30 November 2020: Clinical Research
Clinical Outcomes of 130 Patients with Hormone Receptor-Positive and Human Epidermal Growth Factor Receptor 2-Negative Metastatic Breast Cancer Treated with Palbociclib plus Endocrine Therapy and Subsequent Therapy: A Real-World Single-Center Retrospective Study in China
Chang Liu 12CDEF* , Ting Li 12CDEF* , Zhonghua Tao 12B , Jun Cao 12B , Leiping Wang 12B , Jian Zhang 12B , Biyun Wang 12A* , Xichun Hu 12A*DOI: 10.12659/MSM.927187
Med Sci Monit 2020; 26:e927187
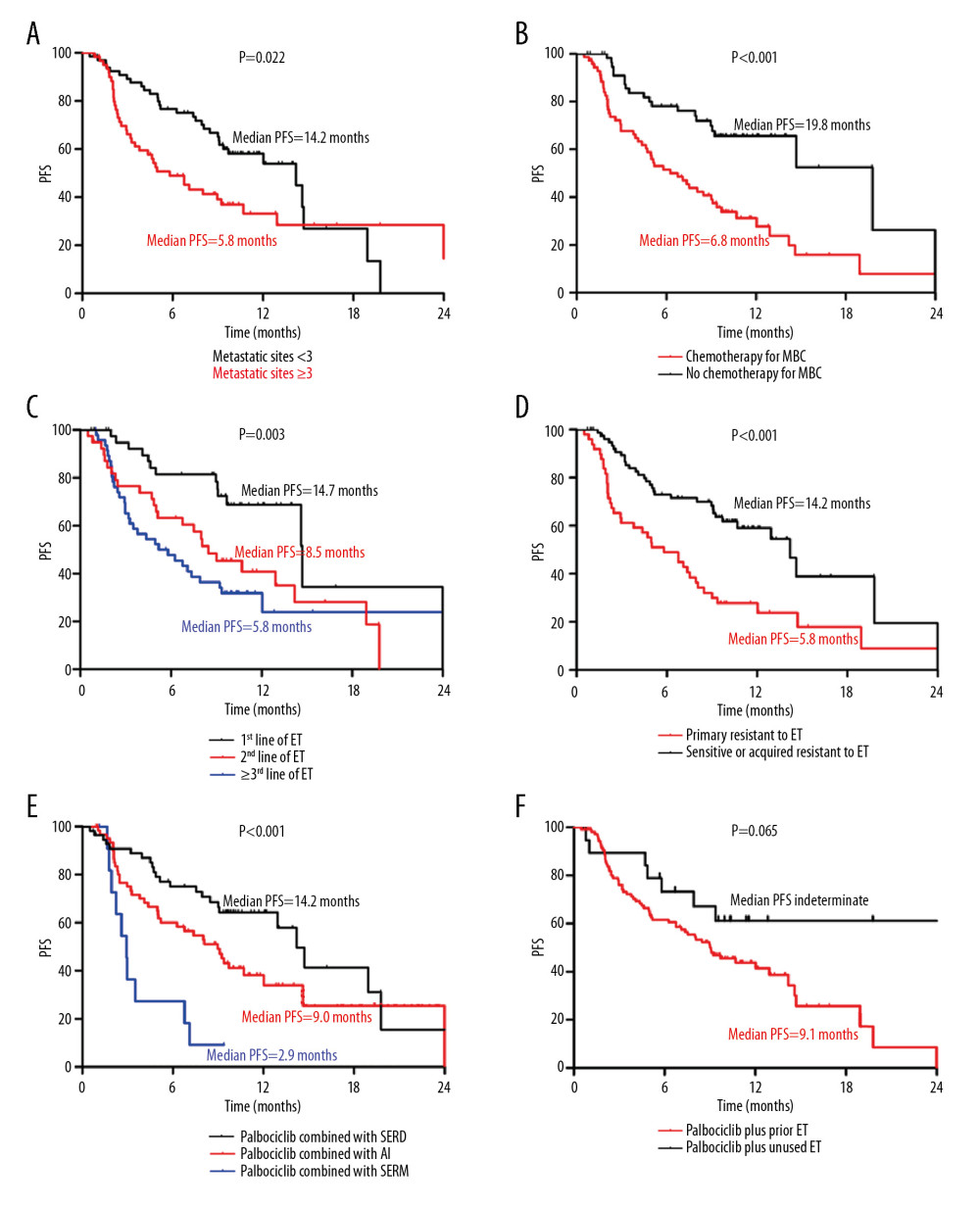
Figure 3 Progression-free survival of palbociclib plus endocrine therapy stratified by patient characteristics. (A) Number of metastatic sites; (B) Whether or not receiving chemotherapy for MBC; (C) Line of palbociclib in ET; (D) response to the most recent ET; (E) Type of combined ET; (F) Palbociclib combined with prior ET or unused ET. Survival curves of PFS were plotted by the Kaplan-Meier method and compared by the log-rank test. P-values of less than 0.05 indicate statistical significance. PFS – progression-free survival; ET – endocrine therapy; MBC – metastatic breast cancer; SERD – selective estrogen receptor degrader; AI – aromatase inhibitor; SERM – selective estrogen receptor modulator.


