19 August 2020: Database Analysis
A Predictive Nomogram for Early Mortality in Stage IV Gastric Cancer
Yuqian Feng1ACDE, Kaibo Guo1ABDE, Huimin Jin1BC, Yuying Xiang1D, Yiting Zhang1E, Shanming Ruan2AFG*DOI: 10.12659/MSM.923931
Med Sci Monit 2020; 26:e923931
Abstract
BACKGROUND: The study was intended to establish predictive nomogram models for predicting total early mortality (the probability of surviving less than or equal to 3 months) and cancer-specific early mortality in patients with stage IV gastric cancer. This was the first study to establish prognostic survival in patients with stage IV gastric cancer.
MATERIAL AND METHODS: Patients from the SEER database were identified using inclusion and exclusion criteria. Their clinical characteristics were statistically analyzed. The Kaplan-Meier method and the log-rank test were used to compare the influences of different factors on survival time. Logistic regression models were conducted to explore the correlative factors of early mortality. A nomogram was established based on factors significant in the logistic regression model and an internal validation was performed.
RESULTS: Of the 11,036 eligible patients included in the study, 4932(44.7%) patients resulted in total early death (42.6% died of the cancer and 2.1% died of other reasons). Larger tumor size, poor differentiation, and liver metastasis were positively related to cancer-specific early mortality. Surgery was negatively related to total early mortality and cancer-specific early mortality, while cardia was only negatively associated with total early death. Predictive nomogram models for total early mortality and cancer-specific early mortality have been validated internally. The areas under the receiver operating characteristics curve were 73.5%, and 68.0%, respectively, and the decision curve analysis also proved the value of the models.
CONCLUSIONS: The nomogram models proved to be a suitable tool for predicting the early mortality in stage IV gastric cancer.
Keywords: Mortality, Premature, nomograms, Aged, 80 and over, SEER Program
Background
Globally, gastric cancer ranks fifth and third in cancer incidence and mortality, respectively. There were more than 1.03 million newly diagnosed gastric cancer cases worldwide and approximately 783,000 death cases in 2018. Gastric cancer morbidity is the highest in eastern Asia [1]. Distant metastases to gastric cancer are common at the time of diagnosis [2]. Metastatic disease is found at the initial diagnosis of 35% to 40% of gastric cancer patients, and 4% to 14% of these have metastatic disease in the liver, followed by the lung, bone, and brain [3,4]. Many factors such as age, tumor location, tumor size, TNM (Tumor-Node-Metastasis), and surgery, affect the prognosis of cancer.
At present, the prognosis of solid cancer is decided by the American Joint Committee on Cancer (AJCC) TNM staging system [5,6]. However, the existing TNM staging does not reflect tumor prognosis well [7,8]. Based on this system, we cannot evaluate the prognosis between patients with stage IV gastric cancer. Therefore, we need to develop a new prognosis prediction model to accurately individualize the early mortality between advanced cancer patients. Large sample studies have been rarely performed, and are urgently needed at present.
This study was based on information about patients with stage IV gastric cancer from the Surveillance, Epidemiology, and End Results (SEER) database to analyze demographic and clinical characteristics, evaluate early mortality, and examine the risk factors of early death when first diagnosed. In addition, the work has produced a predictive nomogram that contained relevant factors for predicting early mortality and internal validation was performed to test the accuracy of the predictive model.
Material and Methods
DATA:
Data was obtained from the SEER database, which provides the cancer relevant factors and survival outcomes from established cancer registries across approximately one-third of the United States population. The database includes information about the clinical characteristics and survival outcomes for different cancer patients. It has certain standards for patient data collection, therefore, its accuracy is guaranteed. SEER*Stat Software version 8.3.5 (
STUDY POPULATION:
This was a population-based cohortstudy. The SEER database did not collect data on organ metastases until 2010. Therefore, the study included patients diagnosed with gastric cancer between 2010 and 2016 (with at least 3 months follow-up). Patients with non-primary tumors, T0, Tis, M0, dead at diagnosis, ambiguous survival time, unknown cause of death, failed to be followed up, or ≤18 years old were excluded. Inclusion criteria were patients with stage IV gastric cancer confirmed at the initial diagnosis (Figure 1). When cancer patients died within 3 months of initial diagnosis, it was defined as early death [9,10].
STATISTICAL ANALYSIS:
The quantitative data involved in the study were described as mean±standard deviation and comparisons between different groups were analyzed using Student’s
The predictive nomograms for total early mortality, and cancer-specific early mortality were based on the results of regression analysis using the R version 3.6.1 (Lucent Technologies, New Jersey, U.S.). Calibration plots for the nomograms were produced. The reliability of the nomograms was evaluated by the C-index, receiver operating characteristic curve, the area under the curve (AUC), and decision curve analysis (DCA). Due to the lack of relevant models for the early death prognosis of patients with stage IV gastric cancer, the nomogram and internal verification with the existing prognostic criterion could not be compared.
Results
DEMOGRAPHIC AND CLINICAL CHARACTERISTICS:
Information was initially collected from the database for 47,553 gastric cancer patients, and 11,036 eligible patients were included in the study after strict screening for inclusion and exclusion criteria. The average age of patients was 63.60±14.28 years, with 64.2% (N=7081) male and 35.8% (N=3955) female patients. The average age of females with gastric cancer was higher than that of males (64.10±15.76 years vs. 63.32±13.38 years, p<0.001). The majority of patients were Caucasians (71.7%, N=7911), 56.3% patients (N=6208) were married, 32.2% patients had cardia gastric cancer, and 88.0% patients had adenocarcinomas. Among the study population, the percentages of liver, lung, bone and brain metastases were 43.0%, 14.7%, 12.8%, and 2.0%, respectively. Patients’ clinical characteristics are shown in Table 1.
INCIDENCE OF EARLY DEATH:
In the present study, 4932 (44.7%) gastric cancer patients had total early deaths, where 4697 (42.6%) patients died of the cancer and 235 (2.1%) patients died of other reasons. Early mortality for males with gastric cancer was higher than females, however, there was no statistical difference (28.6% vs. 16.1%, χ2=0.19, p>0.05). The incidence of total early death fluctuated significantly with age. Early mortality in patients aged 18–40 years decreased with age, while early mortality increased for the other age groups. Trends in early mortality were roughly the same for males and females in the various age groups (Figure 2A).
