30 August 2020: Lab/In Vitro Research
Asiaticoside Antagonizes Proliferation and Chemotherapeutic Drug Resistance in Hepatocellular Carcinoma (HCC) Cells
Ying Ma1BE, Jun Wen1BC, Jing Wang1CE, Chunyan Wang1DF, Yan Zhang1DEF, Lili Zhao1F, Jia Li1A*, Xue Feng1FDOI: 10.12659/MSM.924435
Med Sci Monit 2020; 26:e924435
Abstract
BACKGROUND: Hepatocellular carcinoma (HCC) is the fifth most prevalent malignant tumor in China after lung cancer, gastric cancer, esophageal cancer, and breast cancer, and has a high mortality rate. Though there are a series of therapeutic strategies is now available for HCC in clinical practice, the 5-year survival rate after surgery is still low. In addition, multi-drug resistance (MDR) is one of the most important factors responsible for the low survival rate and poor therapy response in HCC. Hence, novel treatment strategies and molecules for HCC need to be developed.
MATERIAL AND METHODS: We assessed the effect of asiaticoside, a natural product derived from Centella asiatica (L.) Urban, on HCC cell proliferation and drug resistance.
RESULTS: Our data indicated that asiaticoside significantly inhibited the proliferation of HCC cell lines QGY-7703 and Bel-7402 in a dose- and time-dependent manner. Moreover, asiaticoside significantly induced apoptosis in QGY-7703 and Bel-7402 cells. Treatment with asiaticoside also caused G1 cell cycle arrest in QGY-7703 and Bel-7402 cells. Western blot assay results indicated that the mechanism underlying the effects of asiaticoside involves inhibiting the activity of the PI3K/Akt and MAPK/ERK pathways. Furthermore, asiaticoside significantly antagonized P-gp-mediated MDR in HCC cells.
CONCLUSIONS: Our results suggest that asiaticoside has the potential to be applied in the treatment of HCC patients, but further evidence is needed to confirm our results, particularly in vivo efficacy.
Keywords: Carcinoma, Hepatocellular, Cell Cycle Checkpoints, Liver Neoplasms, Phosphatidylinositol 3-Kinases
Background
Hepatocellular carcinoma (HCC), a heterogeneous group of diseases, is the sixth most common cancer worldwide, causing about 60 000 deaths per year [1]. In China, HCC is the fifth most commonly diagnosed cancer after lung cancer, gastric cancer, esophageal cancer, and breast cancer, and it has a high mortality rate [2]. A series of novel therapeutic strategies are now available for HCC, including surgical resection and liver transplantation, which have become primary surgical treatment strategies for HCC [3]; however the 5-year survival rate after surgery is still only 15–40% [4–6]. In addition, although surgical treatment is the main treatment used in early-stage HCC, patients are usually diagnosed at an advanced stage, in which adjuvant therapy, including systemic chemotherapy, is essential [7]. However, development of chemoresistance is a major obstacle in chemotherapy-based treatment of the disease, with poor prognosis [8]. Hence, novel pharmacotherapies to control HCC progression and resistance are urgently required. Natural products usually have considerable therapeutic efficacy and fewer adverse effects than traditional chemotherapeutic drugs [9,10]. In recent years, small molecules derived from natural products have become increasingly important in new drug research into treatment of multiple types of cancer, and provide alternative treatment strategies for HCC [11].
Asiaticoside, one of the primary active compounds derived from
HCC pathogenesis includes a series of genetic and epigenetic mutations, which finally induce aberrant activation of multiple signaling pathways, such as phosphoinositide 3-kinases (PI3Ks) signal transduction pathway 28854942. The PI3K family plays key roles in multiple physiological and pathological processes, including cell proliferation, apoptosis, cell cycle, and cell migration. Mechanistically, PI3Ks transfer extracellular signals, such as a series of cytokines, growth factors, and some chemotherapeutic drugs, to within cells by synthesizing second-message phospholipid PI (3,4,5) P3, and subsequently activate protein kinase B (Akt), as well as downstream effectors. PI3K transduction pathways are often upregulated and excessively activated in multiple types of cancer and have increasingly become the potential targets of novel anti-cancer drugs. There are now a series of PI3K inhibitors approved or in clinical evaluation, including idelalisib, alpelisib [20], BKM120 [21], and gedatolisib [22].
In the present study, we demonstrated that asiaticoside significantly inhibits HCC cell proliferation and clone formation. We found that asiaticoside induced HCC cell apoptosis and cell cycle arrest, which are related to inhibition of PI3K pathways. We also found that asiaticoside reduces HCC cell drug resistance by downregulating the expression level of P-gp by decreasing the ROS level in chemotherapy-resistant HCC cells.
Material and Methods
CHEMICALS AND REAGENTS:
Asiaticoside was purchased from Selleck Chemicals (Houston, TX, USA), Dulbecco’s modified Eagle’s medium (DMEM) and fetal bovine serum (FBS) were purchased from Biological Industries (BI, Israel). Penicillin/streptomycin and trypsin were purchased from Corning Incorporated (Corning, NY, USA). The chemotherapy drugs paclitaxel (PTX), Adriamycin (ADM), colchicine, and vincristine were purchased from Energy Chemicals (Shanghai, China). The primary monoclonal antibodies of PI3K-p110α (#4255), PI3K-p110β (#3011), PI3K-p110γ (#4252), p-PDK1 (Ser241) (#3061), p-Akt (Ser473) (#4060), p-mTOR (Ser2448) (#2976), p-ERK1/2 (#4370), ERK1/2 (#4696), p-JNK1/2 (#9255), JNK1/2 (#9252), p-P38 (#4511), P38 (#8690), β-actin (#4970), and P-gp (ABCB1) (#13342) were purchased from Cell Signaling Technology (Danvers, MA, USA). HRP-conjugated secondary goat anti-mouse antibody, Annexin V-FITC, propidium iodide (PI), and 2′,7′-dichlorofluorescein diacetate (DCFH-DA) were purchased from Beyotime Biotechnology (Nantong, JS, China). All other chemicals were purchased from Sigma Aldrich (MO, USA).
CELL LINES AND CELL CULTURE:
The human HCC cell line QGY-7703 was purchased from the Cell Bank of the Chinese Academy of Science (Shanghai, China). The human HCC cell line Bel-7402 was purchased from the Cell Resource Center, Peking Union Medical College (Beijing, China), and the ADM-resistance Bel-7402/ADM cell line was purchased from KeyGEN Biotech Corporation (Nanjing, JS, China). We used 300 nM of doxorubicin to maintain the resistance level of Bel-7402/ADM cells. Each cell line was cultured in complete DMEM media containing 10% FBS and 1% penicillin/streptomycin at 37°C in a sterile humidified atmosphere containing 5% CO2. Each of the cell lines was passaged when cells occupied 80% of the cell dish. The ADM-resistance cell line Bel-7402/ADM was cultured in drug-free DMEM for more than 14 days before experiments. The cell lines used in this study were authenticated to be free of contamination.
