09 January 2021: Clinical Research
Plasma Heat Shock Protein 90 Alpha: A Valuable Predictor of Early Chemotherapy Effectiveness in Advanced Non-Small-Cell Lung Cancer
Bo Zhong1ACE, Juxin Shen1AEG*, Chunyi Zhang1BDE, Guozhong Zhou2BE, Yuefang Yu1DE, E Qin1BE, Jixian Tang1DE, Dongping Wu3DE, Xiaochao Liang1BEDOI: 10.12659/MSM.924778
Med Sci Monit 2021; 27:e924778
Abstract
BACKGROUND: Heat shock protein-90 alpha (HSP90a) is more abundant in non-small-cell lung cancer (NSCLC) patients than in control individuals. However, whether it can reflect chemotherapy efficacy remains unknown. This study aimed to investigate the association of HSP90a with chemotherapy in advanced NSCLC.
MATERIAL AND METHODS: We retrospectively evaluated data from patients admitted to the Department of Respiratory Medicine, Shaoxing People’s Hospital, from September 2016 to September 2018 with stage IIIB or IV NSCLC and administered 4 cycles of third-generation platinum-based combination chemotherapy (2 drugs simultaneously). Based on the RECIST1.1 criteria, complete remission (CR), partial response (PR), and stable disease (SD) in 60 cases were determined before and after chemotherapy. Before chemotherapy and after 1, 2, and 4 cycles of chemotherapy, plasma HSP90α levels were quantitated by ELISA. Chest CT was performed before and after 2 and 4 cycles of chemotherapy.
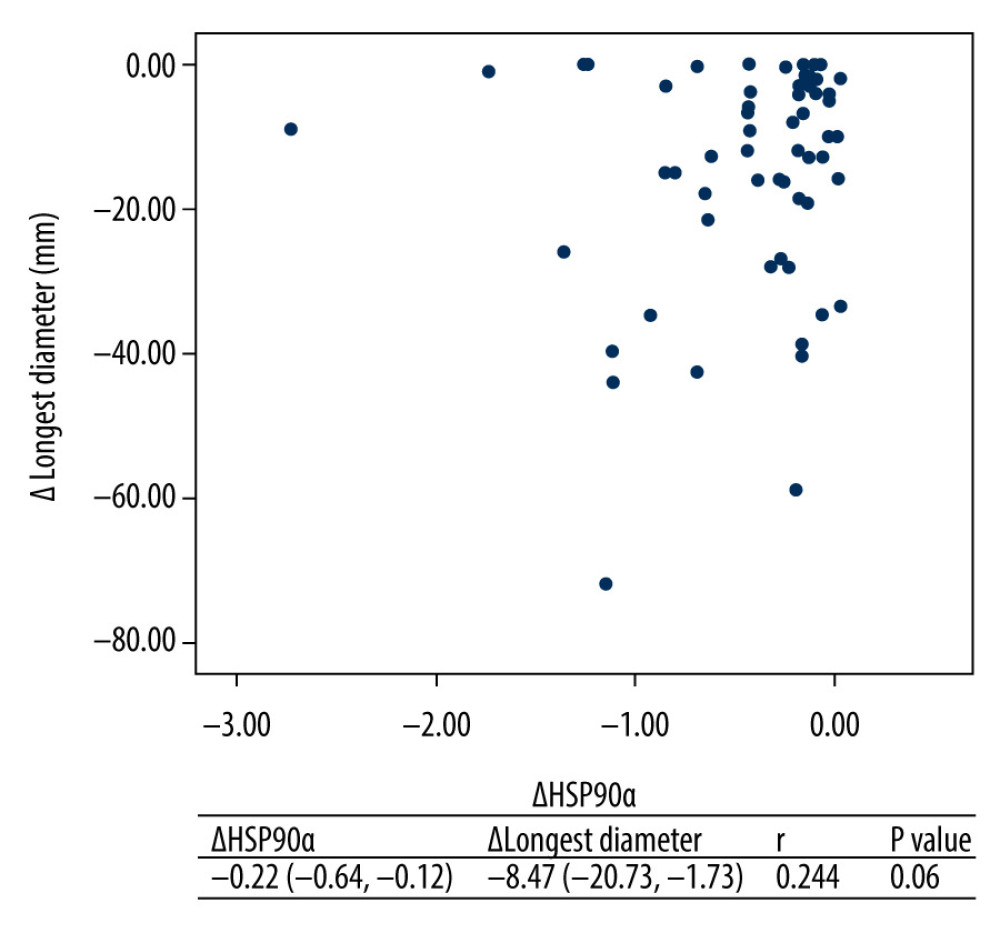
RESULTS: After 1–4 cycles of chemotherapy, plasma HSP90α levels were significantly lower than pre-chemotherapy levels (P<0.05). The sums of the longest tumor diameters after 2 and 4 cycles of chemotherapy were decreased compared with pre-chemotherapy values (P<0.05). Plasma HSP90α levels and tumor size showed no significant correlation before and after chemotherapy (r=0.244, P=0.06).