Early mortality varied with the location of primary tumors. In females, the lowest early mortality was for tumors in the lesser curvature of the stomach (38.6%), followed by the body of the stomach (39.7%) and cardia (40.3%). However, the tumors of the greater curvature of the stomach (49.1%) and fundus of the stomach (48.6%) contributed to higher early mortality. In males, the lowest early mortality was seen in patients with tumors in the cardia (37.8%). However, patients with tumors in the greater curvature of the stomach (52.0%), overlapping lesion of the stomach (48.5%), and body of the stomach (48.1%) contributed to higher early mortality. The cardia cancer (38.3%) presented significantly lower early mortality than the non-cardia cancer (45.0%) (χ2=38.98, p< 0.001) (Figure 2B).
Gastric cancer patients with brain metastases had the highest early mortality (59.0%), followed by lung (53.8%), bone (52.4%), and liver metastases (49.6%). In females, early mortality due to brain metastases (68.3%) was higher than that in males (55.4%) and the total group (59.0%) (Figure 2C).
The early mortality of gastric cancer patients was positively correlated with the number of metastatic organ sites (χ2=164.29, p<0.001). Male patients presented with similar results (χ2=81.435, p<0.001). For males, early mortality in four organ metastases (66.7%) was higher than three organ metastases (58.9%). However, for females, early mortality in four organ metastases (60.0%) was slightly lower than three organ metastases (61.1%) (Figure 2D).
The median survival time of different age groups varied. Patients older than 85 years had the shortest median survival time (p<0.001) compared with the other listed age groups (Figure 3A). Amongst the study population, the median survival time of non-cardia cancer was significantly shorter than patients with cardia gastric cancer (p<0.001) (Figure 3B). Patients with liver metastases had significantly shorter survival times than patients without liver metastases. Similar results were seen for lung metastases (p<0.001) (Figure 3C, 3D). Compared to patients with a lower histological grade, those with a higher histological grade had worse survival prognosis (p<0.001) (Figure 3E). Surgical treatment significantly extended patients’ survival time (p<0.001) (Figure 3F).
FACTORS ASSOCIATED WITH EARLY DEATH:
Univariate logistic regression showed advanced age, marital status, higher T stages, and liver, and lung metastases were all closely related to the total early death, cancer-specific early death and non-cancer-specific early death. Poor differentiation, higher N stages, bone metastases, and surgery were only related to total early death and cancer-specific early death (Table 1).
After incorporating the significant factors into multivariable logistic regression, the results showed that advanced age, primary site, poor differentiation, liver metastases, lung metastases, and surgery were significantly related to total early death. While tumor size, poor differentiation, liver metastases, and surgery were only significantly related to cancer-specific early death. Poor differentiation, liver metastases, and surgery were significantly related to total early death and cancer-specific early death (Table 2).
ESTABLISHMENT OF NOMOGRAMS FOR PREDICTING EARLY MORTALITY:
Based on the previously mentioned factors (age, primary site, tumor size, histological grade, liver metastases, lung metastases, surgery) according to the multivariable model, significant factors related to non-cancer-specific early death were insufficient, only two nomograms were established to predict total early mortality, and cancer-specific early mortality among stage IV gastric cancer patients, respectively. The probability of total early death ranged from 0.05 to 0.90, while cancer-specific early death ranged from 0.10 to 0.95. Therefore, not every total score would have a corresponding probability. The line for the histological grade was the longest in the two prediction models, suggesting that histological grade had the most value in predicting early mortality. In the nomogram for predicting cancer-specific early mortality, surgery and T stage also had great predictive value (Figure 4A, 4B). Internal verification showed that the C-index for the total early mortality nomogram was 0.627 and cancer early mortality was 0.656. The solid lines of the calibration curves approach at a 45°, suggesting accurate prediction by these two models. (Figure 5A, 5B). Moreover, the AUC for the two nomograms were 73.5%, and 68.0%, respectively, exhibiting good discrimination (Figure 5C, 5D). The DCA also proved the value of the two models. The net benefit of our risk models were larger than that in other two scenarios (all screening or none-screening) in a wide range of threshold probabilities (Figure 6A, 6B).
Discussion
LIMITATIONS:
There are several limitations in our study. Firstly, our study only included patients who were initially diagnosed with stage IV gastric cancer, and patients who subsequently developed metastases were not included. The SEER database includes approximately 30% of the total US population only, therefore, the research sample is not extensive enough. Secondly, some factors related to gastric cancer have not been explored and may affect the predictive ability of the nomogram, such as helicobacter pylori, sarcopenia, cachexia, some inflammatory indices, and the Eastern Cooperative Oncology Group performance score. Further studies need to be conducted with consideration of these factors related to gastric cancer. Thirdly, only an internal validation of the nomogram was performed, and external verification is still necessary. We will do our best to validate this prognostic model in future clinical practice.
Conclusions
Based on the aforementioned factors (age, primary site, tumor size, histological grade, liver metastases, lung metastases, surgery), a predictive nomogram was set up. It has a good ability to predict early mortality in patients with stage IV gastric cancer. This model can be widely used in clinical practice, allowing clinicians to develop more personalized treatments for patients with advanced gastric cancer, to give them the best possible prognosis.