CELL VIABILITY ASSAYS:
The anti-proliferation effects of asiaticoside on HCC cells were determined by MTT assay. In brief, human HCC QGY-7703 and Bel-7402 cells were seeded into 96-well plates at a density of 5000 cells per well (200 μL) for overnight culturing, then various concentrations of asiaticoside (gradient dilution) were added and incubated for 48 h, after which MTT was added and incubated at 37°C for 4 h. Subsequently, supernatant was discarded and DMSO was used to dissolve the formazan crystals. The OD value was measured at 490 nm. To assess the time-dependent inhibition effect of asiaticoside, 5 μM of asiaticoside was added into the cells using the protocol described above.
PLATE CLONE FORMATION ASSAY:
After treatment with asiaticoside (1, 3, and 10 μM) or DMSO for 48 h, QGY-7703 and Bel-7402 cells were harvested and pipetted gently to single-cell suspension. Subsequently, 0.7% agarose (containing 10% FBS) in DMEM medium (3 mL) was poured into the bottom of 6-well plates, followed by addition of 0.3% agarose (containing 10% FBS) in DMEM medium (2 mL) to the aforementioned single-cell suspension (5000 cells per mL). After incubating for 15 days, paraformaldehyde-PBS solution (4%) was used to fix the colonies, followed by staining with Giemsa buffer for 20 min and washing with double-distilled water until the colonies were visible and clear.
FLOW CYTOMETRY ASSAYS FOR CELL CYCLE DISTRIBUTION ANALYSIS:
QGY-7703 and Bel-7402 cells were plated into 6-well plates at a density of 2×105 cells per well and treated with DMSO or various concentrations of asiaticoside (1, 3, and 10 μM) and incubated for 48 h. Subsequently, cells were harvested and fixed with ice-cold ethanol (70%) for at least 30 min, after which the cells were centrifuged and resuspended in 0.1% Triton X-100 PBS solution. Cells were then stained by PI for 30–35 min. A BD FACS Verse flow cytometer was used to analyze the cell cycle populations. Data were analyzed using FlowJo software.
FLOW CYTOMETRY ASSAYS FOR APOPTOSIS ANALYSIS BY ANNEXIN V AND PI DOUBLE-STAINING:
Evaluation of apoptotic cells was conducted by Annexin V and PI double-staining, and detected by flow cytometry. In brief, QGY-7703 and Bel-7402 cells were plated into 6-well plates at a density of 2×105 per well and treated with DMSO or various concentrations of asiaticoside (1, 3, and 10 μM), followed by incubation for 48 h. Subsequently, cells were harvested and incubated with Annexin V (5 μL of 100 μL binding buffer) and PI (5 μL of 100 μL binding buffer) for 30 min. Then, the cells were washed and resuspended in 200 μL of binding buffer. A BD FACS Verse flow cytometer was used to detect apoptotic cells. FlowJo software was used to analyze the results.
WESTERN BLOT ASSAYS:
The expression of P-gp and the effects of asiaticoside on PI3K/Akt/MAPKs signaling were determined by Western blot assays. Briefly, cells were seeded into 6-well plates and treated with various concentration of asiaticoside (1, 3, and 10 μM for PI3K/Akt/MAPKs, 2, 4, and 6 μM for expression of P-pg) for 48 h. Subsequently, cells were harvested and total proteins of the cells were extracted with RIPA. Protein concentration was detected by use of a BCA kit and normalized (20 μg) before 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The proteins on the gel were transported to PVDF membranes with 200 mA electricity, and non-fat milk (5%) was used to block the non-specific proteins. Subsequently, primary antibodies were diluted in 5% BSA PBS buffer and incubated overnight in the dark at 4°C. On the following day, after washing twice, the membranes were incubated with diluted HRP-conjugated secondary antibodies for 1 h, and protein bands were visualized by ECL and digitalized by scanning.
REVERSAL OF P-GP-MEDIATED MDR:
As described above in “Cell viability assays”, MTT assay was used to conducted the reversal experiment of asiaticoside on P-gp-mediated drug resistance in HCC cells. Briefly, Bel-7402/ADM cells were seeded into 96-well plates and incubated overnight. On the following day, 2 and 4 μM asiaticoside were added for 2 h. Subsequently, various concentrations of paclitaxel, doxorubicin, colchicine, or vincristine were added, followed by further incubation for 48 h.
DETECTION OF REACTIVE OXYGEN SPECIES (ROS) IN DRUG-RESISTANT HCC CELLS:
Intracellular ROS level was determined by DCFH-DA through flow cytometry assay. In brief, Bel-7402/ADM cells were treated with various concentration (2, 4, and 6 μM) of asiaticoside for 48 h, then stained with DCFH-DA for 30 min. After washing with ice-cold PBS, a FACS Verse flow cytometer was used.
STATISTICS:
All data are presented as mean±SD from at least 3 independent experiments. One-way ANOVA was used to determine the significance of differences between control and experimental groups, with the level of significance set at p value less than 0.05. A dot plot and bar plot were created using GraphPad 7.00.
Results
ASIATICOSIDE SIGNIFICANTLY INHIBITED THE PROLIFERATION OF HUMAN HCC QGY-7703 AND BEL-7402 CELLS:
In MTT assays, asiaticoside showed a significant inhibitory effect on HCC cells. Specifically, asiaticoside significantly inhibited the cell viability of QGY-7703 and Bel-7402 cell lines in dose-dependent (Figure 1A, 1B) and time-dependent (Figure 1C, 1D) manners. The IC50 values of asiaticoside on QGY-7703 cells was 6.724 μM, and that for the Bel-7402 cell line was 6.807 μM. Plate clone formation assays further demonstrated the anti-proliferation effects of asiaticoside on HCC cells. As shown in Figure 1E, the colony formation of QGY-7703 and Bel-7402 cells was significantly decreased, indicating asiaticoside suppressed the colony formation ability of HCC cells.
ASIATICOSIDE INDUCED APOPTOSIS IN HCC CELLS:
As shown in Figure 2A–2C, the ratio of apoptotic cells in QGY-7703 and Bel-7402 cells was significantly increased after treatment with asiaticoside in a concentration-dependent manner. Moreover, the apoptosis-related proteins were evaluated after treatment with asiaticoside. As shown in Figure 2D and 2E, asiaticoside treatment resulted in pronounced upregulation of cleaved-caspase9, cleaved-caspase3, and cleaved-PARP in QGY-7703 and Bel-7402 cells in a dose-dependent manner. These results indicated that asiaticoside inhibits HCC cells proliferation through inducing HCC cells apoptosis.
ASIATICOSIDE INDUCED CELL CYCLE ARREST IN HCC CELLS:
As shown in Figure 3A–3C, an obvious increase in G1 cell population was found after treatment with various concentrations of asiaticoside, indicating that asiaticoside causes G1 cell cycle arrest in HCC cells. Meanwhile, Western blot results showed the effects of asiaticoside on cell cycle checkpoint proteins, including cyclinD1 and p27, which is essential for G1 to S phase transition, and the downstream effector p-pRB. As presented in Figures 3D and 3E, in QGY-7703 and Bel-7402 cells, asiaticoside significantly downregulated the phosphorylation level of pRB and expression level of cyclin D1, and upregulated the p27 expression level, all in a dose-dependent manner, which agrees with the results of flow cytometry and PI staining, indicating that asiaticoside induced growth arrest in HCC cells in vitro.
