrectomy: A prospective, double-blind, randomized, placebo-controlled study: Saudi J Anaesth, 2017; 11(2); 177-84
66. Loadsman JA, McCulloch TJ, Paleologos MS, Peri-operative lidocaine infusion for open radical prostatectomy: Anaesthesia, 2016; 71(10); 1237-38
67. Khan JS, Hodgson N, Choi S, Perioperative pregabalin and intraoperative lidocaine infusion to reduce persistent neuropathic pain after breast cancer surgery: A multicenter, factorial, randomized, controlled pilot trial: J Pain, 2019; 20(8); 980-93
68. Habibi MR, Habibi V, Habibi A, Meta-analysis and meta-regression: Expert Rev Clin Pharmacol, 2018; 11(4); 361-71
69. Kramer ME, Holtan EE, Ives AL, A case report: A A Pract, 2019; 13(3); 96-98
70. Carabalona JF, Delwarde B, Duclos A, Serum concentrations of lidocaine during bariatric surgery: Anesth Analg, 2020; 130(1); e5-e8
71. Khan JS, Yousuf M, Victor JC, A comparative meta-analysis: J Clin Anesth, 2016; 28; 95-104
72. Neal JM, Barrington MJ, Fettiplace MR, The third american society of regional anesthesia and pain medicine practice advisory on local anesthetic systemic toxicity: Executive summary 2017: Reg Anesth Pain Med, 2018; 43(2); 113-23
73. Lauretti GR, Mechanisms of analgesia of intravenous lidocaine: Rev Bras Anestesiol, 2008; 58(3); 280-86
74. Yang X, Wei X, Mu Y, A review of the mechanism of the central analgesic effect of lidocaine: Medicine (Baltimore), 2020; 99(17); e19898
75. Martin F, Cherif K, Gentili ME, Lack of impact of intravenous lidocaine on analgesia, functional recovery, and nociceptive pain threshold after total hip arthroplasty: Anesthesiology, 2008; 109(1); 118-23
76. Bryson GL, Charapov I, Krolczyk G, Intravenous lidocaine does not reduce length of hospital stay following abdominal hysterectomy: Can J Anaesth, 2010; 57(8); 759-66
77. Wuethrich PY, Romero J, Burkhard FC, A randomised, placebo-controlled study: Eur J Anaesthesiol, 2012; 29(11); 537-43
78. Kuo CP, Jao SW, Chen KM, Comparison of the effects of thoracic epidural analgesia and i.v. infusion with lidocaine on cytokine response, postoperative pain and bowel function in patients undergoing colonic surgery: Br J Anaesth, 2006; 97(5); 640-46
79. Das KC, Misra HP, Lidocaine: A hydroxyl radical scavenger and singlet oxygen quencher: Mol Cell Biochem, 1992; 115(2); 179-85
80. Wallin G, Cassuto J, Hogstrom S, Effects of lidocaine infusion on the sympathetic response to abdominal surgery: Anesth Analg, 1987; 66(10); 1008-13
In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952