Figures
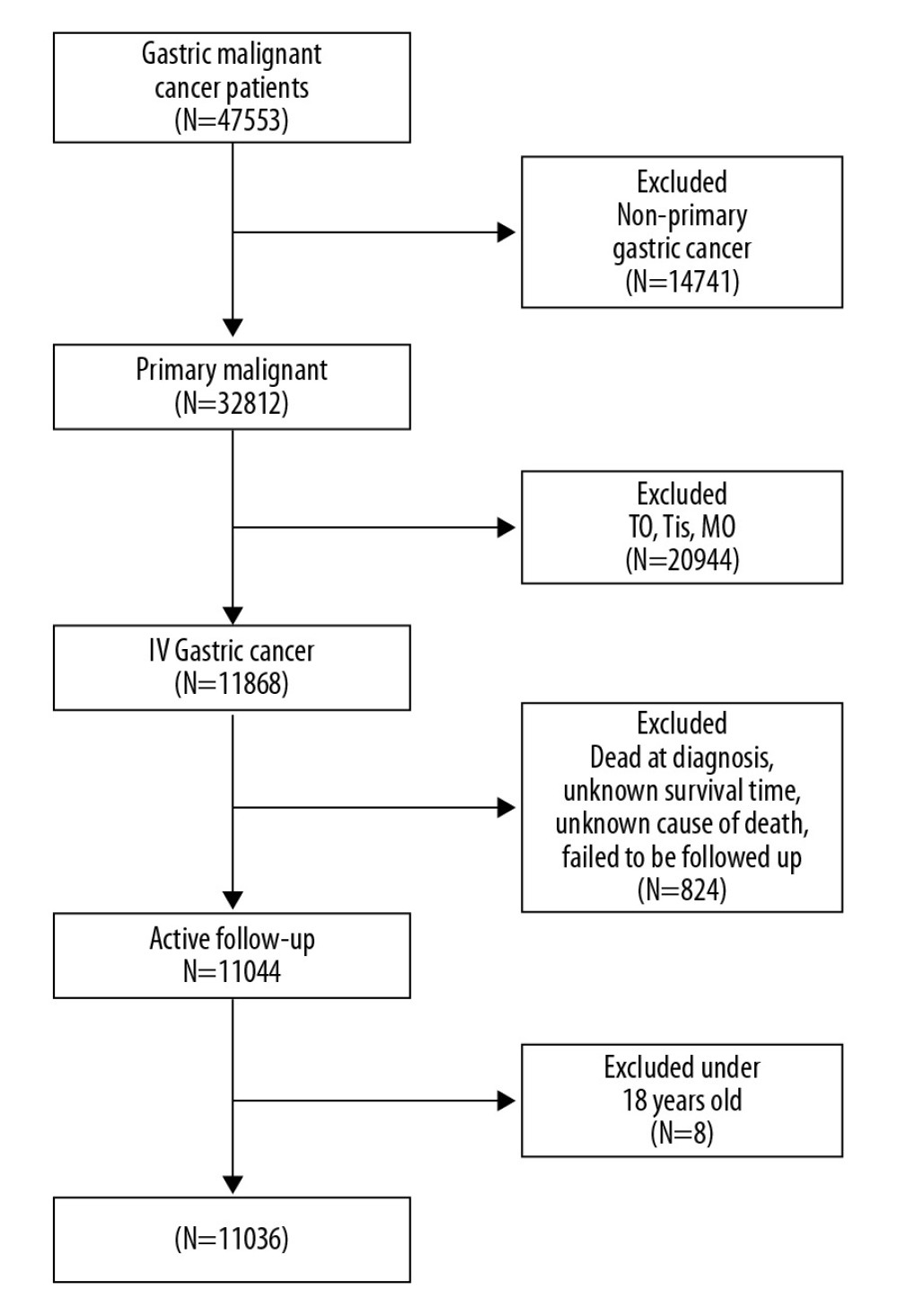 Figure 1. Flowchart for selection of the stage IV gastric cancer patients.
Figure 1. Flowchart for selection of the stage IV gastric cancer patients. 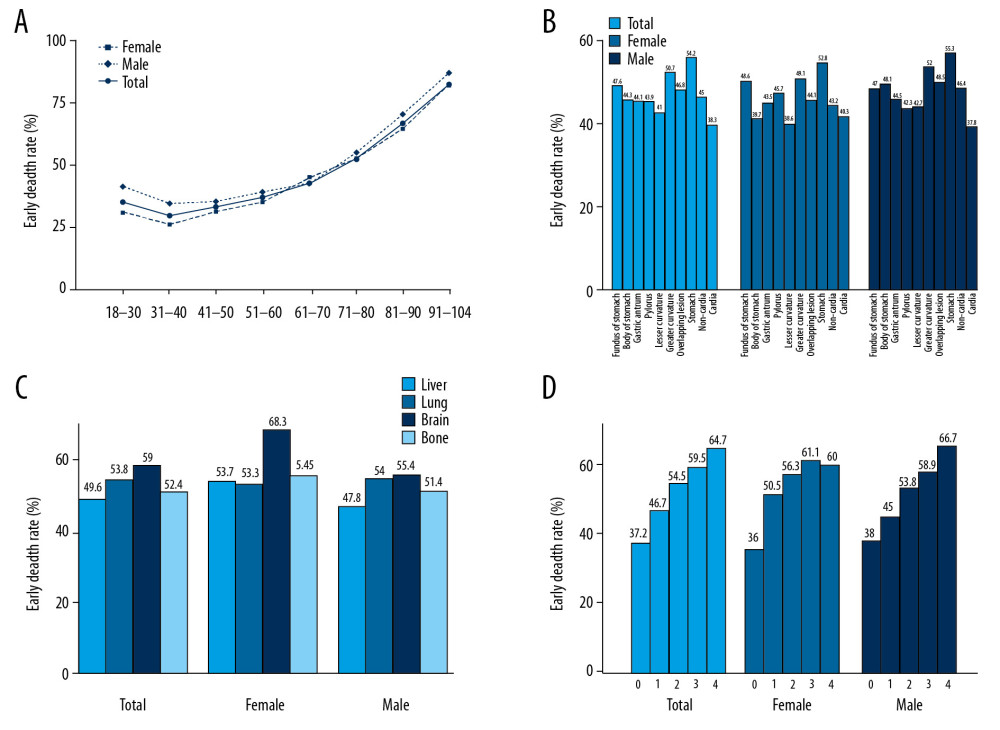 Figure 2. Trend and distribution of early mortality of stage IV gastric cancer patients stratified by: age (A), gastric cancer sites (B), distant metastases by organs (C), number of metastasized organs (D).