ASIATICOSIDE STIMULATED APOPTOSIS AND CELL CYCLE ARREST THROUGH BLOCKING PI3K/AKT AND MAPK/ERK PATHWAYS IN HCC CELLS:
It is widely accepted that PI3K/Akt and MAPKs pathways play important roles in regulating proliferation, apoptosis, and cell cycle in multiple types of cancer, including HCC. Therefore, Western blot assays were conducted to detect the expression and phosphorylation level of key proteins in PI3K/Akt and MAPKs signaling after treatment with asiaticoside. As illustrated in Figure 4A and 4B, in QGY-7703 and Bel-7402 cells, the catalytic subunits of PI3K, p110α, p110β, and p110γ were significantly downregulated by asiaticoside treatment in a concentration-dependent manner. Meanwhile, the phosphorylation level of PDK1, a downstream effector of PI3K, was simultaneously inhibited by asiaticoside. Moreover, downstream of PDK1, the phosphorylation levels of Akt and mTOR were significantly downregulated after treatment with various concentration of asiaticoside in a dose-dependent manner. These results suggested that the apoptotic and cell cycle arrest characters of asiaticoside are related to blocking the PI3K/Akt pathways. In addition, in QGY-7703 and Bel-7402 cells (Figure 5A, 5B), asiaticoside significantly inhibited the phosphorylation level of kinases of ERK, JNK, and P38, which are 3 primary MAPKs molecules in mammalian cells, indicating that the anti-proliferation effects of asiaticoside are related to MAPKs pathways. These results suggested that asiaticoside inhibits the proliferation of HCC cells through blocking the PI3K/Akt and MAPKs pathways.
ASIATICOSIDE P-GP-MEDIATED MDR IN HCC CELLS:
Acquired drug resistance is a major obstacle in cancer chemotherapy, including in HCC. Therefore, we performed a series of experiments to investigate whether asiaticoside could reverse HCC drug resistance. We used the Bel-7402/ADM cell line, which is selected and is resistant to ADM and overexpresses P-gp (ABCB1), an ATP-binding cassette transporter. As illustrated in Figure 6A, low cytotoxic concentrations of asiaticoside significantly lowered the IC50 values of a series of chemotherapeutic drugs, including doxorubicin, paclitaxel, colchicine, and vincristine, in Bel-7402/ADM cells in a dose-dependent manner. Western blot results indicated that the expression level of P-gp in Bel-7402/ADM cells was obviously downregulated (Figure 6E), which could be responsible for the reversal of drug resistance induced by asiaticoside. Our results suggest that the increased level of ROS may be involved in upregulation of P-gp, which resulted in drug resistance. Hence, we performed flow cytometry analysis to detect changes in ROS levels caused by asiaticoside in Bel-7402/ADM cells. As shown in Figure 6F, asiaticoside significantly inhibited the ROS level in Bel-7402/ADM cells.
Discussion
Natural products are extraordinarily diverse and form tremendously large libraries containing abundant bioactive molecules, which are of vital importance in finding novel agents to treat many diseases, especially malignant tumors [23–25]. Compared to the traditional chemotherapeutic drugs, natural product-derived molecules always maintain considerable therapeutic efficacy and have fewer adverse effects in the process of disease treatment [10,26]. A series of natural agents have been approved as anti-cancer drugs, such as vincristine and paclitaxel, which have already shown remarkable efficacy in cancer therapy [9,27]. Hence, it is necessary to evaluate more natural products-derived molecules that have potential effects such as inhibiting proliferation and overcoming drug resistance.
As a triterpene-rich medicinal Chinese traditional herb,
Although extracts of
Our study demonstrated that asiaticoside inhibits HCC cells viability and colony formation, which are 2 major aspects of cell proliferation, in a dose-dependent manner. These results indicate that asiaticoside reduces the proliferation of HCC cells
The PI3K/Akt pathway is one of the most important pathways regulating cell proliferation, apoptosis, and cell cycle in multiple cancer cells, including HCC [33–35]. High expression and aberrant increase of action of PI3K pathways have been widely documented in multiple human cancers [36]. In addition, MAPKs signals, primarily consisting of 3 kinases molecules – ERK, JNK, and P38 – are involved in a series of physiological and pathological processes. We found significant alterations of PI3K/Akt and MAPKs pathways after treatment with asiaticoside. Specifically, the expression of 3 major PI3K catalytic subunits – p110α, p110β, and p110γ – were significantly downregulated by asiaticoside. Meanwhile, the phosphorylation level of the PI3K downstream effector, PDK1, which is also the upstream protein kinase of Akt, was dramatically inhibited by asiaticoside, together with the downregulation of phosphorylation level of Akt or mTOR. Moreover, as key proteins in MAPKs pathways, the phosphorylation levels of ERK, JNK, and P38 were also reduced by asiaticoside. Taken together, these results indicate that the mechanisms of action of asiaticoside on HCC cells proliferation are involved in blocking the activity of PI3K/Akt and MAPKs pathways.
Acquired drug resistance is one of the largest obstacles in cancer chemotherapy, which is always related to the overexpression of P-glycoprotein (P-gp) [37]. In this study, we determined the effect of asiaticoside on reversal of drug resistance mediated by overexpression of P-gp in Bel-7402/ADM HCC cells. Asiaticoside significantly reversed chemotherapeutic resistance in Bel-7402/ADM cells by downregulating the expression level of P-gp in Bel-7402/ADM. It is documented that increased levels of ROS are involved in upregulation of P-gp [38], so we assessed changes in ROS levels caused by asiaticoside in Bel-7402/ADM cells. We found that asiaticoside significantly inhibited the ROS level in Bel-7402/ADM cells, suggesting that the reversal of drug resistance caused by asiaticoside occurs by downregulating P-gp via downregulation of ROS.