CONCLUSIONS: Plasma HSP90α can be considered a valuable predictor of early chemotherapy effectiveness in advanced NSCLC, and is positively correlated with tumor remission after chemotherapy. However, plasma HSP90α level is not correlated with tumor diameter and pathological type.
Keywords: Chemotherapy, Adjuvant, Heat-Shock Proteins, Biomarkers, Tumor, HSP90 Heat-Shock Proteins, Response Evaluation Criteria in Solid Tumors
Background
Lung cancer is one of the deadliest malignancies worldwide [1]. There were more than 1.7 million cancer cases and 606 880 cancer-related deaths in the United States in 2019 [2]. In China, with accelerated industrialization, increased environmental pollution, heavy consumption of tobacco, and population aging, the incidence and mortality of lung cancer are rising [3,4]. Lung cancer includes non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC). NSCLC accounts for about 85% of all lung cancer cases and shows poor prognosis [5]. Indeed, most patients are diagnosed with advanced-stage (stage III–IV) disease and are not eligible for surgical resection [6]. The standard third-generation platinum-containing combination chemotherapy remains the main treatment for advanced NSCLC, with a low effective rate approximating 30% [7]. Therefore, the vast majority of patients do not have good clinical benefits despite pronounced chemotherapy-related toxicities and adverse effects.
The traditional method for evaluating chemotherapy efficacy consists of observing and measuring the changes of tumor lesions on chest CT before and after chemotherapy, according to the RECIST criteria [8]. However, this technique cannot detect subtle and early changes after treatment; indeed, clinicians usually observe anatomical changes by CT several weeks after treatment initiation [9]. In addition, imaging evaluation heavily relies on individual subjective judgment. Therefore, an effective indicator is required for early prediction of chemotherapy efficacy. This would not only reduce pain associated with the adverse effects of chemotherapy, as well as the economic burden, but would also help patients select other treatment options before their physical condition deteriorates.
Heat shock protein 90α (HSP90α), a member of the family of heat shock proteins (HSP), is a highly conserved molecular chaperone that plays important roles in maintaining the stability of various signaling proteins in cells and promoting cell survival and growth [10]. Hsp90α levels in normal human plasma are very low; however, in the tumor state, HSP90α can be specifically secreted by tumor cells to the extracellular space, entering the blood circulation [11,12]. Consequently, plasma Hsp90α levels in patients with lung cancer are significantly higher than those of healthy controls and show close associations with tumor cell invasion and metastasis [13].
However, whether it can reflect chemotherapy efficacy remains unknown. We hypothesized that Hsp90α could rapidly and effectively detect chemotherapy effectiveness in patients with lung cancer. Therefore, this study aimed to assess the plasma levels of HSP90α in advanced-stage NSCLC patients before and after chemotherapy, determining its role in early prediction of chemotherapy effectiveness. We found that plasma HSP90α is positively correlated with tumor remission after chemotherapy, indicating that HSP90α could be considered a valuable predictor of early chemotherapy effectiveness in advanced NSCLC.
Material and Methods
PATIENTS:
This was a retrospective cohort study that assessed patients with untreated stage IIIB or IV NSCLC admitted to the Department of Respiratory Medicine, Shaoxing People’s Hospital, from September 2016 to September 2018. Inclusion criteria were: 1) NSCLC confirmed by pathological examination, including adenocarcinoma and squamous cell carcinoma; 2) no previous NSCLC treatment and lesions not removed after surgical exploration; 3) ECOG score for physical status of 0–1 points; 4) completion of the standard 4-cycle third-generation platinum-containing 2-drug combination chemotherapy. Exclusion criteria were: 1) pregnancy; 2) mental illness; 3) other lung diseases; 4) diseased organs such as the heart, brain, kidney, and/or lung (no lung cancer metastasis); 5) primary malignancies in other systems; 6) infection, trauma, and poisoning; 7) disease progression (PD) after the 4-cycle treatment. The study was approved by the Ethics Committee of Shaoxing People’s Hospital ([2016] 67). Signed informed consent was obtained from all patients or their legal representatives.