Figure 2. Trend and distribution of early mortality of stage IV gastric cancer patients stratified by: age (A), gastric cancer sites (B), distant metastases by organs (C), number of metastasized organs (D). 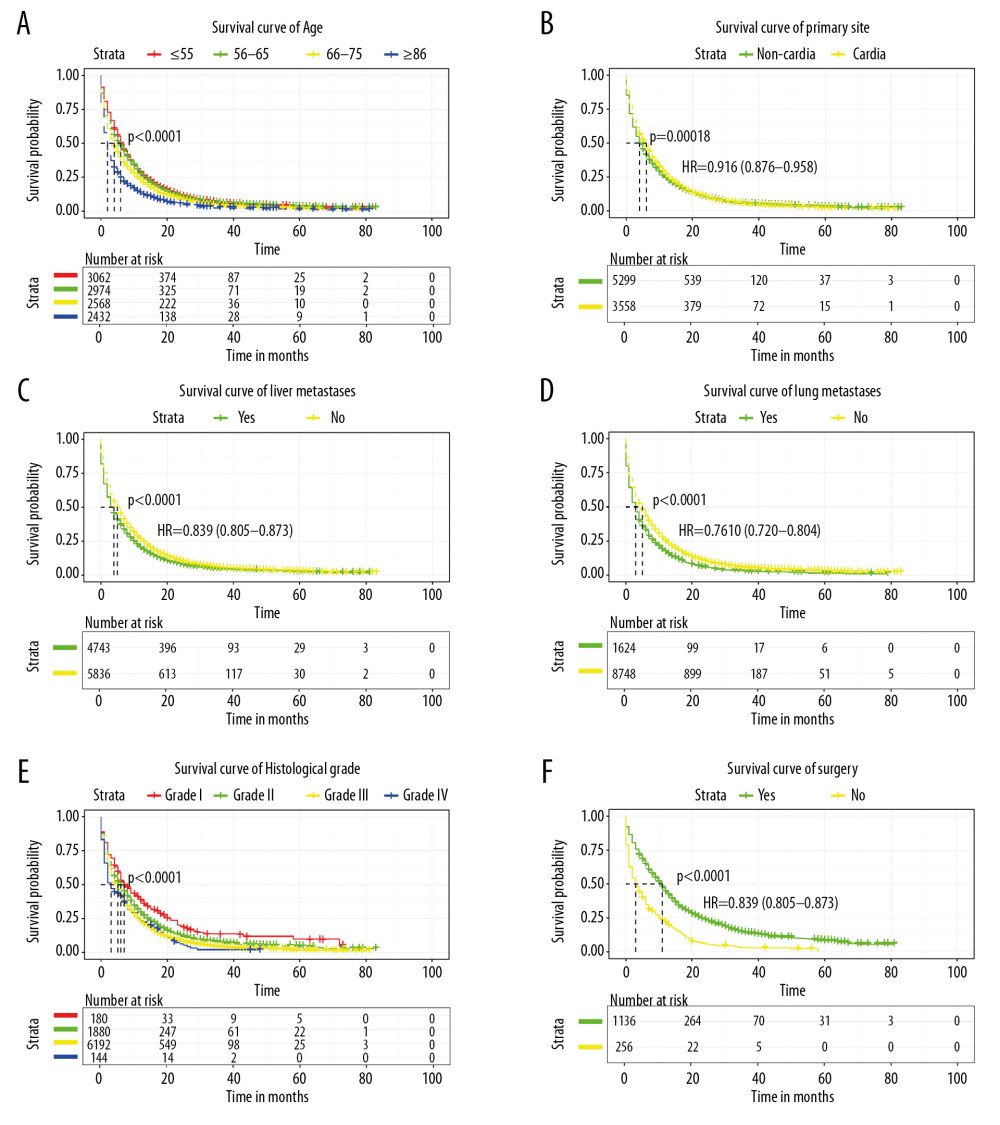 Figure 3. Kaplan-Meier survival curve for (A) age, (B) primary site, (C) liver metastases, (D) lung metastases, (E) histological grade, (F) surgery in stage IV gastric cancer patients.
Figure 3. Kaplan-Meier survival curve for (A) age, (B) primary site, (C) liver metastases, (D) lung metastases, (E) histological grade, (F) surgery in stage IV gastric cancer patients. 