Conclusions
In conclusion, our findings suggest that asiaticoside could be a novel candidate for use in HCC therapy, with promising anti-cancer activities, and the ability to reduce drug resistance in HCC cancer cells. Nevertheless, more studies are needed to further determine the mechanism of action underlying the anti-HCC effects of asiaticoside; for example,
Figures
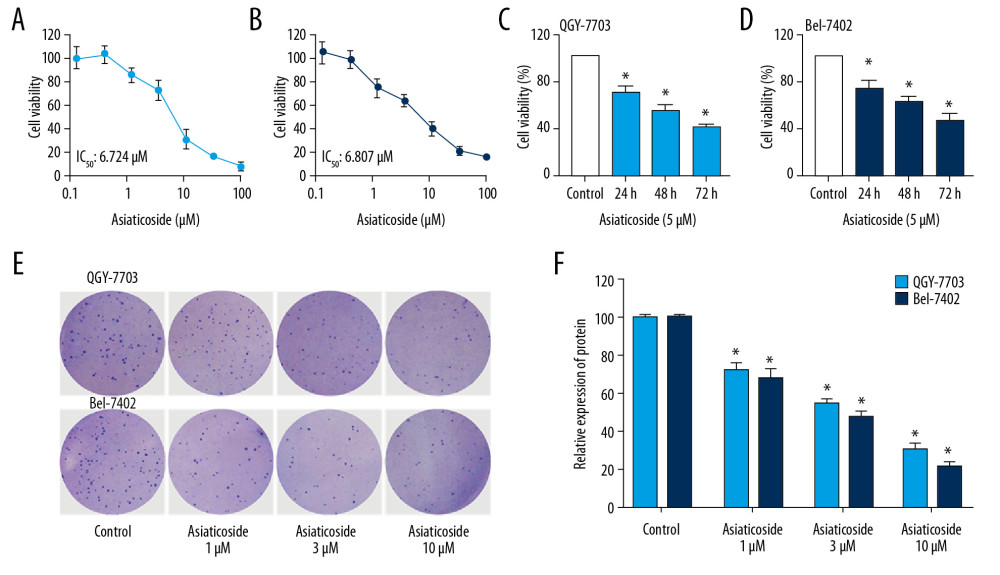 Figure 1. The inhibitory effect of asiaticoside on HCC cells proliferation. Concentration-viability curves of asiaticoside on QGY-703 cells (A) and Bel-7402 cells (B) after treatment for 48 h. Asiaticoside (5 μM) inhibited the proliferation of QGY-7703 cells (C) and Bel-7402 cells (D) in a time-dependent manner. (E) Colony formation assays of asiaticoside in QGY-7703 and Bel-7402 cells. (F) Statistical analysis of colony formation assays. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 1. The inhibitory effect of asiaticoside on HCC cells proliferation. Concentration-viability curves of asiaticoside on QGY-703 cells (A) and Bel-7402 cells (B) after treatment for 48 h. Asiaticoside (5 μM) inhibited the proliferation of QGY-7703 cells (C) and Bel-7402 cells (D) in a time-dependent manner. (E) Colony formation assays of asiaticoside in QGY-7703 and Bel-7402 cells. (F) Statistical analysis of colony formation assays. Data are presented as mean±SD, which are representative of at least 3 independent experiments. 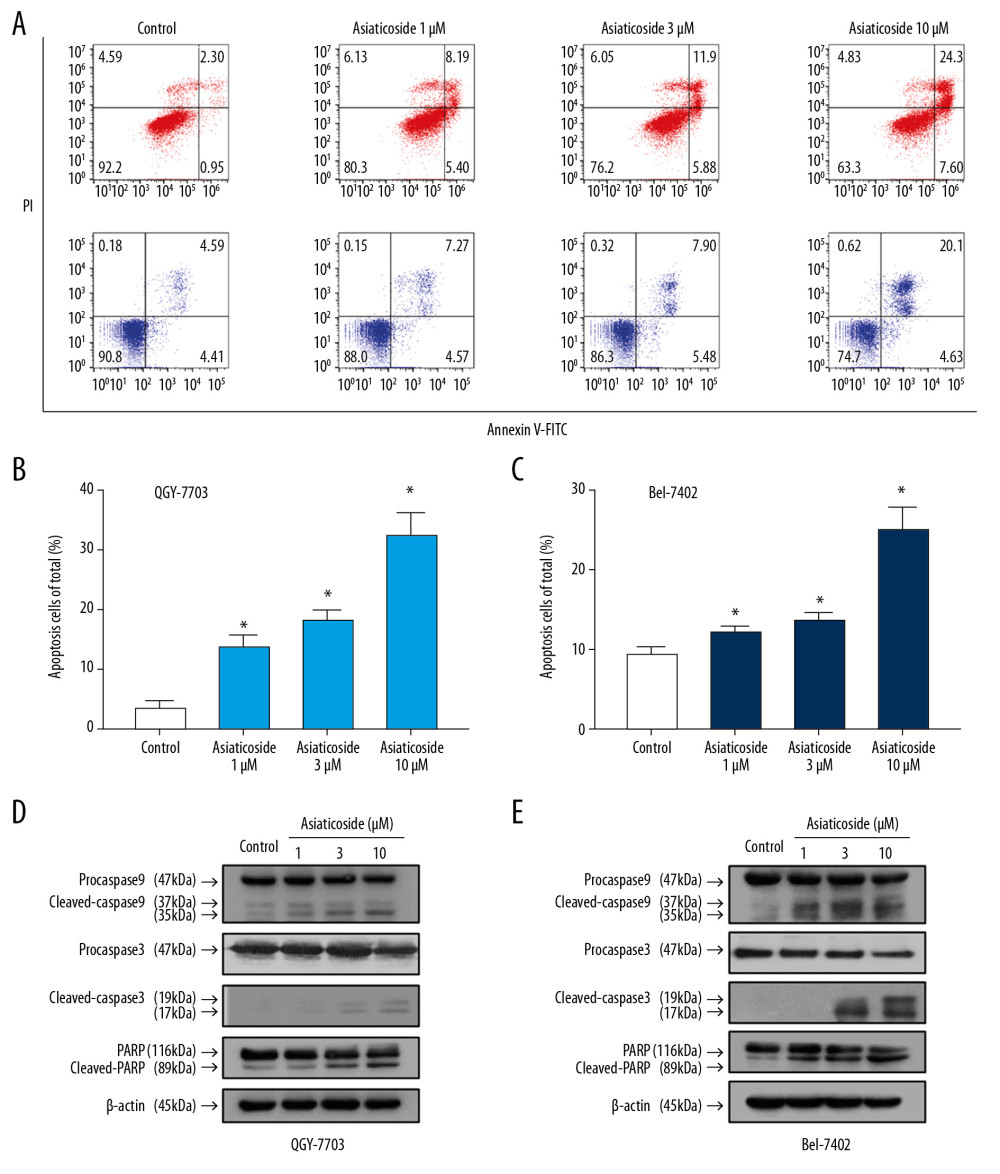 Figure 2. Asiaticoside induced apoptosis in HCC cells. (A) Dot plot of apoptotic cells in QGY-7703 and Bel-7402 cells conducted by flow cytometry. The “red” dot plots showed the results in QGY-7703 cells, while the “blue” dot plot showed the results in Bel-7402 cells. (B) The percentage of apoptotic cells in QGY-7703 cells after treatment with asiaticoside. (C) The percentage of apoptotic cells in Bel-7402 cells after treatment with asiaticoside. (D) Apoptotic-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) Apoptotic-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 2. Asiaticoside induced apoptosis in HCC cells. (A) Dot plot of apoptotic cells in QGY-7703 and Bel-7402 cells conducted by flow cytometry. The “red” dot plots showed the results in QGY-7703 cells, while the “blue” dot plot showed the results in Bel-7402 cells. (B) The percentage of apoptotic cells in QGY-7703 cells after treatment with asiaticoside. (C) The percentage of apoptotic cells in Bel-7402 cells after treatment with asiaticoside. (D) Apoptotic-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) Apoptotic-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments. 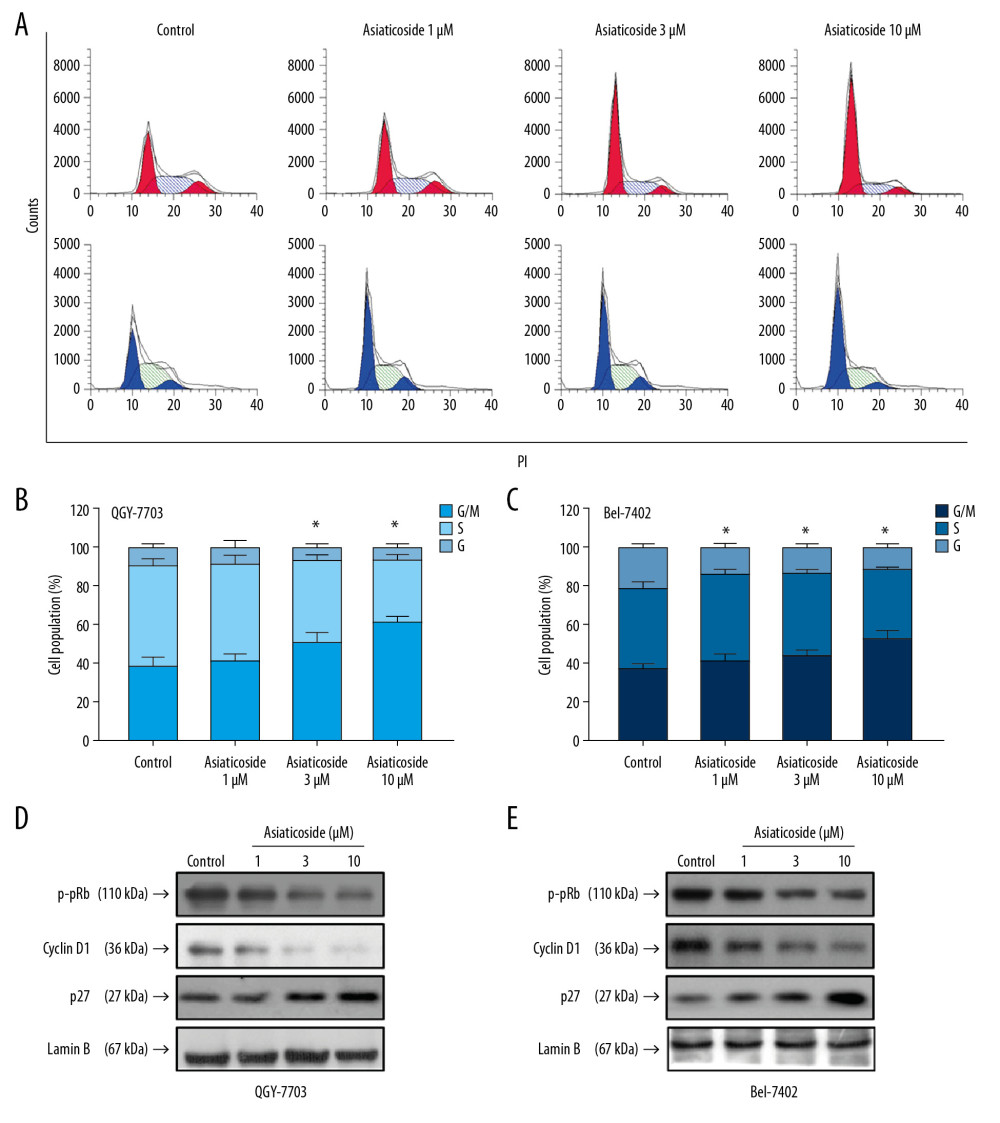 Figure 3. Asiaticoside induced G1 phase cell cycle arrest in HCC cells. (A) The peak plot showed results of cell cycle analysis for QGY-7703 cells (red) and Bel-7402 cells (blue) after treatment with asiaticoside. (B) The percentages of the QGY-7703 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (C) The percentages of the Bel-7402 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (D) G1 cell cycle-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) G1 cell cycle-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 3. Asiaticoside induced G1 phase cell cycle arrest in HCC cells. (A) The peak plot showed results of cell cycle analysis for QGY-7703 cells (red) and Bel-7402 cells (blue) after treatment with asiaticoside. (B) The percentages of the QGY-7703 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (C) The percentages of the Bel-7402 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (D) G1 cell cycle-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) G1 cell cycle-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments. 