EVALUATION OF RESPONSE TO CHEMOTHERAPY:
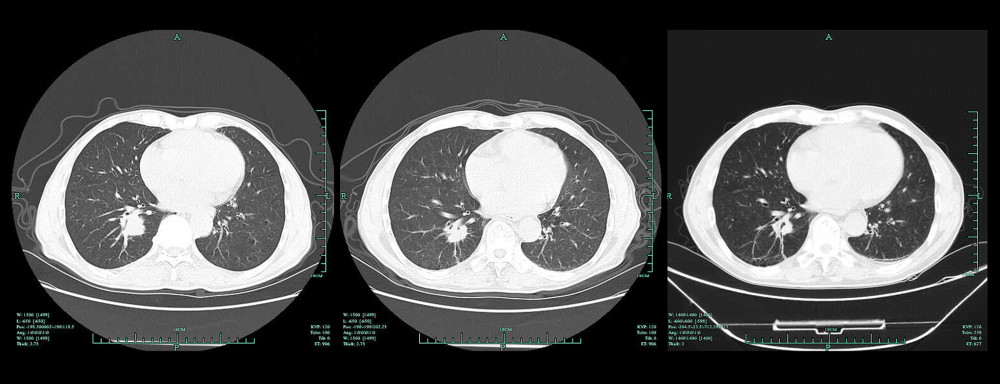
Chest CT was performed before chemotherapy and after 2 (6 weeks from baseline) and 4 (12 weeks from baseline) cycles of chemotherapy, respectively. The greatest diameter of the tumor lesion was measured as the baseline value by chest CT (scan thickness of 3.75 mm). When there were multiple measurable lesions at baseline, all lesions were recorded and measured, and the 2 largest target lesions were selected as the baseline lesions. Tumor lesions before and after chemotherapy were observed and measured, and disease remission was assessed according to the RECIST 1.1 criteria, determining complete remission (CR), partial remission (PR), stable disease (SD), and progressive disease (PD).
DETERMINATION OF PLASMA HSP90α LEVELS:
Peripheral venous blood (3 ml) from the enrolled patients was collected into EDTA-K2 anticoagulation tubes the day before chemotherapy (baseline levels) and after 1 (3 weeks from baseline), 2, and 4 cycles of chemotherapy in the fasting state (morning). After centrifugation, 1 ml of plasma was obtained. The HSP90α quantitative test kit (#BMS2090, Invitrogen, USA) was used to quantitate plasma HSP90α levels by ELISA, strictly following the manufacturer’s instructions and using a ThermoFisher Scientific microplate reader. The changes in HSP90α levels from baseline values were determined.
STATISTICAL ANALYSIS:
SPSS 22.0 statistical software (SPSS, USA) was utilized for data analysis. Categorical and measurement data are presented as n (%) and mean±standard deviation (SD), respectively. A paired
Results
BASELINE PATIENT CHARACTERISTICS:
A total of 60 patients were included in this study according to the above inclusion and exclusion criteria and received 4 cycles of standard third-generation platinum-containing 2-drug combination chemotherapy. The patients were 45–79 years old, with an average age of 66.38±4.25 years. They included 32 males and 28 females. There were 38 and 22 stage IIIB and IV cases, respectively. The detailed baseline characteristics of the patients are summarized in Table 1.
PLASMA HSP90α LEVEL AND TUMOR SIZE CHANGES AFTER CHEMOTHERAPY:
The average HSP90α measurement before chemotherapy was 0.534±0.141 in males and 0.761±0.603 in females (P=0.163). Plasma HSP90α levels gradually decreased with the duration of chemotherapy (Table 2). Pairwise comparisons showed statistically significant differences between all group pairs (P<0.001), except pre-chemotherapy vs. 1 cycle of chemotherapy.
The greatest diameters of tumor lesions after 2 and 4 cycles of chemotherapy were markedly decreased compared with the values obtained before chemotherapy (Table 3). Pairwise comparisons showed statistically significant differences between all group pairs (P<0.05). Figure 1 shows a representative case.