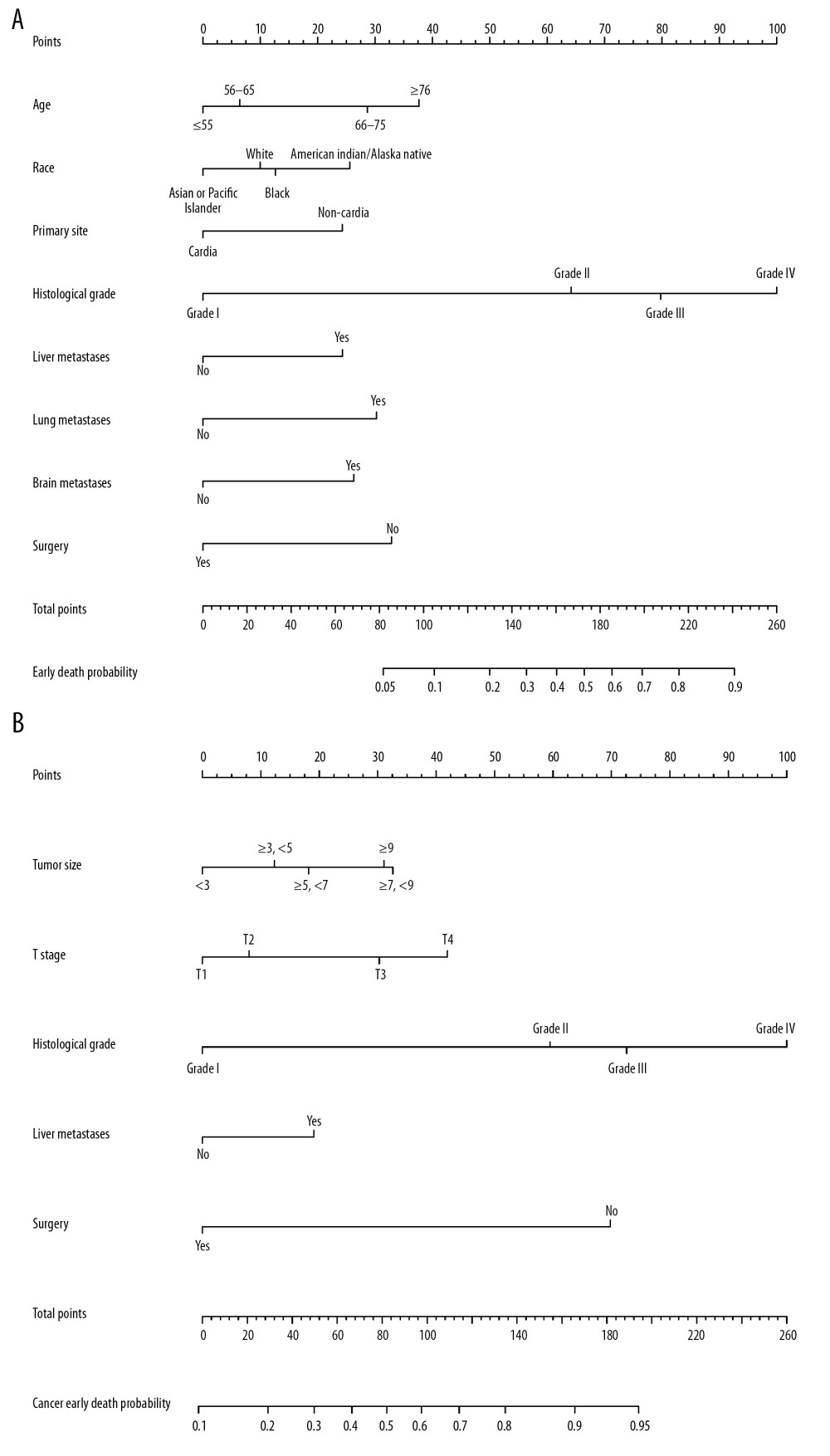 Figure 4. Nomogram for predicting all causes of early mortality (A) and cancer-specific early mortality in stage IV gastric cancer patients (B).
Figure 4. Nomogram for predicting all causes of early mortality (A) and cancer-specific early mortality in stage IV gastric cancer patients (B). 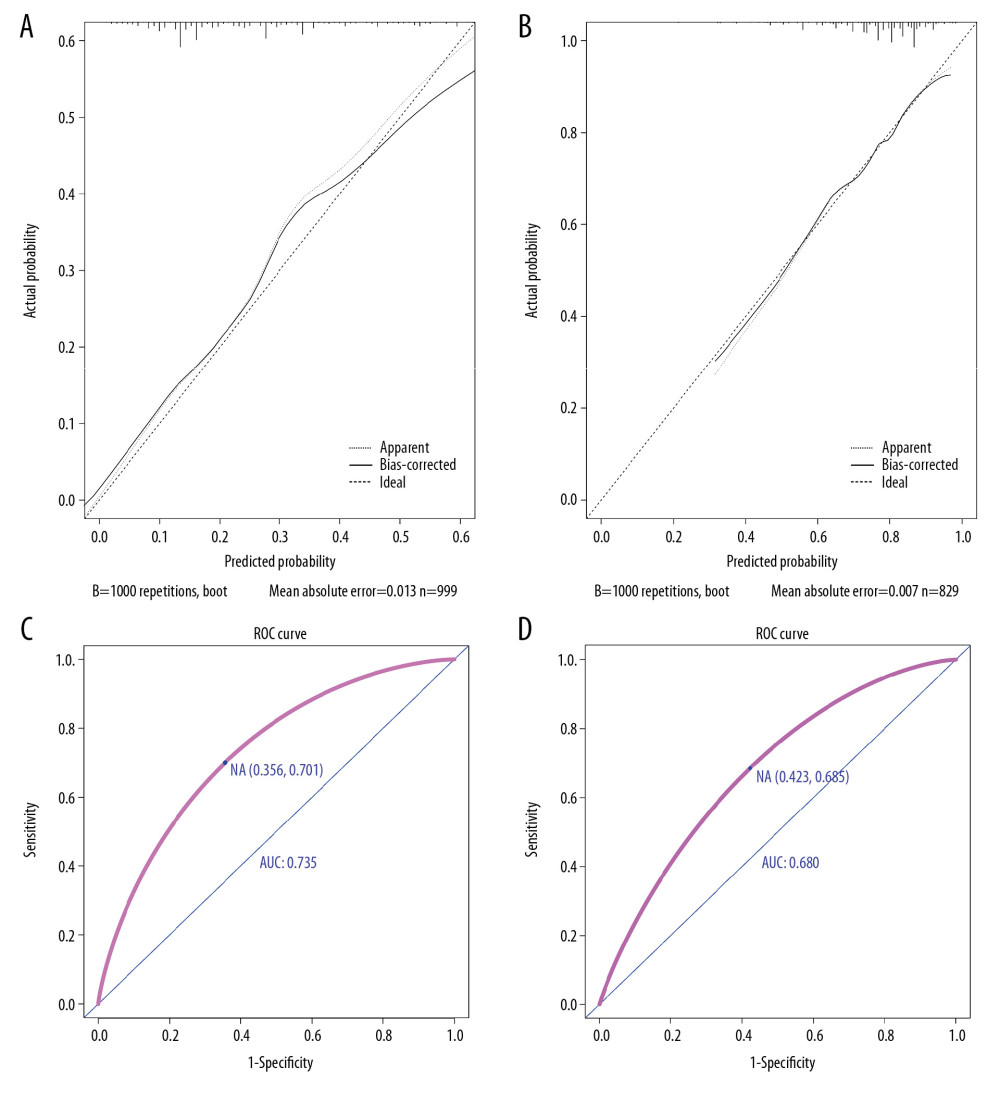 Figure 5. The calibration curve and receiver operating characteristics curve for assessing the calibration and discrimination of the nomogram in predicting all causes of early mortality (A, C) and cancer-specific early mortality (B, D).