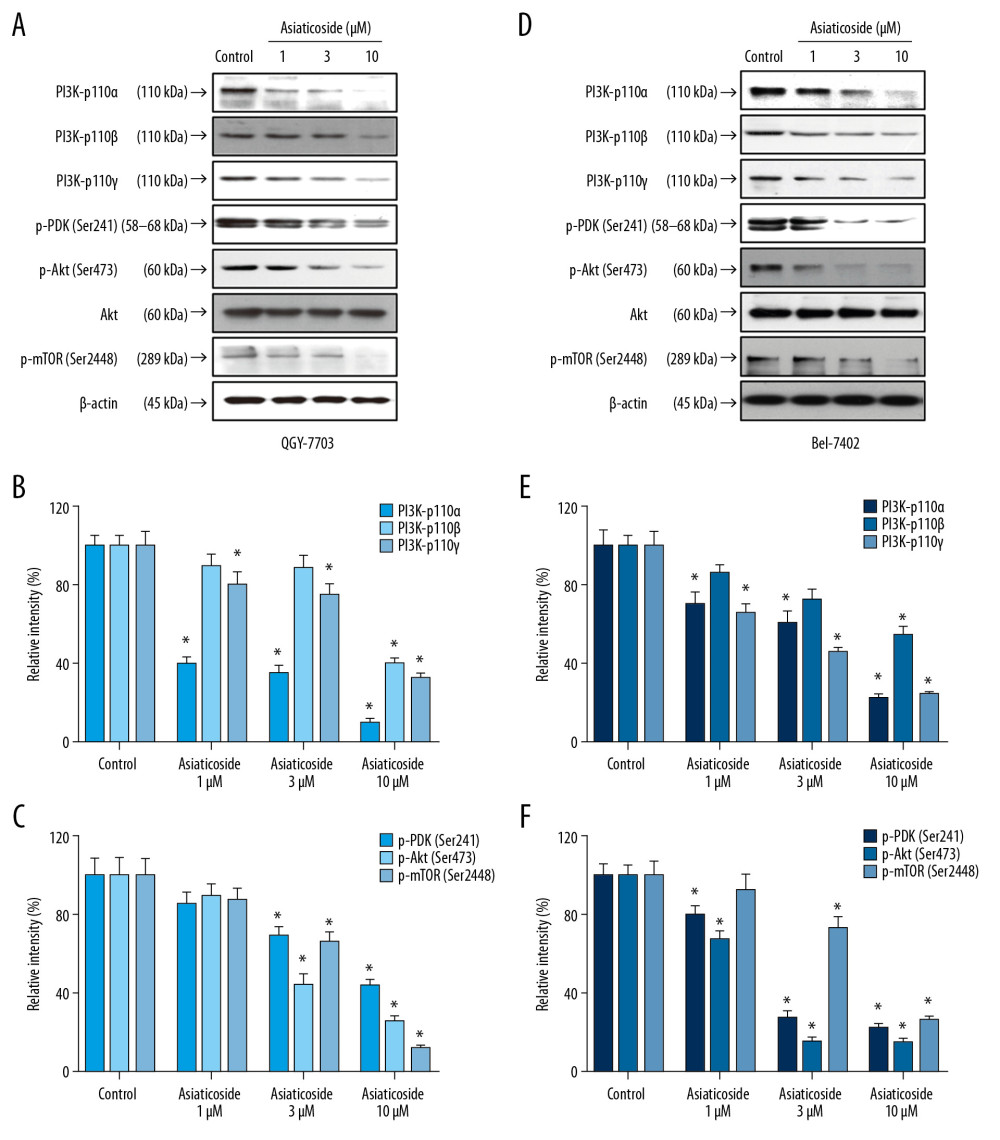 Figure 4. Asiaticoside inhibited PI3K/Akt pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B, C) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in QGY-7703 cells. (D) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (E, F) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 4. Asiaticoside inhibited PI3K/Akt pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B, C) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in QGY-7703 cells. (D) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (E, F) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments. 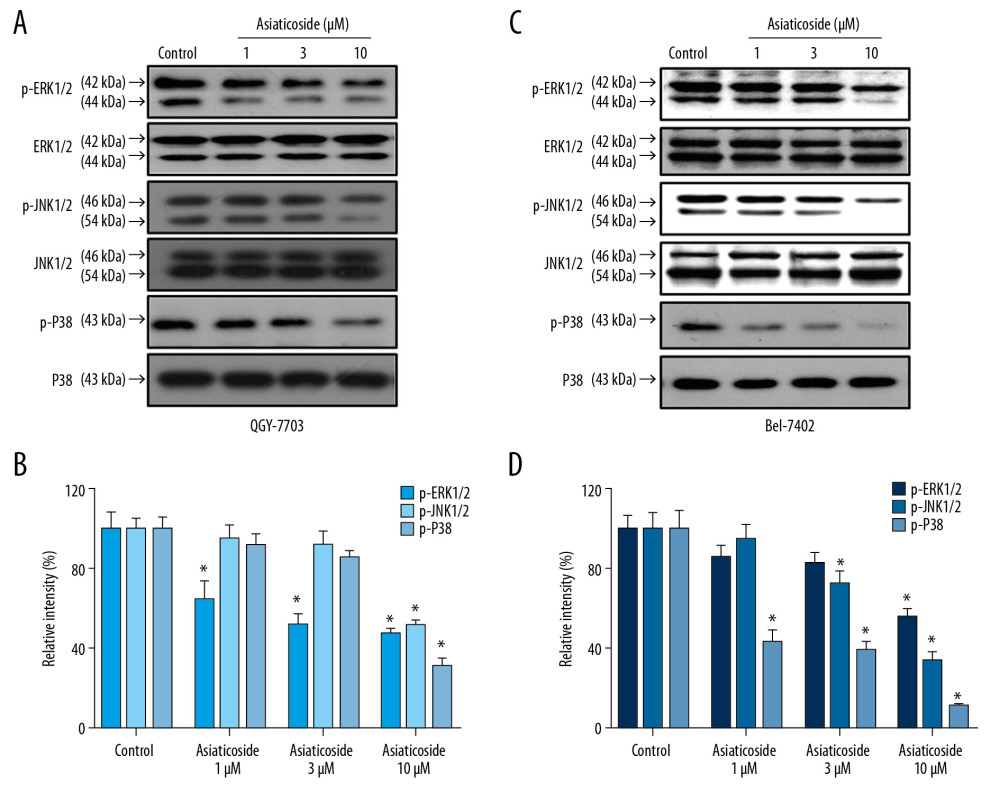 Figure 5. Asiaticoside inhibited MAKPs pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in QGY-7703 cells. (C) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (D) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 5. Asiaticoside inhibited MAKPs pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in QGY-7703 cells. (C) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (D) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments. 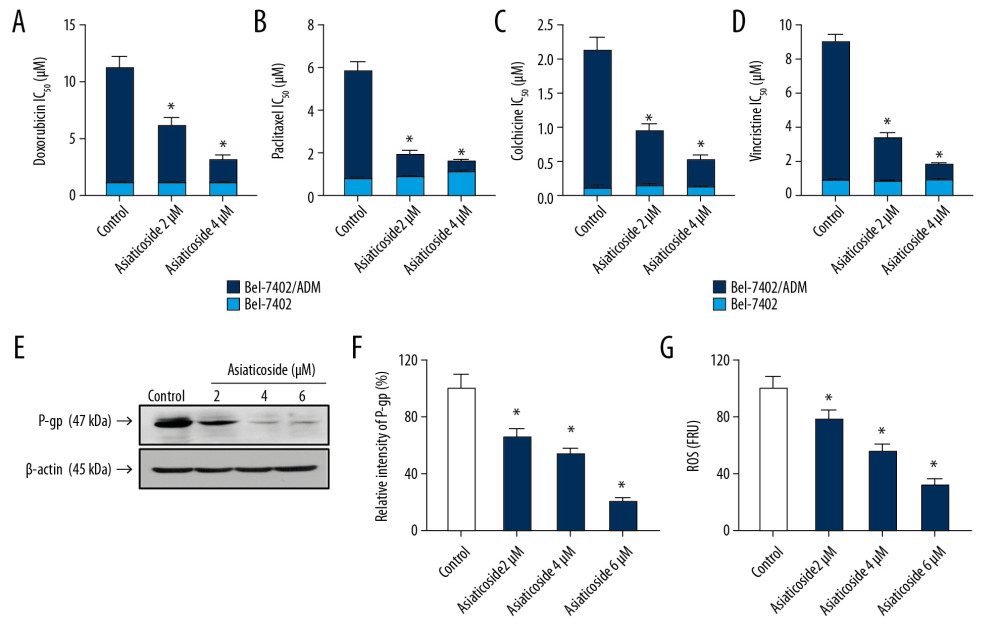 Figure 6. Asiaticoside antagonized P-gp-mediated MDR in HCC cells by decreasing the expression level of P-gp through decreasing the intracellular ROS level. The colored bar plot shows the IC50 values of doxorubicin (A), paclitaxel (B), colchicine (C), and vincristine (D) after treatment with indicated concentrations of asiaticoside in parental Bel-7402 cells as well as chemoresistance of Bel-7402/ADM cells. Western blot assays showed the inhibitory effect of asiaticoside on P-gp protein expression (E), and relative intensity of P-gp after treatment with asiaticoside in Bel-7402/ADM cells is shown in (F). The intracellular ROS level in Bel-7402/ADM cells after treatment with asiaticoside for 48 h (G). Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 6. Asiaticoside antagonized P-gp-mediated MDR in HCC cells by decreasing the expression level of P-gp through decreasing the intracellular ROS level. The colored bar plot shows the IC50 values of doxorubicin (A), paclitaxel (B), colchicine (C), and vincristine (D) after treatment with indicated concentrations of asiaticoside in parental Bel-7402 cells as well as chemoresistance of Bel-7402/ADM cells. Western blot assays showed the inhibitory effect of asiaticoside on P-gp protein expression (E), and relative intensity of P-gp after treatment with asiaticoside in Bel-7402/ADM cells is shown in (F). The intracellular ROS level in Bel-7402/ADM cells after treatment with asiaticoside for 48 h (G). Data are presented as mean±SD, which are representative of at least 3 independent experiments. References
1. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2019: Cancer J Clin, 2019; 69(1); 7-34
2. Li Z, Wang Y, Duan S, Expression of TBX3 in hepatocellular carcinoma and its clinical implication: Med Sci Monit, 2018; 24; 9324-33
3. Lang H, Heinrich S, Bartsch FSurgical treatment of hepatic tumors-liver resection and transplantation: Internist (Berl), 2020; 61(2); 147-57 [in German]
4. Forner A, Llovet JM, Bruix J, Hepatocellular carcinoma: Lancet, 2012; 379(9822); 1245-55
5. Chen W, Zheng R, Baade PD, Cancer statistics in China, 2015: Cancer J Clin, 2016; 66(2); 115-32
6. Chen W, Zheng R, Zhang S, Cancer incidence and mortality in China in 2013: An analysis based on urbanization level: Chin J Cancer Res, 2017; 29(1); 1-10
7. De Rosa M, Pace U, Rega D, Genetics, diagnosis and management of colorectal cancer (review): Oncol Rep, 2015; 34(3); 1087-96
8. Lohitesh K, Chowdhury R, Mukherjee S, Resistance a major hindrance to chemotherapy in hepatocellular carcinoma: An insight: Cancer Cell Int, 2018; 18; 44
9. Stonik VA, Fedorov SN, Marine low molecular weight natural products as potential cancer preventive compounds: Mar Drugs, 2014; 12(2); 636-71