ASSOCIATIONS OF PLASMA HSP90α LEVELS WITH THERAPEUTIC RESPONSE:
The correlation between plasma HSP90α levels and tumor lesion size before and after chemotherapy was not significant as analyzed by the Spearman’s rank correlation method (Figure 2; r=0.244, P=0.06).
Before chemotherapy, the patients were divided into an adenocarcinoma group and a squamous cell carcinoma group, which showed similar HSP90a levels (
Discussion
This study demonstrated that plasma HSP90α could predict early chemotherapy effectiveness in advanced NSCLC, showing a positive correlation with tumor remission even after just 1 cycle of chemotherapy.
HSP expression in cells is significantly increased under stimulation by certain environmental factors or stress conditions [13]. HSP play important roles in protein synthesis, folding, assembly, secretion, transport, and degradation, regulate transcription factors and protein kinases, and maintain the stability of multiple intracellular signaling proteins, thereby promoting cell survival and growth [14–16]. HSP90, an important member of the HSP family, participates in protein folding and activation, among other processes [17]. Since its substrate proteins are mostly overexpressed or mutated EGFR, p53, HER2, c-MET, RAF, and other proteins associated with tumor generation and development, HSP90 has become a hotspot in the field of cancer research. Under the tumor state, HSP90 is overexpressed and plays an important role in protecting and maintaining the stability of tumor cells, which could increase malignancy [18]. HSP90 expression is high in lung cancer, and this high expression might enhance the proliferation and metastatic ability of lung cancer [16,19,20]. Decreasing HSP90 expression also decreases lung cancer proliferation, metastasis, and mortality [16,19,20]. HSP90 has 2 subtypes, including HSP90α and HSP90β. Under normal physiological conditions, HSP90α and HSP90β are present in cells, with extremely low extracellular levels; under hypoxia, injury, oxidation, and other stress states, intracellular HSP90α and HSP90β levels are significantly increased, but only HSP90α can be secreted by a variety of cells [13]. In cancer, HSP90α is continuously secreted to the extracellular space and acts in the following aspects: (1) extracellular HSP90α functions as a molecular chaperone to keep mutant signaling proteins active; and (2) extracellular HSP90α affects the proto-oncogenes Her-2 and metalloproteinase 2 on the cell surface, promoting the infiltration and metastasis of tumor cells [13].
As shown above, plasma HSP90α levels were significantly decreased as early as right after completion of the first cycle of chemotherapy in patients with stage IIIB and IV NSCLC, and this reduction persisted throughout the 4 treatment cycles. Therefore, plasma levels of HSP90α could reflect chemotherapy effectiveness, as supported by Shi et al. [21]. Similar studies should be performed in other cancers, since in addition to NSCLC [22], HSP90α has been demonstrated to be highly expressed in prostate cancer [23], multiple myeloma [24], pancreatic cancer [25], liver cancer [26–28], and cholangiocarcinoma [29].
In addition, changes in plasma HSP90α levels after chemotherapy were positively correlated with disease remission based on the RECIST1.1 criteria. This confirms that plasma HSP90α could be used as an early predictor of chemotherapy efficacy in patients with advanced NSCLC. We also found that plasma HSP90α content was not correlated with tumor diameter, indicating that it may help detect subtle positive effects of chemotherapy not revealed by CT.
Previous studies assessing HSP90α have focused on tumor histology, with few reports evaluating the clinical significance of plasma HSP90α levels. Meanwhile, it is known that plasma HSP90α levels in patients with lung cancer are increased and closely related to tumor occurrence, development, and prognosis [13]. The current study provides additional information regarding this important cancer marker. Based on our findings, HSP90α could be used as early as 3 weeks after treatment initiation in advanced NSCLC to determine chemotherapy efficacy. This would help decide between maintaining the same regimen and applying other chemotherapeutics for further patient care. As a result, patient prognosis and quality of life may be improved (increased treatment efficacy), while the financial burden on the families and health care system is relieved (avoiding use of inefficient chemotherapeutics).
A few limitations of this study should be mentioned. First of all, this was a retrospective study with inherent shortcomings. In addition, the sample size was relatively small, and all patients were enrolled in the same center. Although the RECIST1.1 criteria were used for assessing therapeutic outcomes, we did not include other known prognostic markers as controls. Therefore, well-designed, large, multicenter prospective studies are required to confirm the present findings.