Figure 5. The calibration curve and receiver operating characteristics curve for assessing the calibration and discrimination of the nomogram in predicting all causes of early mortality (A, C) and cancer-specific early mortality (B, D). 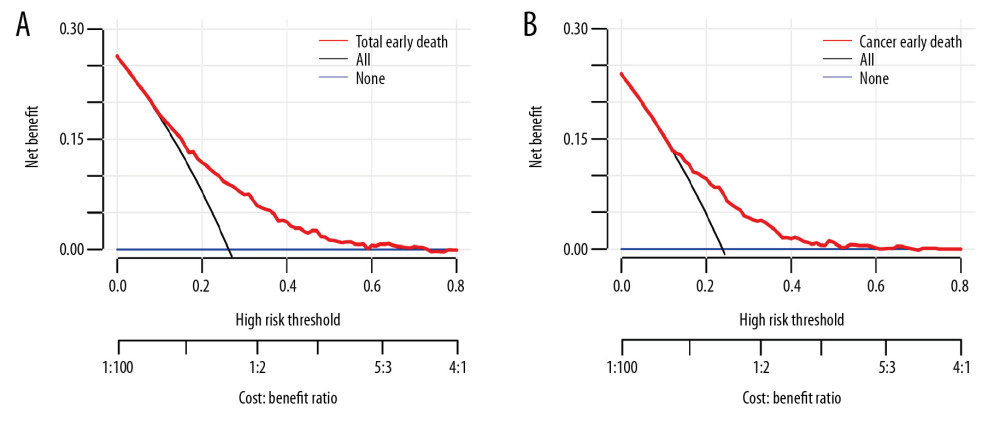 Figure 6. The decision curve analysis for assessing clinical utility of the nomogram in predicting all causes of early mortality (A) and cancer-specific early mortality (B).
Figure 6. The decision curve analysis for assessing clinical utility of the nomogram in predicting all causes of early mortality (A) and cancer-specific early mortality (B). References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68(6); 394-424
2. Rodriguez-de-Santiago E, Hernanz N, Marcos-Prieto HM, A multicentric Spanish study on the characteristics and survival of gastric adenocarcinoma under the age of 60: Gastroenterol Hepatol, 2019; 42(10); 595-603
3. Thomassen I, van Gestel YR, van Ramshorst B, Peritoneal carcinomatosis of gastric origin: A population-based study on incidence, survival and risk factors: Int J Cancer, 2014; 134(3); 622-28
4. Martella L, Bertozzi S, Londero AP, Surgery for liver metastases from gastric cancer: A meta-analysis of observational studies: Medicine, 2015; 94(31); e1113
5. Edge SB, Compton CCThe American Joint Committee on Cancer: Ann Surg Oncol, 2010; 17(6); 1471-74
6. Cao LL, Lu J, Li P, Evaluation of the Eighth Edition of the American Joint Committee on Cancer TNM Staging System for Gastric Cancer: An analysis of 7371 patients in the SEER database: Gastroenterol Res Pract, 2019; 2019 6294382
7. Zhou R, Wu Z, Zhang J, Clinical significance of accurate identification of lymph node status in distant metastatic gastric cancer: Oncotarget, 2016; 7(1); 1029-41
8. Fuse N, Bando H, Chin K, Adjuvant capecitabine plus oxaliplatin after D2 gastrectomy in Japanese patients with gastric cancer: A phase II study: Gastric Cancer, 2017; 20(2); 332-40
9. Jochems A, El-Naqa I, Kessler M, A prediction model for early death in non-small cell lung cancer patients following curative-intent chemoradiotherapy: Acta Oncol, 2018; 57(2); 226-30
10. Inoue T, Tamiya M, Tamiya A, Analysis of early death in Japanese patients with advanced non-small-cell lung cancer treated with nivolumab: Clin Lung Cancer, 2018; 19(2); e171-76
11. Qiu MZ, Shi SM, Chen ZH, Frequency and clinicopathological features of metastasis to liver, lung, bone, and brain from gastric cancer: A SEER-based study: Cancer Med, 2018; 7(8); 3662-72
12. Sun Z, Zheng H, Yu J, Liver metastases in newly diagnosed gastric cancer: A population-based study from SEER: J Cancer, 2019; 10(13); 2991-3005
13. Sleeman J, Steeg PS, Cancer metastasis as a therapeutic target: Eur J Cancer, 2010; 46(7); 1177-80
14. Han J, Tu J, Tang C, Clinicopathological characteristics and prognosis of cT1N0M1 gastric cancer: A population-based study: Dis Markers, 2019; 2019 5902091
15. Yagi Y, Seshimo A, Kameoka S, Prognostic factors in stage IV gastric cancer: Univariate and multivariate analyses: Gastric Cancer, 2000; 3(2); 71-80
16. Wong JU, Tai FC, Huang CC, An examination of surgical and survival outcomes in the elderly (65–79 years of age) and the very elderly (>/=80 years of age) who received surgery for gastric cancer: Current Med Res Opin, 2020; 36(2); 229-33
17. Wang F, Liu X, Mao P, Relationship between the body mass index and tumor site postoperative complications and prognosis in gastric adenocarcinoma: Am Surg, 2018; 84(12); 1861-68
18. Troncone E, Marafini I, Stolfi C, Monteleone G, Transforming growth factor-beta1/Smad7 in intestinal immunity, inflammation, and cancer: Front Immunol, 2018; 9; 1407
19. O’Connell JB, Maggard MA, Livingston EH, Yo CK, Colorectal cancer in the young: Am J Surg, 2004; 187(3); 343-48
20. Zu H, Wang H, Li C, Clinico-pathological features and prognostic analysis of gastric cancer patients in different age groups: Hepatogastroenterology, 2015; 62(137); 225-30
21. Park HJ, Ahn JY, Jung HY, Clinical characteristics and outcomes for gastric cancer patients aged 18–30 years: Gastric Cancer, 2014; 17(4); 649-60
22. Zhao J, Zhao J, Du F, Cardia and non-cardia gastric cancer have similar stage-for-stage prognoses after R0 resection: A large-scale, multicenter study in China: J Gastrointest Surg, 2016; 20(4); 700-7