10. Raffa D, Maggio B, Raimondi MV, Recent discoveries of anticancer flavonoids: Eur J Med Chem, 2017; 142; 213-28
11. Lichota A, Gwozdzinski K, Anticancer activity of natural compounds from plant and marine environment: Int J Mol Sci, 2018; 19(11); 3533
12. He L, Hong G, Zhou L, Asiaticoside, a component of Centella asiatica attenuates RANKL-induced osteoclastogenesis via NFATc1 and NF-kappaB signaling pathways: J Cell Physiol, 2019; 234(4); 4267-76
13. Jiang JZ, Ye J, Jin GY, Asiaticoside mitigates the allergic inflammation by abrogating the degranulation of mast cells: J Agric Food Chem, 2017; 65(37); 8128-35
14. Wang X, Cai X, Wang W, Effect of asiaticoside on endothelial cells in hypoxiainduced pulmonary hypertension: Mol Med Rep, 2018; 17(2); 2893-900
15. Duggina P, Kalla CM, Varikasuvu SR, Protective effect of centella triterpene saponins against cyclophosphamide-induced immune and hepatic system dysfunction in rats: Its possible mechanisms of action: J Physiol Biochem, 2015; 71(3); 435-54
16. Yingchun L, Huihan W, Rong Z, Antitumor activity of asiaticoside against multiple myeloma drug-resistant cancer cells is mediated by autophagy induction, activation of effector caspases, and inhibition of cell migration, invasion, and STAT-3 signaling pathway: Med Sci Monit, 2019; 25; 1355-61
17. Kwon KJ, Bae S, Kim K, Asiaticoside, a component of Centella asiatica, inhibits melanogenesis in B16F10 mouse melanoma: Mol Med Rep, 2014; 10(1); 503-7
18. Rahman AA, Makpol S, Jamal R, Tocotrienol-rich fraction, [6]-gingerol and epigallocatechin gallate inhibit proliferation and induce apoptosis of glioma cancer cells: Molecules, 2014; 19(9); 14528-41
19. Al-Saeedi FJ, Study of the cytotoxicity of asiaticoside on rats and tumour cells: BMC Cancer, 2014; 14; 220
20. Markham A, Alpelisib: First global approval: Drugs, 2019; 79(11); 1249-53
21. Bahrami A, Khazaei M, Hasanzadeh M, Therapeutic potential of targeting PI3K/AKT pathway in treatment of colorectal cancer: Rational and progress: J Cell Biochem, 2018; 119(3); 2460-69
22. Zirlik K, Veelken H, Idelalisib: Recent Results Cancer Res, 2018; 212; 243-64
23. Yuan R, Hou Y, Sun W, Natural products to prevent drug resistance in cancer chemotherapy: A review: Ann NY Acad Sci, 2017; 1401(1); 19-27
24. Parvez MK, Arbab AH, Al-Dosari MS, Al-Rehaily AJ, Antiviral natural products against chronic hepatitis B: Recent developments: Curr Pharm Des, 2016; 22(3); 286-93
25. Llovet JM, Zucman-Rossi J, Pikarsky E, Hepatocellular carcinoma: Nat Rev Dis Primers, 2016; 2; 16018
26. Liu X, Li J, Qu X, Clinical outcomes for systemic corticosteroids versus vincristine in treating kaposiform hemangioendothelioma and tufted angioma: Medicine (Baltimore), 2016; 95(20); e3431
27. Stage TB, Bergmann TK, Kroetz DL, Clinical pharmacokinetics of paclitaxel monotherapy: An updated literature review: Clin Pharmacokinet, 2018; 57(1); 7-19
28. Razali NNM, Ng CT, Fong LY, Cardiovascular protective effects of centella asiatica and its triterpenes: a review: Planta Med, 2019; 85(16); 1203-15
29. Park BC, Bosire KO, Lee ES, Asiatic acid induces apoptosis in SK-MEL-2 human melanoma cells: Cancer Lett, 2005; 218(1); 81-90
30. Ren L, Cao QX, Zhai FR, Asiatic acid exerts anticancer potential in human ovarian cancer cells via suppression of PI3K/Akt/mTOR signalling: Pharm Biol, 2016; 54(11); 2377-82
31. Cui Q, Ren J, Zhou Q, Effect of asiatic acid on epithelial-mesenchymal transition of human alveolar epithelium A549 cells induced by TGF-beta1: Oncol Lett, 2019; 17(5); 4285-92
32. Sakonsinsiri C, Kaewlert W, Armartmuntree N, Anti-cancer activity of asiatic acid against human cholangiocarcinoma cells through inhibition of proliferation and induction of apoptosis: Cell Mol Biol (Noisy-le-grand), 2018; 64(10); 28-33