Conclusions
In conclusion, plasma HSP90α is significantly reduced in stage IIIB, and IV NSCLC patients administered 1–4 cycles of third-generation platinum-based combination chemotherapy and can be considered a valuable predictor of early chemotherapy effectiveness in advanced NSCLC. In addition, plasma HSP90α levels are positively correlated with tumor remission after chemotherapy. Overall, plasma HSP90α content can detect subtle positive effects of chemotherapy not revealed by CT.
References
1. Zaorsky NG, Churilla TM, Egleston BL, Causes of death among cancer patients: Ann Oncol, 2017; 28(2); 400-7
2. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2019: Cancer J Clin, 2019; 69(1); 7-34
3. Parascandola M, Xiao L, Tobacco and the lung cancer epidemic in China: Transl Lung Cancer Res, 2019; 8(Suppl 1); S21-30
4. Bode AM, Dong Z, Wang H, Cancer prevention and control: Alarming challenges in China: Natl Sci Rev, 2016; 3(1); 117-27
5. Smith DL, Acquaviva J, Sequeira M, The HSP90 inhibitor ganetespib potentiates the antitumor activity of EGFR tyrosine kinase inhibition in mutant and wild-type non-small cell lung cancer: Target Oncol, 2015; 10(2); 235-45
6. Farbicka P, Nowicki A, Palliative care in patients with lung cancer: Contemp Oncol (Pozn), 2013; 17(3); 238-45
7. Germonpré P, Van den Wyngaert T, Second-line erlotinib after failure of pemetrexed-containing chemotherapy in advanced non-small cell lung cancer (NSCLC): Real-world effectiveness, safety and tolerability: PLoS One, 2019; 14(4); e0215135-35
8. Subbiah V, Chuang HH, Gambhire D, Kairemo K, Defining clinical response criteria and early response criteria for precision oncology: Current state-of-the-art and future perspectives: Diagnostics (Basel), 2017; 7(1); 10
9. Hwang K-E, Kim H-R, Response evaluation of chemotherapy for lung cancer: Tuberc Respir Dis (Seoul), 2017; 80(2); 136-42
10. Sottile ML, Nadin SB, Heat shock proteins and DNA repair mechanisms: An updated overview: Cell Stress Chaperones, 2018; 23(3); 303-15
11. Song X, Wang X, Zhuo W, The regulatory mechanism of extracellular Hsp90{alpha} on matrix metalloproteinase-2 processing and tumor angiogenesis: J Biol Chem, 2010; 285(51); 40039-49
12. Wang X, Song X, Zhuo W, The regulatory mechanism of Hsp90alpha secretion and its function in tumor malignancy: Proc Natl Acad Sci USA, 2009; 106(50); 21288-93
13. Zhou Y, Deng X, Zang N, Transcriptomic and proteomic investigation of HSP90A as a potential biomarker for HCC: Med Sci Monit, 2015; 21; 4039-49
14. Song X, Zhao Z, Qi X, Identification of epipolythiodioxopiperazines HDN-1 and chaetocin as novel inhibitor of heat shock protein 90: Oncotarget, 2015; 6(7); 5263-74