23. Nguyen TH, Mallepally N, Hammad T: Dig Dis Sci, 2019; 14; 1-9
24. Amini N, Spolverato G, Kim Y, Clinicopathological features and prognosis of gastric cardia adenocarcinoma: A multi-institutional US study: J Surg Oncol, 2015; 111(3); 285-92
25. Saito H, Fukumoto Y, Osaki T, Distinct recurrence pattern and outcome of adenocarcinoma of the gastric cardia in comparison with carcinoma of other regions of the stomach: World J Surg, 2006; 30(10); 1864-69
26. Marchegiani G, Andrianello S, Malleo G, Does size matter in pancreatic cancer?: Reappraisal of tumour dimension as a predictor of outcome beyond the TNM: Ann Surg, 2017; 266(1); 142-48
27. Tran B, Roshan D, Abraham E, The prognostic impact of tumor size in papillary thyroid carcinoma is modified by age: Thyroid, 2018; 28(8); 991-96
28. Wang HM, Huang CM, Zheng CH, Tumor size as a prognostic factor in patients with advanced gastric cancer in the lower third of the stomach: World J Gastroenterol, 2012; 18(38); 5470-75
29. Wang X, Wan F, Pan J, Tumor size: A non-neglectable independent prognostic factor for gastric cancer: J Surg Oncol, 2008; 97(3); 236-40
30. Zhang J, Gold KA, Lin HY, Relationship between tumor size and survival in non-small-cell lung cancer (NSCLC): An analysis of the surveillance, epidemiology, and end results (SEER) registry: J Thorac Oncol, 2015; 10(4); 682-90
31. Dai CY, Lin CY, Tsai PC, Impact of tumor size on the prognosis of hepatocellular carcinoma in patients who underwent liver resection: J Chin Med Assoc, 2018; 81(2); 155-63
32. Bernards N, Creemers GJ, Nieuwenhuijzen GA, No improvement in median survival for patients with metastatic gastric cancer despite increased use of chemotherapy: Ann Oncol, 2013; 24(12); 3056-60
33. Kong JH, Lee J, Yi CA, Lung metastases in metastatic gastric cancer: Pattern of lung metastases and clinical outcome: Gastric Cancer, 2012; 15(3); 292-98
34. Lasithiotakis K, Antoniou SA, Antoniou GA, Gastrectomy for stage IV gastric cancer. a systematic review and meta-analysis: Anticancer Res, 2014; 34(5); 2079-85
Figures
 Figure 1. Flowchart for selection of the stage IV gastric cancer patients.
Figure 1. Flowchart for selection of the stage IV gastric cancer patients. Figure 2. Trend and distribution of early mortality of stage IV gastric cancer patients stratified by: age (A), gastric cancer sites (B), distant metastases by organs (C), number of metastasized organs (D).
Figure 2. Trend and distribution of early mortality of stage IV gastric cancer patients stratified by: age (A), gastric cancer sites (B), distant metastases by organs (C), number of metastasized organs (D). Figure 3. Kaplan-Meier survival curve for (A) age, (B) primary site, (C) liver metastases, (D) lung metastases, (E) histological grade, (F) surgery in stage IV gastric cancer patients.
Figure 3. Kaplan-Meier survival curve for (A) age, (B) primary site, (C) liver metastases, (D) lung metastases, (E) histological grade, (F) surgery in stage IV gastric cancer patients. Figure 4. Nomogram for predicting all causes of early mortality (A) and cancer-specific early mortality in stage IV gastric cancer patients (B).
Figure 4. Nomogram for predicting all causes of early mortality (A) and cancer-specific early mortality in stage IV gastric cancer patients (B). Figure 5. The calibration curve and receiver operating characteristics curve for assessing the calibration and discrimination of the nomogram in predicting all causes of early mortality (A, C) and cancer-specific early mortality (B, D).
Figure 5. The calibration curve and receiver operating characteristics curve for assessing the calibration and discrimination of the nomogram in predicting all causes of early mortality (A, C) and cancer-specific early mortality (B, D). Figure 6. The decision curve analysis for assessing clinical utility of the nomogram in predicting all causes of early mortality (A) and cancer-specific early mortality (B).
Figure 6. The decision curve analysis for assessing clinical utility of the nomogram in predicting all causes of early mortality (A) and cancer-specific early mortality (B). Tables
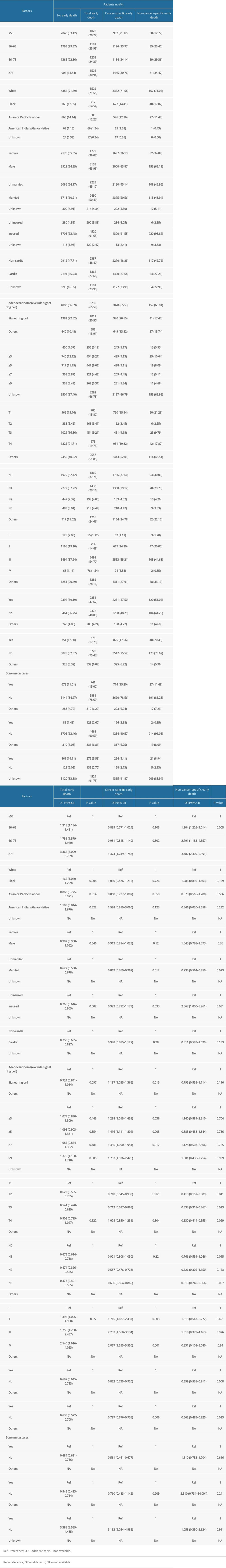 Table 1. Univariable logistic regression for analyzing the risk factors for early death.