33. Xi X, Liu N, Wang Q, ACT001, a novel PAI-1 inhibitor, exerts synergistic effects in combination with cisplatin by inhibiting PI3K/AKT pathway in glioma: Cell Death Dis, 2019; 10(10); 757
34. Ruggiero CF, Malpicci D, Fattore L, ErbB3 phosphorylation as central event in adaptive resistance to targeted therapy in metastatic melanoma: Early detection in CTCs during therapy and insights into regulation by autocrine neuregulin: Cancers (Basel), 2019; 11(10); 1425
35. Jin Y, Liu M, Sa R, Mouse models of thyroid cancer: Bridging pathogenesis and novel therapeutics: Cancer Lett, 2020; 469; 35-53
36. Noorolyai S, Shajari N, Baghbani E, The relation between PI3K/AKT signalling pathway and cancer: Gene, 2019; 698; 120-28
37. Ji N, Yang Y, Cai CY, Selonsertib (GS-4997), an ASK1 inhibitor, antagonizes multidrug resistance in ABCB1- and ABCG2-overexpressing cancer cells: Cancer Lett, 2019; 440–441; 82-93
38. Cao B, Li M, Zha W, Metabolomic approach to evaluating adriamycin pharmacodynamics and resistance in breast cancer cells: Metabolomics, 2013; 9(5); 960-73
Figures
 Figure 1. The inhibitory effect of asiaticoside on HCC cells proliferation. Concentration-viability curves of asiaticoside on QGY-703 cells (A) and Bel-7402 cells (B) after treatment for 48 h. Asiaticoside (5 μM) inhibited the proliferation of QGY-7703 cells (C) and Bel-7402 cells (D) in a time-dependent manner. (E) Colony formation assays of asiaticoside in QGY-7703 and Bel-7402 cells. (F) Statistical analysis of colony formation assays. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 1. The inhibitory effect of asiaticoside on HCC cells proliferation. Concentration-viability curves of asiaticoside on QGY-703 cells (A) and Bel-7402 cells (B) after treatment for 48 h. Asiaticoside (5 μM) inhibited the proliferation of QGY-7703 cells (C) and Bel-7402 cells (D) in a time-dependent manner. (E) Colony formation assays of asiaticoside in QGY-7703 and Bel-7402 cells. (F) Statistical analysis of colony formation assays. Data are presented as mean±SD, which are representative of at least 3 independent experiments. Figure 2. Asiaticoside induced apoptosis in HCC cells. (A) Dot plot of apoptotic cells in QGY-7703 and Bel-7402 cells conducted by flow cytometry. The “red” dot plots showed the results in QGY-7703 cells, while the “blue” dot plot showed the results in Bel-7402 cells. (B) The percentage of apoptotic cells in QGY-7703 cells after treatment with asiaticoside. (C) The percentage of apoptotic cells in Bel-7402 cells after treatment with asiaticoside. (D) Apoptotic-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) Apoptotic-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 2. Asiaticoside induced apoptosis in HCC cells. (A) Dot plot of apoptotic cells in QGY-7703 and Bel-7402 cells conducted by flow cytometry. The “red” dot plots showed the results in QGY-7703 cells, while the “blue” dot plot showed the results in Bel-7402 cells. (B) The percentage of apoptotic cells in QGY-7703 cells after treatment with asiaticoside. (C) The percentage of apoptotic cells in Bel-7402 cells after treatment with asiaticoside. (D) Apoptotic-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) Apoptotic-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments. Figure 3. Asiaticoside induced G1 phase cell cycle arrest in HCC cells. (A) The peak plot showed results of cell cycle analysis for QGY-7703 cells (red) and Bel-7402 cells (blue) after treatment with asiaticoside. (B) The percentages of the QGY-7703 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (C) The percentages of the Bel-7402 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (D) G1 cell cycle-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) G1 cell cycle-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 3. Asiaticoside induced G1 phase cell cycle arrest in HCC cells. (A) The peak plot showed results of cell cycle