15. Mazaira GI, Camisay MF, De Leo S, Biological relevance of Hsp90-binding immunophilins in cancer development and treatment: Int J Cancer, 2016; 138(4); 797-808
16. Rasola A, HSP90 proteins in the scenario of tumor complexity: Oncotarget, 2017; 8(13); 20521-22
17. Wang H, Zou X, Wei Z, Hsp90α forms a stable complex at the cilium neck for the interaction of signalling molecules in IGF-1 receptor signalling: J Cell Sci, 2015; 128(1); 100-8
18. Yang J, Song X, Chen Y, PLCγ1-PKCγ signaling-mediated Hsp90α plasma membrane translocation facilitates tumor metastasis: Traffic, 2014; 15(8); 861-78
19. Rong B, Yang S, Molecular mechanism and targeted therapy of Hsp90 involved in lung cancer: New discoveries and developments (Review): Int J Oncol, 2018; 52(2); 321-36
20. Mittal S, Rajala MS, Heat shock proteins as biomarkers of lung cancer: Cancer Biol Ther, 2020; 21(6); 477-85
21. Shi Y, Liu X, Lou J, Plasma levels of heat shock protein 90 alpha associated with lung cancer development and treatment responses: Clin Cancer Res, 2014; 20(23); 6016-22
22. Seo YH, Discovery of 2′,4′-dimethoxychalcone as a Hsp90 inhibitor and its effect on iressa-resistant non-small cell lung cancer (NSCLC): Arch Pharm Res, 2015; 38(10); 1783-88
23. Seidel C, Schnekenburger M, Mazumder A, 4-Hydroxybenzoic acid derivatives as HDAC6-specific inhibitors modulating microtubular structure and HSP90α chaperone activity against prostate cancer: Biochem Pharmacol, 2016; 99; 31-52
24. Suzuki R, Hideshima T, Mimura N, Anti-tumor activities of selective HSP90α/β inhibitor, TAS-116, in combination with bortezomib in multiple myeloma: Leukemia, 2015; 29(2); 510-14
25. Ghadban T, Dibbern JL, Reeh M, HSP90 is a promising target in gemcitabine and 5-fluorouracil resistant pancreatic cancer: Apoptosis, 2017; 22(3); 369-80
26. Guan ZB, Sun MX, Song JF, Regulation mechanism exploration of hsp90α expression by hepatitis B virus X protein in human hepatocarcinoma cells: Oncology Progress, 2017; 15(11); 1672-535
27. Liu W, Li J, Zhang P, A novel pan-cancer biomarker plasma heat shock protein 90alpha and its diagnosis determinants in clinic: Cancer Sci, 2019; 110(9); 2941-59
28. Sourbier C, Plasma HSP90α and liver cancer: A potential biomarker?: EBioMedicine, 2017; 25; 7-8
29. Boonjaraspinyo S, Juasook A, Boonmars T, A promising serum autoantibody marker, anti-heat shock protein 90α, for cholangiocarcinoma: Asian Pac J Cancer Prev, 2015; 16(14); 5779-85
Figures
Tables
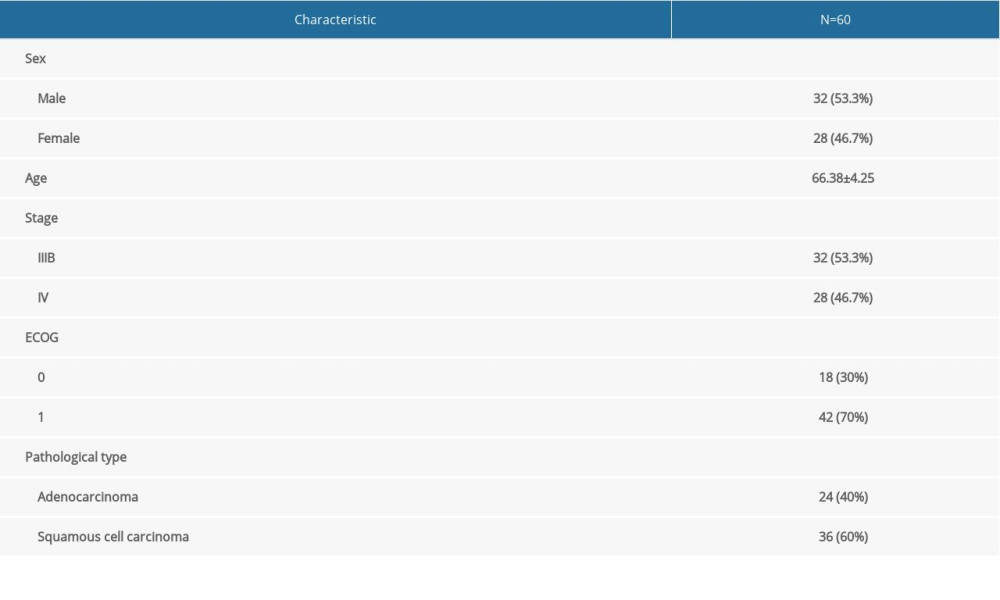 Table 1. Patient baseline data.