Table 1. Univariable logistic regression for analyzing the risk factors for early death.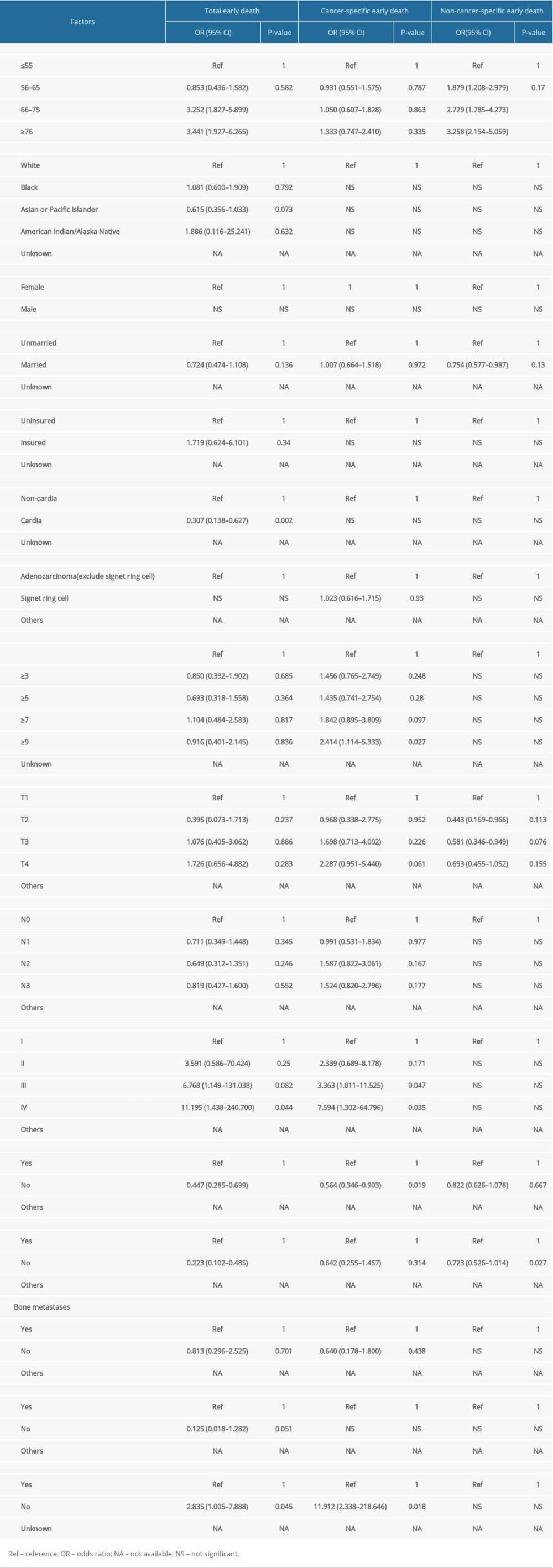 Table 2. Multivariable logistic regression for analyzing the risk factors for early death.
Table 2. Multivariable logistic regression for analyzing the risk factors for early death. Table 1. Univariable logistic regression for analyzing the risk factors for early death.
Table 1. Univariable logistic regression for analyzing the risk factors for early death. Table 2. Multivariable logistic regression for analyzing the risk factors for early death.
Table 2. Multivariable logistic regression for analyzing the risk factors for early death.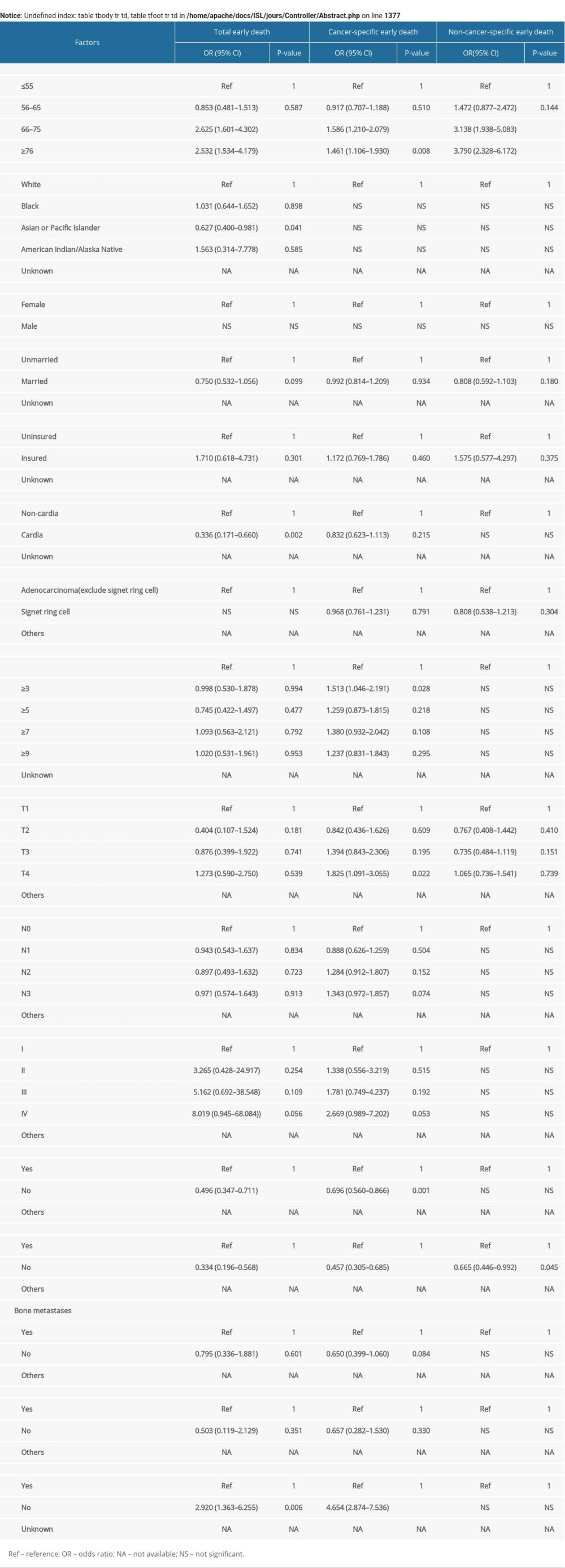 Supplementary Table 1. Multivariable Cox regression for analyzing the risk factors for early death.
Supplementary Table 1. Multivariable Cox regression for analyzing the risk factors for early death. In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