analysis for QGY-7703 cells (red) and Bel-7402 cells (blue) after treatment with asiaticoside. (B) The percentages of the QGY-7703 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (C) The percentages of the Bel-7402 cells population in G1, S, and G2/M phases after treatment with asiaticoside. (D) G1 cell cycle-related proteins alteration in QGY-7703 cells after treatment with asiaticoside. (E) G1 cell cycle-related proteins alteration in Bel-7402 cells after treatment with asiaticoside. Data are presented as mean±SD, which are representative of at least 3 independent experiments. Figure 4. Asiaticoside inhibited PI3K/Akt pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B, C) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in QGY-7703 cells. (D) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (E, F) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 4. Asiaticoside inhibited PI3K/Akt pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B, C) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in QGY-7703 cells. (D) Protein bands showed alteration of key proteins in PI3K/Akt pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (E, F) The relative intensity of protein expression of PI3K subunits p110α, p110β, and p110γ, and p-PDK, p-Akt, and p-mTOR after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments. Figure 5. Asiaticoside inhibited MAKPs pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in QGY-7703 cells. (C) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (D) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 5. Asiaticoside inhibited MAKPs pathways in QGY-7703 and Bel-7402 cells. (A) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in QGY-7703 cells. (B) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in QGY-7703 cells. (C) Protein bands showed alteration of key proteins in MAKPs pathways after asiaticoside treatment for 48 h in Bel-7402 cells. (D) The relative intensity of protein expression of MAPKs key proteins after treatment with asiaticoside for 48 h in Bel-7402 cells. Data are presented as mean±SD, which are representative of at least 3 independent experiments. Figure 6. Asiaticoside antagonized P-gp-mediated MDR in HCC cells by decreasing the expression level of P-gp through decreasing the intracellular ROS level. The colored bar plot shows the IC50 values of doxorubicin (A), paclitaxel (B), colchicine (C), and vincristine (D) after treatment with indicated concentrations of asiaticoside in parental Bel-7402 cells as well as chemoresistance of Bel-7402/ADM cells. Western blot assays showed the inhibitory effect of asiaticoside on P-gp protein expression (E), and relative intensity of P-gp after treatment with asiaticoside in Bel-7402/ADM cells is shown in (F). The intracellular ROS level in Bel-7402/ADM cells after treatment with asiaticoside for 48 h (G). Data are presented as mean±SD, which are representative of at least 3 independent experiments.
Figure 6. Asiaticoside antagonized P-gp-mediated MDR in HCC cells by decreasing the expression level of P-gp through decreasing the intracellular ROS level. The colored bar plot shows the IC50 values of doxorubicin (A), paclitaxel (B), colchicine (C), and vincristine (D) after treatment with indicated concentrations of asiaticoside in parental Bel-7402 cells as well as chemoresistance of Bel-7402/ADM cells. Western blot assays showed the inhibitory effect of asiaticoside on P-gp protein expression (E), and relative intensity of P-gp after treatment with asiaticoside in Bel-7402/ADM cells is shown in (F). The intracellular ROS level in Bel-7402/ADM cells after treatment with asiaticoside for 48 h (G). Data are presented as mean±SD, which are representative of at least 3 independent experiments. In Press
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