Table 1. Patient baseline data.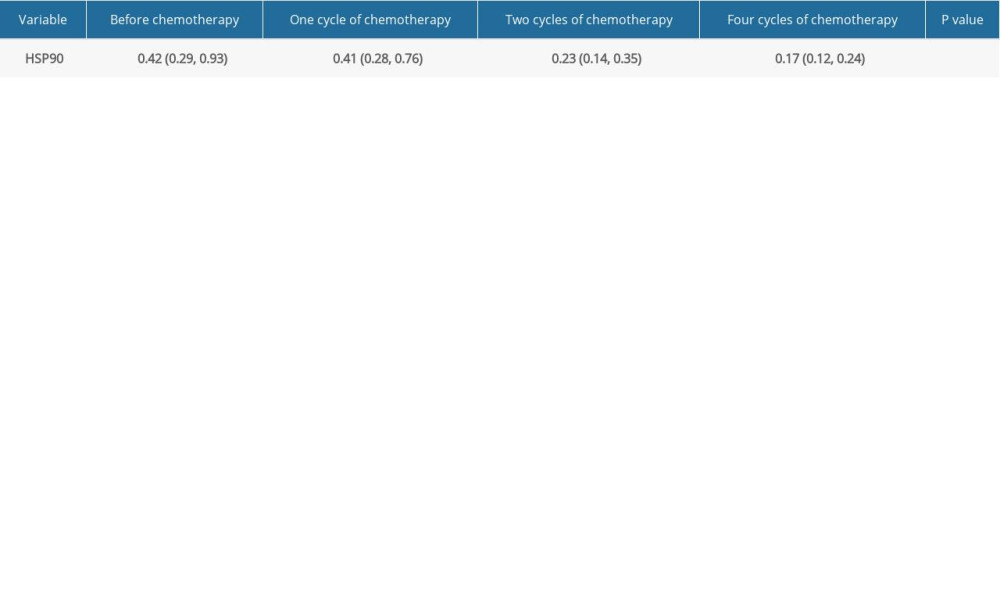 Table 2. Plasma HSP90α levels at different times before and after chemotherapy.
Table 2. Plasma HSP90α levels at different times before and after chemotherapy.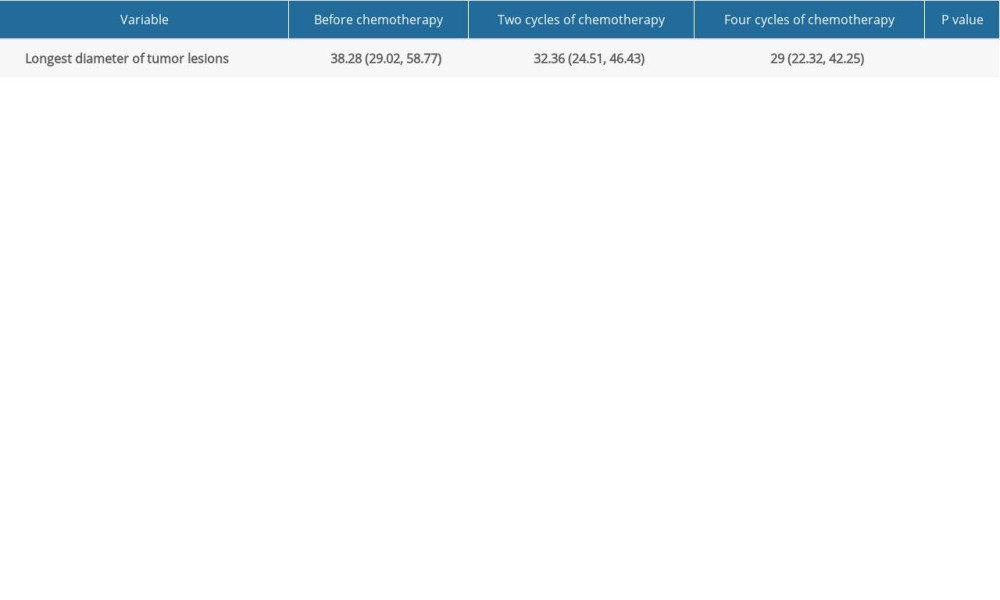 Table 3. Tumor lesion diameter at different times before and after chemotherapy.
Table 3. Tumor lesion diameter at different times before and after chemotherapy. Table 1. Patient baseline data.
Table 1. Patient baseline data. Table 2. Plasma HSP90α levels at different times before and after chemotherapy.
Table 2. Plasma HSP90α levels at different times before and after chemotherapy. Table 3. Tumor lesion diameter at different times before and after chemotherapy.
Table 3. Tumor lesion diameter at different times before and after chemotherapy. In Press
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952